Business Editors
NEWTOWN, Pa.--(BUSINESS WIRE)--Jan. 6, 2003
Study Includes CRP Measurement to Confirm Positive
Findings from Earlier Studies
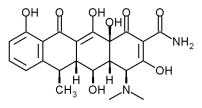
CollaGenex Pharmaceuticals, Inc. (Nasdaq:CGPI) today announced that the first patients have entered a multicenter, double-blinded, placebo-controlled Phase 4 clinical study to evaluate the combined efficacy of Periostat(R) (doxycycline hyclate tablets) 20mg, a systemic medication, and Atridox(R) (doxycycline hyclate) 10%, a locally-applied antimicrobial gel, in the treatment of adult periodontitis.
This study, which will enroll up to 180 patients, calls for full-mouth scaling and root planing at baseline, followed by either six months of Periostat or a matching placebo. Atridox will be applied to selected tooth sites in the active group at baseline and at three months. In addition to the evaluation of traditional clinical indices, a subset of patients will be tested for levels of the inflammatory marker, C-reactive protein (CRP) at baseline and during the course of the study, to establish the impact of periodontal pharmacotherapy on this key marker of cardiovascular risk.
"Recent studies suggest that there is a positive correlation between the extent of a patient's periodontal disease and their levels of CRP, and that elevated CRP in patients with periodontitis may signal an elevated risk for cardiovascular disease(1)," said Dr. John Novak, University of Kentucky, the lead investigator for the study. "The recent finding that Periostat significantly reduced levels of CRP in patients hospitalized for acute coronary syndromes suggests that Periostat may have a protective benefit in patients with periodontitis who are at risk of cardiovascular disease(2). Furthermore a previous study had illustrated that administration of Atridox could also impact CRP levels in periodontitis patients(3)."
In addition to Dr Novak, other investigators in the study include Dr. Sebastian Ciancio, State University of New York at Buffalo, Dr. Connie Drisko, University of Louisville, Dr. Maria Ryan, State University of New York at Stony Brook, Dr. Ingvar Magnussen, University of Florida, and Dr. Alan Polson, University of Pennsylvania.
"Both Periostat and Atridox have leading clinical data in the treatment of periodontitis," noted Brian M. Gallagher, PhD, chairman, president and chief executive officer of CollaGenex. "We anticipate that by combining Periostat and Atridox with mechanical treatment, it may be possible to establish a new standard of care in treating patients with periodontitis as well as those with an elevated risk of cardiovascular disease."
CollaGenex Pharmaceuticals, Inc. is a specialty pharmaceutical company currently focused on providing innovative medical therapies to the dental and dermatology markets. Currently, the Company's 120-person professional dental pharmaceutical sales force markets Periostat(R), the first and only pharmaceutical to treat periodontal disease by inhibiting the enzymes that destroy periodontal support tissues, Vioxx(R), a Merck & Co. drug that CollaGenex co-promotes for the treatment of acute dental pain, Denavir(R), a Novartis Group prescription anti-viral medication for the treatment of cold sores, and Atridox(R), Atrisorb(R) and Atrisorb-D(R), Atrix Laboratories Inc.'s products for the treatment of adult periodontitis. Research has shown that the enzyme suppression technology underlying Periostat may also be applicable to other diseases involving inflammation and/or destruction of the body's connective tissues, including acne, rosacea, meibomianitis and cancer metastasis, among others. CollaGenex is further developing Periostat, as well as a series of novel, proprietary compounds known as IMPACS (Inhibitors of Multiple Proteases and Cytokines), to address these applications. In addition, CollaGenex has licensed the Restoraderm(TM) technology, a unique, proprietary dermal drug delivery system, in order to develop a range of topical dermatological products with enhanced pharmacologic and cosmetic properties.
To receive additional information on the Company, please visit our Web site at www.collagenex.com.
This press release contains forward-looking statements within the meaning of Section 21E of the Securities and Exchange Act of 1934, as amended. Investors are cautioned that forward-looking statements involve risks and uncertainties that may affect the company's business and prospects. The company's business of selling, marketing and developing pharmaceutical products is subject to a number of significant risks, including marketing plans; risks inherent in research and development activities; risks associated with conducting business in a highly regulated environment and uncertainty relating to clinical trials of products under development, all as discussed in the company's periodic filings with the U.S. Securities and Exchange Commission.
Periostat(R), IMPACS(R) and Restoraderm(TM) are trademarks of CollaGenex Pharmaceuticals, Inc.
Pandel(R) is a trademark of Taisho Pharmaceuticals
VIOXX(R) is a trademark of Merck & Co., Inc.
Atridox(R), Atrisorb(R) and Atrisorb-D(R) are trademarks of Atrix Laboratories, Inc.
Denavir(R) is a trademark of the Novartis Group.
References:
(1) Noack, B et al: Relationship between periodontal disease
status, periodontal organisms, and C-reactive protein. J
Periodontol 72:1221-1227, 2001.
(2) Brown D et al: Clinical and Biochemical Results of the
Metalloproteinase Inhibition with Low-Dose Doxycycline to
Prevent Acute Coronary Syndromes (MIDAS) Pilot Trial.
Circulation 106: (Spec Issue 19; Abstract 2253):II-455, 2002.
(3) Alibhai, Z et al: Effect of periodontal therapy on systemic
inflammatory mediators. J Dent Res 81(Spec Issue A; Abstract
4076):A-498, 2002.
COPYRIGHT 2003 Business Wire
COPYRIGHT 2003 Gale Group