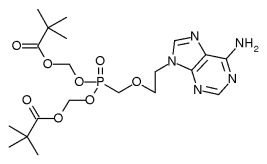
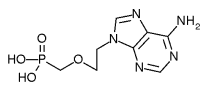
Gilead Sciences, Inc. (Nasdaq:GILD), Foster City, Calif., has submitted a Marketing Authorisation Application (MAA) to the European Medicines Evaluation Agency (EMEA) for adefovir dipivoxil 10 mg. Gilead is seeking an indication for the treatment of patients with chronic hepatitis B, including treatment-naive and treatment-experienced patients. The application is supported by data from Phase III studies in hepatitis B "e" antigen-positive patients, hepatitis B "e" antigen-negative patients and chronic hepatitis B patients with lamivudine-resistant HBV. This regulatory milestone follows the submission of a New Drug Application (NDA) to the U.S. Food and Drug Administration (FDA) for marketing approval.
"The rapid completion of the Marketing Authorisation Application for adefovir dipivoxil in Europe, following our U.S. NDA submission by less than a week, underscores our commitment to addressing the unmet medical needs in the treatment of chronic hepatitis B," said John C. Martin, PhD, President and CEO, Gilead Sciences. "The breadth of clinical data generated to date for adefovir dipivoxil, including data in precore mutant chronic hepatitis B -- a strain of the virus more common in many European countries -- suggest this drug may be a promising option for many patients suffering from this life-threatening, chronic infectious disease."
The MAA will be reviewed under the centralized licensing procedure, which, if approval is granted, provides a marketing license valid in all the 15 member states of the European Community. Review of the application will be coordinated by the EMEA. The French authorities will act as rapporteur for the application, and the Danish authorities will act as co-rapporteur.
About Adefovir Dipivoxil
Adefovir dipivoxil belongs to a class of drugs called nucleotide analogues which are designed to work by blocking hepatitis B virus (HBV) DNA polymerase, an enzyme involved in the replication of the virus in the body.
Data from two pivotal studies and a number of supportive studies of adefovir dipivoxil in a variety of chronic hepatitis B patient populations were presented at the 37th Annual Meeting of the European Association for the Study of the Liver (EASL). Presentations included data from studies of hepatitis B "e" antigen-positive patients, precore mutant (hepatitis B "e" antigen-negative) patients, those with lamivudine-resistant HBV, patients post-liver transplantation and patients co-infected with HIV. Since 1999, Gilead has provided access to adefovir dipivoxil through Study 435 to approximately 400 chronic hepatitis B patients with lamivudine-resistant HBV who are wait listed for or have received a liver transplant. Adefovir dipivoxil is an investigational compound and has not yet been determined safe or efficacious in humans.
Chronic Hepatitis B
Worldwide, there are approximately 350 million chronic carriers of hepatitis B, of which approximately one million die each year from complications of the disease, making chronic hepatitis B one of the 10 most common causes of death. Complications of chronic hepatitis B include cirrhosis (scarring of the liver), liver failure and primary liver cancer (hepatocellular carcinoma). Between one quarter and one third of people with chronic hepatitis B are expected to develop progressive liver disease. Patients infected with the precore mutant strain of hepatitis B may be predisposed to more severe and progressive liver injury. Precore mutant hepatitis B infects up to approximately 50 percent of the 350 million chronic hepatitis B carriers worldwide and is most prevalent in countries of the Mediterranean and Southeast Asia, where between 30-80 percent of chronic hepatitis B patients are estimated to be infected with this strain.
Early Access Program Initiated
In March 2002, Gilead announced the initiation of an early access program in the United States to provide adefovir dipivoxil to chronic hepatitis B patients with lamivudine-resistant HBV. This program is now open in the United States. A similar program opened in France in July 2001 and has enrolled 289 patients to date, and additional programs in Canada, Australia and in other countries in Europe will open in the coming months as appropriate regulatory approvals are obtained.
For more information regarding the adefovir dipivoxil early access program, or to request program registration materials, physicians may call 1-800-GILEAD-5 or 1-650-574-3000.
Gilead Sciences
Gilead Sciences is a biopharmaceutical company that discovers, develops and commercializes therapeutics to advance the care of patients suffering from life-threatening diseases worldwide. The company has five marketed products and focuses its research and clinical programs on anti-infectives, including antivirals, antifungals and antibacterials. Headquartered in Foster City, CA, Gilead has operations in the United States, Europe and Australia.
For more information on Gilead Sciences, please visit the company's web site at www.gilead.com or call the Gilead Corporate Communications Department at 1-800-GILEAD-5 or 1-650-574-3000.
COPYRIGHT 2002 Worldwide Videotex
COPYRIGHT 2002 Gale Group