The article "Diabetes mellitus and its chronic complications" is the basis for this AORN Journal independent study. The behavioral objectives and examination for this program were prepared by Barbara K. Bailes, RN, EdD, CS, NP-C, with consultation from Susan Bakewell, RN, MS, education program professional, Center for Perioperative Education.
A minimum score of 70% on the multiple-choice examination is necessary to earn 3.5 contact hours for this independent study. Participants receive feedback on incorrect answers. Each applicant who successfully completes this study will receive a certificate of completion. The deadline for submitting this study is August 31, 2005.
Send the completed application form, multiple-choice examination, learner evaluation, and appropriate fee to
AORN Customer Service c/o Home Study Program 2170 S Parker Rd, Suite 300 Denver, CO 80231-5711
or fax the information with a credit card number to (303) 750-3212.
BEHAVIORAL OBJECTIVES
After reading and studying this article on diabetes mellitus and its complications, the nurse will be able to
(1) compare and contrast type 1 and type 2 diabetes mellitus,
(2) define the pathogenesis of type 2 diabetes as it evolves,
(3) explain the major complications of diabetes mellitus,
(4) discuss recent findings regarding the pathogenesis of vascular disease and its major complications,
(5) describe the clinical manifestations of peripheral neuropathy,
(6) discuss management of patients with type 2 diabetes, and
(7) identify possible complications associated with type 2 diabetes.
This program meets criteria for CNOR and CRNFA recertification, as well as other continuing education requirements.
Diabetes Mellitus and its Chronic Complications
Diabetes mellitus often is referred to as a silent killer because it annually contributes to approximately 18% of all deaths in the United States among patients who are age 25 and older. Diabetes is a significant risk factor for early onset of atherosclerotic vascular disease and coronary heart disease. The rate of heart disease in patients with diabetes is double that of the general population. Heart disease is the leading cause of death in patients with diabetes and has a mortality rate that is six times greater among male patients with diabetes and four times greater among female patients with diabetes. (1) Other complications of diabetes are retinopathy, nephropathy, and neuropathy. (2)
Every day, patients with diabetes undergo scheduled or emergency surgical procedures. Perioperative nurses often are aware of the chronic complications that trouble patients with diabetes, but they may not be familiar with current findings and treatment goals. The purpose of this article is to provide a brief overview of the pathogenesis of type 1 and type 2 diabetes mellitus; the chronic complications and underlying pathophysiology of diabetes mellitus; various treatment protocols being investigated to prevent, reverse, or alleviate chronic complications of diabetes; treatment protocols; and nursing interventions that can be used to provide intraoperative care for patients with diabetes undergoing surgery.
EPIDEMIOLOGY
The prevalence of diabetes mellitus worldwide has increased dramatically during the past few decades, and it is expected to increase even more in the future. In the United States, approximately 16 million Americans have diabetes mellitus, and more than 90% of these have type 2 diabetes mellitus. Each year, approximately 800,000 Americans are diagnosed with diabetes mellitus, and many others remain undiagnosed. In addition, there are countless people with impaired glucose tolerance (ie, prediabetic condition) or insulin resistance. Diabetes mellitus is confirmed when an individual has a fasting plasma glucose of 126 mg per dL or higher. (3) Table 1 lists major risk factors for type 2 diabetes mellitus. (4)
The incidence of diabetes mellitus essentially is the same for males and females, and it increases with age. The prevalence of known cases rises from about 6% in people age 45 to 64 years to approximately 12% in those age 65 and olden In the older age group, more women than men are diagnosed with diabetes each year. High-risk groups include African Americans, Hispanics, Native Americans, Asian Americans, and Pacific Islanders. Regardless of race or ethnicity, however, obesity and advancing age both are correlated to the increased incidence of diabetes mellitus. (5) Table 2 lists criteria for the diagnosis of diabetes mellitus. (6)
CATEGORIZING DIABETES MELLITUS
Diabetes mellitus is a group of metabolic diseases caused by a complex interaction of genetic, immunological, and environmental factors, as well as life-style choices. These diseases also feature chronic hyperglycemia. Chronic hyperglycemia is associated with microvascular and macrovascular changes that result in organ damage, dysfunction, or failure. Experts believe that microvascular changes begin when the diabetic state becomes overt (ie, fasting blood glucose is at or higher than 126 mg per dL), and macrovascular changes begin many years earlier. (7)
Microvascular changes affect the eyes and kidneys, and macrovascular changes predominately affect the cardiovascular system. Long-term complications involving the eyes include retinopathy and potential loss of vision. Nephropathy that is not aggressively treated leads to end-stage renal failure. Atherosclerotic changes increase cardiovascular risk factors so much that diabetes now is considered a heart disease equivalent. (8)
Although there are different methods of categorizing diabetes, the American Diabetes Association (ADA) has identified four major categories of diabetes mellitus including
* type 1 diabetes mellitus,
* type 2 diabetes mellitus,
* gestational diabetes, and
* impaired glucose tolerance (ie, prediabetes).
