BOULDER, Colo., Dec. 8 /PRNewswire-FirstCall/ -- Pharmion Corporation reported today that data from several Vidaza(R) studies will be presented at the American Society of Hematology 47th Annual Meeting and Exposition in Atlanta (December 10-13, 2005). Vidaza is the only approved therapy for myelodysplastic syndromes (MDS) and is indicated for the treatment of all five subtypes of the condition.
These subtypes include: refractory anemia (RA) or refractory anemia with ringed sideroblasts (RARS) (if accompanied by neutropenia or thrombocytopenia or requiring transfusions), refractory anemia with excess blasts (RAEB), refractory anemia with excess blasts in transformation (RAEB-T), and chronic myelomonocytic leukemia (CMMoL).
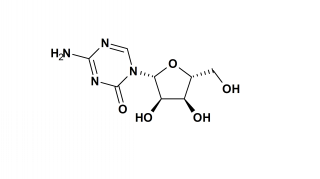
Vidaza is believed to exert its antineoplastic effects by causing hypomethylation of DNA and direct cytotoxicity on abnormal hematopoietic cells in the bone marrow. The concentration of azacitidine required for maximum inhibition of DNA methylation in vitro does not cause major suppression of DNA synthesis. Hypomethylation may restore normal function to genes that are critical for differentiation and proliferation. The cytotoxic effects of azacitidine cause the death of rapidly dividing cells, including cancer cells that are no longer responsive to normal growth control mechanisms. Non-proliferating cells are relatively insensitive to Vidaza.
Important Safety Information
Vidaza is contraindicated in patients with a known hypersensitivity to azacitidine or mannitol and in patients with advanced malignant hepatic tumors. In clinical studies, the most commonly occurring adverse reactions were nausea (70.5%), anemia (69.5%), thrombocytopenia (65.5%), vomiting (54.1%), pyrexia (51.8%), leukopenia (48.2%), diarrhea (36.4%), fatigue (35.9%), injection site erythema (35.0%), constipation (33.6%), neutropenia (32.3%) and ecchymosis (30.5%). Other adverse reactions included dizziness (18.6%), chest pain (16.4%), febrile neutropenia (16.4%), myalgia (15.9%), injection site reaction (13.6%), aggravated fatigue (12.7%) and malaise (10.9%). Because treatment with Vidaza is associated with neutropenia and thrombocytopenia, complete blood counts should be performed as needed to monitor response and toxicity, but at a minimum, prior to each dosing cycle.
Because azacitidine is potentially hepatotoxic in patients with severe pre-existing hepatic impairment, caution is needed in patients with liver disease. In addition, azacitidine and its metabolites are substantially excreted by the kidneys and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, it may be useful to monitor renal function. Vidaza may cause fetal harm. While receiving treatment with Vidaza, women of childbearing potential should avoid becoming pregnant, and men should avoid fathering a child. In addition, women treated with Vidaza should not nurse.
About MDS
The highest prevalence of MDS is in patients over 60 years of age. According to the American Cancer Society and the Aplastic Anemia and MDS International Foundation, there are an estimated 10,000-30,000 new cases of MDS in the United States each year. Survival ranges from six months to many years for the different subtypes of MDS.
About Pharmion
Pharmion is a pharmaceutical company focused on acquiring, developing and commercializing innovative products for the treatment of hematology and oncology patients in the U.S., Europe and additional international markets. For additional information about Pharmion, please visit the company's website at http://www.pharmion.com/ .
For more information or complete prescribing information about Vidaza, please call 1-866-PHARMION.
Safe Harbor Statement under the Private Securities Litigation Reform Act of 1995: This release contains forward-looking statements, which express the current beliefs and expectations of management. Such statements are based on current expectations and involve a number of known and unknown risks and uncertainties that could cause Pharmion's future results, performance or achievements to differ significantly from the results, performance or achievements expressed or implied by such forward-looking statements.
Important factors that could cause or contribute to such differences include the status and timing or regulatory approvals for Thalidomide Pharmion 50mg and Vidaza; the impact of competition from other products under development by Pharmion's competitors; the regulatory environment and changes in the health policies and structure of various countries; acceptance and demand for new pharmaceutical products and new therapies, uncertainties regarding market acceptance of products newly launched, currently being sold or in development; Pharmion's ability to successfully acquire rights to, develop and commercialize additional pharmaceutical products; fluctuations in currency exchange rates, and other factors that are discussed in Pharmion's filings with the U.S. Securities and Exchange Commission. Forward-looking statements speak only as of the date on which they are made, and Pharmion undertakes no obligation to update publicly or revise any forward-looking statement, whether as a result of new information, future developments or otherwise.
2005426
2005 (C) Pharmion Corporation. All rights reserved.
CONTACT: Breanna Burkart or Anna Sussman, Directors, Investor Relations and Corporate Communications of Pharmion Corporation, +1-720-564-9150
Web site: http://www.pharmion.com/
COPYRIGHT 2005 PR Newswire Association LLC
COPYRIGHT 2005 Gale Group