Types 1 and 2 diabetes mellitus predominate. Gestational diabetes appears during pregnancy, and blood glucose levels typically return to normal upon completion of the pregnancy; however, these women have a greater risk of developing overt diabetes later in life. Impaired glucose tolerance (ie, prediabetes) is a state in which patients have blood glucose levels that are higher than normal, but the glucose levels are not high enough for a diagnosis of diabetes. Data from research studies show that many people with prediabetes can prevent or delay the development of diabetes through weight reduction and a regular exercise program. (9)
Type 1 diabetes formerly was known as juvenile diabetes or insulin-dependent diabetes. People with type 1 diabetes usually are young and thin, although it can and does occur in older adults. Type 1 diabetes is caused by the synergistic effects of genetic, environmental, and immunological factors that destroy the pancreatic beta cells, resulting in an absolute deficiency of insulin secretion. Without insulin, glucose levels rise and cause polyuria, polydipsia, polyphagia, and weight loss. If not diagnosed and treated, marked hyperglycemia can progress to ketoacidosis, coma, and death. Current therapy for type 1 diabetes is to provide the patient with exogenous insulin to maintain normal plasma glucose levels, prevent hyperglycemia and ketoacidosis, and prevent or delay chronic complications. (10)
Type 2 diabetes mellitus previously was known as adult onset or noninsulin-dependent diabetes. Patients with type 2 diabetes generally are older, although there is an alarming increase in the incidence of type 2 diabetes in children and adolescents. (11) Many patients with type 2 diabetes have insulin-resistance syndrome (eg, central obesity, hypertension, hyperlipidemia) for many years. There are three major pathophysiologic abnormalities in patients with type 2 diabetes, including
* early loss of first-phase insulin production associated with defective beta cell secretion,
* peripheral resistance to insulin action primarily in muscle tissue and the liver, and
* excessive hepatic glucose production as disease progresses. (12)
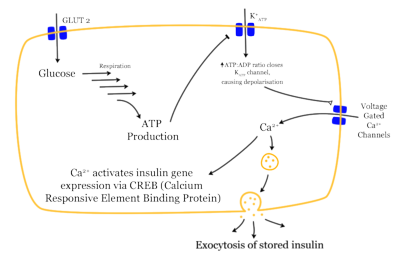
Normally, first-phase insulin secretion exerts an inhibitory effect on hepatic glucose production and output. When a patient has beta cell defects, first-phase insulin secretion is impaired and eventually lost, which results in fasting hyperglycemia. The body's attempt to moderate blood glucose levels results in enhanced second-phase insulin secretion, and hyperinsulinism occurs. Beta cells may secrete high levels of insulin to normalize blood glucose levels and successfully maintain normoglycemia for many years. Gradually, however, the beta cells will begin to falter, and insulin secretion decreases. As hepatic glucose production increases, both fasting and postprandial glucose levels become elevated. (13)
Insulin resistance implies that the body's cells are less sensitive to the action of insulin. Insulin resistance affects the disposal of glucose in muscle tissue and suppresses the output of hepatic glucose. Consequently, blood glucose levels rise, even though the beta cells produce more insulin. In patients with insulin resistance, however, hyperinsulinism does not suppress gluconeogenesis, and chronic hyperglycemia develops. Insulin sensitivity can decline by at least 70% before fasting plasma glucose concentrations become abnormal, and it may take up to 20 years to reach that point. Experts are not certain yet about the mechanism underlying insulin resistance, but they know that obesity, particularly central obesity, increases insulin resistance. They further speculate that defects in intracellular signaling prevent glucose from entering cells. (14)
Treatment goals for patients with type 2 diabetes include achieving and maintaining normoglycemia through weight reduction and exercise, diet and portion control, use of exogenous insulin, use of oral agents that stimulate the secretion of insulin or reduce insulin resistance, or use of a combination of oral agents and exogenous insulin. A secondary, but equally important goal, is to prevent or delay the onset of long-term complications. (15)
COMPLICATIONS OF DIABETES MELLITUS
In recent years, important advances have been made in understanding the pathogenesis of diabetes. Scientists are learning more about how diabetes affects the cardiovascular, renal, nervous, and ophthalmological systems. Some of the complications from diabetes include
* peripheral vascular disease (PVD),
* diabetic neuropathy,
* diabetic foot problems,
* diabetic retinopathy, and
* nephropathy.
Peripheral vascular disease. Atherosclerosis is common in patients with diabetes, and it causes increased incidences of microangiopathy and macroangiopathy. Macroangiopathy causes an accelerated form of atherosclerosis that affects important vessel sites, including the coronary, carotid, aortic, iliac, femoral, popliteal, tibial, and peroneal vessels. (16)
Patients with diabetes are approximately four times more likely to have PVD than patients without diabetes. Many patients with PVD have few symptoms, and others have significant clinical manifestations. Patients with diabetes who also have PVD have an increased incidence of calf vessel involvement (eg, peroneal and tibial arteries). Patients with diabetes also have greater difficulty developing collateral blood supply in the lower extremities, although the reason for this is not clear. Symptoms include claudication or leg pain on rest, loss of foot hair, dependent rubor and coolness of the feet, and diminished or absent peripheral pulses. (17)
It is well established that diabetes develops early and patients with diabetes have accelerated atherosclerosis. Experts theorize that the pathophysiology underlying atherosclerosis in patients with diabetes includes many variables, (eg, dyslipidemia, hyperglycemia, altered platelet and coagulation function, the presence of adhesion molecules, excessive levels of plasminogen activator inhibitor type I). Plasminogen activator inhibitor type I fosters altered platelet function and plaque formation and contributes to atherogenesis in patients with diabetes. (18)
The vascular endothelium now is recognized as a multifunctional specialty organ strategically placed between vessel walls and circulating blood, and it performs important roles in vascular hemostasis and renal function. (19) The endothelium produces several regulatory mediators, including nitric oxide and prostacyclin, which are vasodilators, and endothelin, which is a vasoconstrictor. In a normal state, the endothelium actively decreases vascular tone; regulates vascular permeability to nutrients, hormones, and other macromolecules; increases vascular permeability to leukocytes; inhibits platelet adhesion and aggregation; limits activation of the coagulation cascade; and regulates fibrinolysis. (20)
Nitric oxide, a principle physiological dilator, produces local vasodilatation and inhibits platelet adherence and aggregation. Nitric oxide also inhibits proliferation of smooth muscles, reduces leukocyte adhesiveness, and regulates endothelial permeability, to lipoproteins and other constituents of plasma. (21)
There is evidence that insulin resistance decreases the synthesis of nitric oxide. When nitric oxide is deficient or used inadequately, endothelium becomes a procoagulant instead of an anticoagulant, and vasoactive molecules, cytokines, and growth factors are formed. The endometrium becomes more permeable, which permits the increased presence of lipoproteins, monocytes, and macrophages. These substances increase smooth muscle cell migration; foster the formation and proliferation of atherosclerotic plaques; alter the function and structure of blood vessels; and, eventually, jeopardize organ function. (22) Diabetic neuropathy. Of all of the long-term complications of diabetes, diabetic neuropathy is the most common and least understood. Fifty percent of all patients with diabetes will experience diabetic neuropathy when they have had the disorder for 25 years or more. Some patients with type 2 diabetes may develop signs of diabetic neuropathy soon after they are diagnosed. Diabetic neuropathy leads to visual, gastrointestinal, sexual, and peripheral vascular disturbances. (23)
As with atherogenesis, the pathogenesis of diabetic neuropathy is multifactoral with metabolic and vascular involvement. Microangiopathy of the vasa nervorum (ie, small arteries located in the outer and middle coats of the larger arteries and veins) develop and impair normal blood flow. Diminished nerve blood flow, in turn, reduces nerve perfusion and oxygenation, which ultimately results in damage to axons and the myelin sheath. (24)
In the past, experts believed that these metabolic defects were caused by chronic hyperglycemia. They were able to show that tight glucose control dramatically reduced the incidence of clinical neuropathy; however, tight glucose control did not completely prevent diabetic neuropathy, so investigators concluded that other factors were involved. These factors include the presence of microvascular disease, nonenzymatic glycation and oxidative stress via activation of the polyol pathway, altered lipid metabolism leading to long-chain fatty acid toxicity, reduction in neurotrophic factor binding, and altered insulin mediation. (25)
The polyol pathway uses the enzyme aldose reductase to convert glucose to sorbitol. Sorbitol, in turn, is converted to fructose by sorbitol dehydrogenase. When glucose levels increase, more fructose is produced to enhance oxidative stress nonenzymatic glycation. During nonenzymatic glycation, glucose, fructose, galactose, and other sugars interact with red blood cells and other proteins, undergo chemical rearrangements, and form advanced glycation end products. These glycochelates (ie, heavily glycated proteins) accumulate metals, especially copper, causing a catalytic destruction of the nitric oxide-endothelium-derived-relaxing factor and producing chronic vasoconstriction. (26) Although glycochelates amass slowly and naturally during normal aging, in patients with diabetes, these substances accumulate more rapidly in the lens, retinas, kidneys, peripheral nerves, and other tissues. (27)
Diabetic neuropathy also affects the sensory, motor, or autonomic systems or a combination of these. Diabetic neuropathy includes several distinct syndromes, which are classified in different ways. One system, based on anatomic classification, is divided into diffuse or focal syndromes, including
* distal symmetric sensorimotor polyneuropathy,
* focal and multifocal neuropathies,
* entrapment neuropathy,
* autonomic neuropathy, and
* sensorimotor polyneuropathy. (28)
Distal symmetric sensorimotor polyneuropathy. Distal symmetric sensorimotor polyneuropathy is the most common form of diabetic neuropathy. This syndrome is insidious. It begins in the toes and gradually moves up the legs, causing a stocking pattern of sensory loss. Later, the upper extremities develop a glove pattern sensory loss beginning in the fingertips and progressing up the arms. Small fiber sensory neurons usually are affected first with loss of pain and temperature sensation. Large sensory fibers are affected later, and this results in a loss of vibration and proprioceptive sensation and some gait and balance disturbances. (29)
Focal and multifocal neuropathies. Focal and multifocal neuropathies may be ischemic or entrapment neuropathies. Ischemic focal neuropathies are acute, asymmetric, self-limiting events involving a single vessel or group of vessels. Oculomotor neuropathy presents with eye pain, diplopia, or ptosis and usually resolves within a few weeks. Femoral nerve ischemia causes proximal (ie, hip, thigh) muscle weakness and severe pain. Residual deficits linger, and recovery may take many months. A thorocolumbar neuropathy/radiculopathy may cause chest or abdominal pain. The pain pattern follows the nerve root, and sensory loss follows a dermatome. (30)
Entrapment neuropathy. Entrapment neuropathy includes carpal tunnel and ulnar nerve entrapment. Although these syndromes also are seen in patients without diabetes, they are more common in patients with diabetes. (31)
Autonomic neuropathy. Autonomic neuropathy is a serious form of neuropathy with different presentations. Abnormalities include impotence, gastrointestinal disturbances, bladder dysfunction, diabetic diarrhea, and orthostatic hypotension. (32) Patients with autonomic neuropathy may develop an auto-sympathectomy-like state. In this situation, there is a loss of sweating of the feet, and the skin of the feet becomes dry and thickened. Cracks and fissures develop, especially in the heels. This increases the risk of ulceration. If no large vessel disease is present, arteriovenous shunting increases blood flow to the feet. Even though the feet are warm, however, the risk of ulceration and infection are high. (33)
Pain symptoms in autonomic neuropathy may be episodic or continuous and are described as lacinating (ie, lightening-like), burning, or aching. These symptoms often are worse at night and may cause sleep disturbances. Individuals with autonomic neuropathy also may experience paresthesia, allodynia (ie, pain upon benign stimuli, such as a sheet touching the feet), and hypesthesia (ie, increased sensations in affected areas). Sensation is impaired and eventually lost as the nerves die. (34)
Sensorimotor polyneuropathy. A variant of sensorimotor polyneuropathy is painful diabetic neuropathy. Individuals with this form of neuropathy experience severe, relentless leg pain that is worse at night. In addition, cutaneous hyperesthesia may develop that is so severe that affected individuals are unable to endure the touch of a sheet or clothing on their feet. (35)
Diabetic foot problems. Both PVD and diabetic neuropathy are the indirect cause of many diabetic foot problems. Approximately 20% of all hospital admissions of patients with diabetes are for diabetic foot conditions (eg, ulceration, infection, foot deformities, Charcot neuroarthropathy, osteoarthritis, amputation). (36)
Sensory neuropathy is the underlying cause of most foot ulcers. Initial symptoms usually begin with paresthesia and dysesthesia and culminate in a loss of sensation. As previously discussed, the presence of autonomic neuropathy causes a decline in skin temperature regulation and loss of normal sweating of the feet so that the skin becomes dry and scaly. Consequently, fissures develop in the heels, the skin barrier is disrupted, and the risk of infection increases. (37)
After ulceration develops, wound healing is difficult and, in some cases, impossible. Patients with diabetes have impaired neutrophils, fibroblasts, and leukocyte activity, as well as impaired migration and action of inflammatory cells to the ulcer site. In older adult patients with diabetes, subcutaneous fat decreases, elastin fibers become fragmented, and epidermal turnover and collagen production decrease and further compromise healing. (38)
Motor neuropathy leads to significant muscle wasting and an imbalance of the intrinsic muscles of the feet, with the flexor muscles dominating the extensor muscles. This syndrome, known as "intrinsic feet," leads to hammer and claw toes and depression of the metatarsal heads. The metatarsal footpad gradually becomes displaced laterally, pressure on the footpad is increased, and cushioning is lost. In addition, the flexion deformities of the interphalangeal joints result in continual pressure and rubbing by the shoe, which causes irritation and, ultimately, ulceration. The two most common causes of minor foot trauma are improper cutting of toenails and poorly fitted shoes. Minor trauma, such as cuts, blisters, and tinea pedis (ie, athlete's foot), may result in infection and ulceration. The goal is to prevent foot trauma from occurring. (39)
Charcot osteoarthropathy is a highly destructive but somewhat painless disorder that occurs in people with neuropathy and involves one or multiple joints of the feet. Although Charcot's foot can occur in patients with various disorders, it is seen most commonly in patients with diabetes. Experts believe that Charcot's phenomenon has both a neurotrauma and neurovascular element, resulting in neuropathic bone and joint changes. When trauma and injury to the foot are not recognized and treated early, further joint destruction and deformity occurs. If sufficiently severe, hyperkeratosis or ulceration may develop, increasing the potential for infection that often is resistant to treatment because of impaired blood flow and an impaired immune system. (40)
Diabetic foot infections may be minor and non-limb threatening or major and limb threatening. Minor infections do not involve bones or joints and usually are caused by staphylococcus aureus. Major and limb-threatening infections usually are polymicrobial and may involve aerobic gram positive cocci, gram negative bacilli, anaerobes, and enterococci. Approximately one-third to two-thirds of patients with diabetes who have foot infections will develop osteomyelitis. (41) These patients are at high risk for amputation. (42)
Diabetic retinopathy. Diabetic retinopathy is the leading cause of blindness in people younger than 75 years of age in the United States. The risk of retinopathy is related directly to the degree and control or lack of control of hyperglycemia. It is present in almost all people who have had diabetes for 20 years or more. The 20-year prevalence of retinopathy is approximately 80% in people who require insulin and approximately 20% in those who do not require insulin. (43)
Diabetic retinopathy develops as a result of several processes, and it progresses from a mild nonproliferative to moderate to severely proliferative condition. These processes include
* the presence of microaneurysms of the retinal capillaries;
* excessive vascular permeability;
* vessel occlusion;
* neovascularization (ie, the development of new blood vessels);
* fibrous tissue on the retina and optic disc; and
* contraction of the vitreous, vessels, and fibrous tissue.
Microaneurysms of the retinal capillaries develop first and are markers for the development of diabetic retinopathy. Macular edema (ie, retinal thickening caused by accumulation of fluid) may develop and may be accompanied by hard exudates (ie, yellow-white lipid deposits). Macular edema may come and go without impairing vision, but the presence of lipid deposits, especially beneath the center of the macula, is associated with retinal damage and loss of vision. People with microaneurysms, with or without macular edema, have mild, nonproliferative retinopathy. (44)
Vision loss from diabetes mellitus is caused by several factors. Macular edema or nonprofusion of the retina may impair central vision. The new vessels may rupture and bleed. When this happens, fibrous tissue contracts, and the retina may become detached, resulting in a potentially irreversible loss of vision if not diagnosed and treated immediately. (45)
The main objective in the prevention of diabetic retinopathy is tight glucose control and early screening to find and prevent diabetic retinopathy from developing and progressing. All patients with diabetes should undergo annual dilated eye examinations by an ophthalmologist or optometrist. Individuals who have macular edema, proliferative retinopathy, or severely nonproliferative retinopathy should be referred to a retinal specialist. The current standard of care for diabetic retinopathy is laser photocoagulation. (46)
Nephropathy. Diabetic nephropathy is characterized by albuminuria, hypertension, and progressive loss of renal function. It is a microvascular complication that is the most common single cause of end-stage renal disease if not treated adequately. The earliest clinical indicator of nephropathy is microalbuminuria, which is low but abnormal levels of albumin in the urine (ie, [greater than or equal to] 30 mg per day or 20 [micro]g per minute). (47)
As with the other complications of diabetes, the pathogenesis of diabetic nephropathy is complex and involves direct effects by high plasma glucose concentrations on vascular, glomerular, tubular, and interstitial cellular function. Hyperglycemia and the formation of advanced glycosylation end products and protein kinase C isoforms are implicated in the development of diabetic nephropathy. Experts agree that diabetic nephropathy develops in the presence of an intact renin-angiotension system that secretes angiotensin II. Angiotensin II increases glomerular capillary pressures, stimulates cellular hypertrophy, and causes accumulation of extracellular matrix. This, combined with other microvascular changes, results in diabetic nephropathy . (48)
The main objective is to prevent diabetic nephropathy because there is sufficient evidence that early diabetic nephropathy need not progress to end-stage renal disease. All patients with diabetes who also have proteinuria should have annual microalbuminuria and 24-hour urine testing. Treatment includes strict plasma glucose control to achieve and maintain normoglycemia and administration of angiotensin converting enzyme inhibitors to decrease the progress of diabetic nephropathy. Both systolic and diastolic hypertension accelerates the progression of renal damage, so all patients with diabetes who also have hypertension need to maintain a blood pressure of less than 130/80. (49)
MEDICATIONS
For more than 25 years, pharmaceutical companies have conducted clinical trials on different formulations of aldose reductase inhibitors (ARI) hoping to find a medication that would retard or even reverse the effects of diabetic neuropathy. (50) Zebarestat was the last ARI to be developed and tested. In clinical trials, fat diabetic rats were given controlled doses of Zebarestat. Although the medication did reduce some nerve sorbitol and there was some improvement in nerve conduction, the medication's efficacy was limited, and adverse effects were problematic. Clinical trials of Zebarestat have been discontinued. (51)
Other medications that have undergone clinical trials to treat diabetic neuropathy include essential fatty acids (eg, gamma-lenoleic acid) (52) and antioxidants (eg, alpha-lipoic acid). (53) Individuals involved in these studies had limited relief of symptoms but no long-term improvement. Clinical trials are ongoing.
Currently, neuropathic pain is treated with a variety of medications, including tricyclics, anticonvulsives (eg, phenytoin, gabapentin), beta blockers (eg, propranolol), benzodiazepine (eg, lorazepam), lidocaine, codeine, capsaicin, nonsteroidal anti-inflammatory drugs, and magnesium. Of these, tricyclics are the most effective in at least half of the patients using them. Amitriptyline often is prescribed, even though it causes significant drowsiness. Two other tricyclics, desipramine and nortriptyline, are just as effective and do not cause as much drowsiness as amitriptyline. (54)
Topical capsaicin, which is made from hot peppers, provides significant relief for some people when larger than normal doses are used. Unfortunately, capsaicin produces an almost painfully hot sensation when first applied, and many people refuse to use it after one or two applications. To alleviate this discomfort, investigators in one study administered a regional anesthetic first. Although trials showed that capsaicin did provide relief, the discomfort it caused did not make it useful for routine relief of neuropathic pain. (55)
Anticonvulsive medications also have been used with some success to treat neuropathic pain because there are similarities in a number of biochemical and pathophysiologic changes that occur in seizure activity and neuropathic dysfunction. Gabapentin, a newer anticonvulsive, has been used with some success, even though significant adverse effects were noted by some of the participants in the clinical trial. (56)
Aspirin and ticlopidine both are prescribed for arterial insufficiency. Aspirin, having antiplatelet properties, retards or prevents platelet thrombogenesis on atherosclerotic plaques. An enteric-coated aspirin in doses of 81 mg to 325 mg per day is recommended for all patients older than the age of 21 unless there is a contraindication. Ticlopidine, also an antiplatelet medication, improves walking distances and delays the need for vascular surgery; however, it is associated with adverse effects, such as neutrapenia and thrombotic thrombocytopenia. (57)
Several new medications have been approved by the US Food and Drug Administration to treat patients who experience intermittent claudication or pain at rest, which can indicate more severe limb ischemia. Pentoxifyline is a weak antithrombotic agent that is reported to reduce blood viscosity and reduce abnormal erythrocyte deformities. Experts have reported only mild improvement in walking distances (ie, 20% to 25%) with pentoxifyline use. (58) Cilostrazol is a more promising medication than pentoxifyline. It is a type III phosphodiesterase inhibitor and is both an antiplatelet and vasodilating agent. Cilostrazol improves walking distance when compared with a placebo in about 40% to 60% of patients tested. (59)
Other medications that currently are being investigated to treat peripheral arterial disease include L-carnitine, L-arginine, vasodilator prostaglandins, fish oils, nerve growth factor, and ethylenediaminetetraacetic acid chelation therapy. L-camitine shows some promise in clinical trials. It appears to facilitate the transfer of acylated fatty acids across mitochondrial membranes, enhance available energy stores, and improve oxidative muscle metabolism. Nerve growth factor, a protein that lengthens the life of nerve cells, also is a medication that may not only prevent the onset of diabetic neuropathy but also may reverse it. Clinical trials continue. (60)
CURRENT PHARMACOLOGICAL THERAPY
The goals of therapy for patients with diabetes mellitus are to improve and normalize blood glucose levels and reduce the overall risks of microvascular complications. The goals of therapy recommended by the ADA are shown in Table 3. (61)
Patients with type 1 diabetes have an absolute deficiency of beta cells and insulin secretion, so exogenous insulin of different formulations and action are used to maintain normal blood glucose levels. Patients with type 2 diabetes have functioning beta cells and produce insulin. Treatment includes weight reduction and an exercise program, diet modification with portion control, oral diabetic medications, and insulin. In some patients with diabetes, weight reduction, exercise, and diet modification are sufficient to normalize blood glucose values. Other patients will need monotherapy or a combination of different classes of oral diabetic medications. Eventually, most patients with diabetes will require a combination of oral medication and insulin as their beta cells fail to produce sufficient insulin. (62)
Medications for patients with type 2 diabetes include various insulin formulations as well as oral diabetic medications, which are divided into broad categories according to their action. (63) Table 4 lists the available insulin preparations, and Table 5 lists commonly used oral diabetic medications. (64)
PERIOPERATIVE NURSING INTERVENTIONS
Although perioperative nurses have limited contact with patients before surgery, an assessment can be performed with sufficient knowledge and a review of the risk factors for diabetes and its complications. Just as with other patients, patients with diabetes who are undergoing major surgery have food and fluids withheld for approximately eight hours before the procedure. The physician writes specific orders regarding whether and how the patient should take his or her medication. If there is an opportunity to make a timely preoperative visit, the perioperative nurse should know what these instructions are and review them with the patient. If the procedure is major, the patient usually is given IV fluids with regular insulin. If it is a minor procedure, the patient may be instructed to simply hold the medication until the surgery is completed and food can be taken.
The patient's chart is reviewed for laboratory work, specifically blood glucose, blood urea nitrogen and creatinine, and electrolytes, although all abnormal or unusual values need to be noted and reported to the surgeon and anesthesia care provider. Patients with diabetes may present with higher blood glucose levels than normal because the metabolic stress response that occurs with illness or a stressor, such as surgery, increases the secretion of counterregulatory hormones. These hormones accelerate catabolism, hepatic glucose gluconeogenesis, and lipolysis, further elevating the blood glucose level. Although strict glycemic control should be the goal for all patients with diabetes--except for older, frail adults--physicians may allow more liberal glucose levels of approximately 150 to 200 mg per dL to prevent hyperosmolar, nonketotic states from occurring during hospitalization. Patients with diabetes whose conditions have been controlled via diet or an oral diabetic medication may be on insulin for several days. They need to be assured that their diabetes is not getting worse and that they will be able to resume oral medication after their blood glucose level has returned to preoperative levels.
Many diabetic patients will have stocking or glove types of neuropathy and have little or no sensation in their feet, lower legs, hands, or upper extremities. The circulation in the lower extremities of patients with diabetes already is impaired, so extremities need extra protection from the cold temperatures found in the OR. They also need good alignment and extra padding to avoid compromising blood flow. Although it is not necessary to emphasize good aseptic technique to perioperative nurses, it is important to stress that patients with diabetes are at increased risk for poor wound healing and infection. The nursing care plan in Table 6 may help perioperative nurses provide appropriate preoperative and intraoperative care for patients with diabetes. (65)
CASE STUDY
Mr D is a 60-year-old man with diabetes who is scheduled for left total knee prosthesis because of severe arthritis and pain. Mr D is 6 ft tall and weighs 200 lbs. At the time of admission, his blood pressure is 130/70, pulse is 88, respiration is 16, and temperature is 98.6[degrees]F. Mr D's medical history includes type 2 diabetes mellitus of 10 years duration, coronary heart disease, hypertension, and hyperlipidemia. His past surgical history includes multiple surgical procedures on the knee and coronary artery bypass surgery four years ago. Current medications include ramipril, 10 mg per day; anlodipine, 5 mg twice daily; glimepiride, 4 mg; rosiglitazone 2 mg; gemfibrozil 250 mg twice daily; and aspirin, 325 mg every day.
During the preoperative interview, the admitting nurse learns that Mr D's knowledgeable about his disease and takes an active role in its control. He acknowledges that he is overweight, but he is unable to exercise because of his painful knee. He reports that his glycosylated hemaglobin was 6% last month; however, his triglycerides still were elevated. His last report noted his triglycerides as 400 mg per dL. He does not experience chest pain or shortness of breath. He does report some tingling and burning in his feet, and he has little sensation in his toes. Mr D's feet are cool but the skin is unbroken and free from lesions. In addition, Mr D's primary physician and surgeon have decided that he will receive lactated Ringer's solution with glucose and regular insulin. He has withheld aspirin for four days, and his hyperglycemia will be controlled with IV fluids and insulin until he can take food and fluids. After that, his diabetes will be monitored with bedside glucose testing. His medications for diabetes will be a combination of oral medication and insulin, if required. Mr D will receive an epidural for pain relief. He anticipates a smooth and fairly fast recovery and does not seem overly anxious about the procedure.
On the morning of surgery, Mr D's blood glucose level is 160 mg per dL. He takes his ramipril and anlodipine with a small amount of water as directed by his physician. His blood pressure is 130/70, pulse is 80, respiration is 16, and temperature is 98.6[degrees] F. His laboratory tests are within normal limits except for albumin in the urine. The electrocardiogram report shows normal sinus rhythm. Mr D's blood glucose levels are elevated, so the preoperative nurse should reassure him that these glucose values are within acceptable limits during this surgical experience, and remind him that he will be receiving both insulin and glucose during the procedure and that his blood glucose levels will be checked regularly. (66)
SUMMARY
Diabetes mellitus affects millions of people in the United States and worldwide and is associated with several chronic disorders that are potentially life threatening. Perioperative nurses regularly provide care to patients with diabetes who are undergoing surgery. Patients with diabetes can achieve normal blood glucose levels with diet and portion control, weight reduction and exercise, oral diabetic medication, or insulin given as either monotherapy or combination therapy. Many patients with diabetes who are unable or unwilling to achieve ideal glycemic control develop chronic complications of diabetes, and others follow diabetic protocols and still develop chronic complications. Researchers continue to look for the underlying causes of diabetes mellitus and search for new medications that can prevent, halt, or reverse the chronic effects of diabetes. Treatment goals are to prevent the development of diabetes mellitus, detect its onset early, and provide comprehensive and timely treatment to prevent or delay chronic complications of diabetes mellitus.
NOTES
(1.) R Singh, S E Weigers, B J Goldstein, "The impact of gender on diabetes mellitus and its associated cardiovascular risk factors," Clinical Geriatrics 9 (June 2001) 63-72; "American Diabetes Association. 61st scientific sessions. Friday, June 22-Tuesday, June 26, 2001, Philadelphia, Pennsylvania," Diabetes 50 suppl (June 2001) 1-106.
(2.) M I Harris, R C Eastman, "Early detection of undiagnosed diabetes mellitus: A US perspective," Diabetes/Metabolism Research and Reviews 16 (July/August 2000) 230-236; S H Rao, "Treating to target: A rational approach based on pathophysiology," Consultant 41 no 13 suppl (November 2001) S 14-S21.
(3.) Ibid; Singh, Weigers, Goldstein, "The impact of gender on diabetes mellitus and its associated cardiovascular risk factors," 63-72.
(4.) American Diabetes Association, "Position statements: Standards of medical care for patients with diabetes mellitus," Diabetes Care 25, no 1 suppl (2001) S33-S49.
(5.) Singh, Weigers, Goldstein, "The impact of gender on diabetes mellitus and its associated cardiovascular risk factors," 63-72.
(6.) "Report of the expert committee on the diagnosis and classification of diabetes mellitus," Diabetes Care 20 (July 1997) 1183-1197; Rao, "Treating to target: A rational approach based on pathophysiology," S 14-S21; American Diabetes Association, "Position statements: Screening for diabetes," Diabetes Care 25, no 1 suppl (2002) S21-S22.
(7.) M. Qian, J W Eaton, "Glycochelates and the etiology of diabetic peripheral neuropathy," Free Radical Biology & Medicine 28 (Feb 15, 2000) 652-656.
(8.) "Report of the expert committee on the diagnosis and classification of diabetes mellitus," 1183-1197; A M Valente, W Strong, A R Sinaiko, "Results of experts meetings: Conducting pediatric cardiovascular trials--Obesity and insulin resistance in young people," The American Heart Journal 142, no 3 (2001) 440-444.
(9.) "Report of the expert committee on the diagnosis and classification of diabetes mellitus," 1183-1197; J Tuomilehto et al, "Prevention of type 2 diabetes mellitus by changes in lifestyle among subjects with impaired glucose tolerance," The New England Journal of Medicine 344 (May 3, 2001) 1343-1350; "Pre-diabates," American Diabetic Association, http://diabetes.org/ main/info/pre-diabetes.jsp (accessed 14 June 2002).
(10.) "Report of the expert committee on the diagnosis and classification of diabetes mellitus," 1183-1197.
(11.) Ibid; S Del Prato, A Tiango, "The importance of first-phase insulin secretion: Implications for the therapy of type 2 diabetes mellitus," Diabetes/Metabolism Research and Reviews 17 (May/June 2001) 164-174; G Sesti et al, "Review articles--Molecular mechanism of insulin resistance in type 2 diabetes mellitus: Role of the insulin receptor variant forms," Diabetes/Metabolism Research and Reviews 17 (July/August 2001) 363-373.
(12.) Del Prato, Tiango, "The importance of first-phase insulin secretion: Implications for the therapy of type 2 diabetes mellitus," 164-174; Sesti et al, "Review articles--Molecular mechanism of insulin resistance in type 2 diabetes mellitus: Role of the insulin receptor variant forms," 363-373.
(13.) Del Prato, Tiango, "The importance of first-phase insulin secretion: Implications for the therapy of type 2 diabetes mellitus," 164-174.
(14.) M T Sheehan, M D Jensen, "Metabolic complications of obesity: Pathophysiologic considerations," Medical Clinics of North America 84 (March 2000) 363-385; A H Mokdad et al, "The continuing epidemics of obesity and diabetes in the United States," JAMA 286 (Sept 12, 2001) 1195-2000.
(15.) E Strock, "Type 2 diabetes: Strategies for patient care and education," Consultant 41 no 13 suppl (November 2001) S6-S 13.
(16.) W R Aengevaeren, "Beyond lipids--the role of the endothelium in coronary artery disease," Atherosclerosis 147 suppl (Sept 9, 1999) S11-S16; C D Stehouwer et al, "Endothelial dysfunction and pathogenesis of diabetic angiopathy," Cardiovascular Research 34 (April 1997) 55-68; D W Laight, M J Carrier, E E Anggard, "Endothelial cell dysfunction and the pathogenesis of diabetic macroangiopathy," Diabetes/ Metabolism Research and Reviews 15 (July/August 1999) 274-282.
(17.) N C Schaper, M H Nabuurs-Franssen, M S Huijberts, "Peripheral vascular disease and type 2 diabetes mellitus," Diabetes/Metabolism Research and Reviews 16 suppl (September/October 2000) S11-S15.
(18.) Stehouwer et al, "Endothelial dysfunction and pathogenesis of diabetic angiopathy," 55-68; Aengevaeren, "Beyond lipids--the role of the endothelium in coronary artery disease," S11-S16.
(19.) Aengevaeren, "Beyond lipids--the role of the endothelium in coronary artery disease," S11-S16.
(20.) Ibid.
(21.) L H Honing et al, "Nitric oxide availability in diabetes mellitus," Diabetes-Metabolism Reviews 14 (September 1998) 241-249; H Li, U Forstermann, "Nitric oxide in the pathogenesis of vascular disease," Journal of Pathology 190 (February 2000) 244-254.
(22.) Aengevaeren, "Beyond lipids--the role of the endothelium in coronary artery disease," S11-S16.
(23.) K Sugimoto, Y Murakawa, A A Sima, "Diabetic neuropathy: A continuing enigma," Diabetes/ Metabolism Research and Reviews 16 (November/December 2000) 408-433; P D Vaillancourt, H M Langevin, "Painful peripheral neuropathies," Medical Clinics of North America 83 (May 1999) 627-642.
(24.) D Porte Jr, "Clinical importance of insulin secretion and its interaction with insulin resistance in the treatment of type 2 diabetes mellitus and its complications," Diabetes/Metabolism Research and Reviews 17 (May/June 2001) 181-188; Sugimoto, Murakawa, Sima, "Diabetic neuropathy: A continuing enigma," 408-433.
(25.) Sugimoto, Murakawa, Sima, "Diabetic neuropathy: A continuing enigma," 408-433.
(26.) Qian, Eaton, "Glycochelates and the etiology of diabetic peripheral neuropathy," 652-656.
(27.) M E Garay-Sevilla et al, "Advanced glycosylation end products (AGEs), insulin-like growth factor-1 ( IGF-1) and IGF-binding protein-3 (IGFBP-3) in patients with type 2 diabetes mellitus," Diabetes/ Metabolism Research and Reviews 16 (March/April 2000) 106-113.
(28.) D A Greene, M J Stevens, E L Feldman, "Diabetic neuropathy: Scope of the syndrome," American Journal of Medicine 107 (Aug 30, 1999) 2S-8S.
(29.) Ibid.
(30.) Ibid.
(31.) Ibid.
(32.) Ibid.
(33.) M S Wallace, J S Dunn, T L Yaksh "Pain: Nociceptive and neuropathic mechanisms with clinical correlates," Anesthesiology, Clinics of North America 15 (June 1997) 229-463.
(34.) Greene, Stevens, Feldman, "Diabetic neuropathy: Scope of the syndrome," 2S-8S.
(35.) Wallace, Dunn, Yaksh "Pain: Nociceptive and neuropathic mechanisms with clinical correlates," 229-463.
(36.) G E Reiber, B A Lipsky, G W Gibbons, "The burden of diabetic foot ulcers," American Journal of Surgery 176 suppl (August 1998) 5S-10S; J E Shaw, A J Boulton, "The pathogenesis of diabetic foot problems: An overview," Diabetes 46 suppl (September 1997) S58-S61.
(37.) Reiber, Lipsky, Gibbons, "The burden of diabetic foot ulcers," 5S-10S.
(38.) J Jansma, K Van Voris, A M Reyzelman, "Management of diabetic wounds and infections in the elderly patient," Clinical Geriatrics 9 (June 2001) 52-60.
(39.) Reiber, Lipsky, Gibbons, "The burden of diabetic foot ulcers," 5S-10S.
(40.) G M Caputo et al, "The Charcot foot in diabetes: Six key points," American Family Physician 57 (June 1998) 2705-2710.
(41.) L Vileikyte, "Review articles--Diabetic foot ulcers: A quality of life issue," Diabetes/Metabolism Research and Reviews 17 (July/August 2001) 246-249.
(42.) Ibid.
(43.) F L Ferris, M D Davis, L M Aiello, "Review article--Drug therapy: Treatment of diabetic retinopathy," The New England Journal of Medicine 341 (Aug 26, 1999) 667-678.
(44.) L P Aiello, M T Cahill, J S Wong, "Systemic considerations in the management of diabetic retinopathy," American Journal of Ophthalmology 132 (November 2001) 760-776.
(45.) American Diabetes Association, "Position statements: Diabetic retinopathy," Diabetes Care 24, no 1 suppl (2001) S73-S76.
(46.) Aiello, Cahill, Wong, "Systemic considerations in the management of diabetic retinopathy," 760-776.
(47.) R J Zoorob, M D Hagen, "Guidelines on the care of diabetic nephropathy, retinopathy, and foot disease," American Family Physician 56 (Nov 15, 1997) 2021-2128; C Arauz-Pacheco, M A Parrott, P Raskin, "The treatment of hypertension in adult patients with diabetes," (Technical Review) Diabetes Care 25 (January 2002) 134-147.
(48.) K D Bums, "Angiotensin II and its receptors in the diabetic kidney," American Journal of Kidney Diseases 36 (September 2000) 449-467; American Diabetes Association, "Position statements: Diabetic nephropathy," Diabetes Care 24, no 1 suppl (2001) S69-S72.
(49.) Strock, "Type 2 diabetes: Strategies for patient care and education," S6-S13; Zoorob, Hagen, "Guidelines on the care of diabetic nephropathy, retinopathy, and foot disease," American Family Physician 56 (Nov 15, 1997) 2021-2128.
(50.) D A Greene, J C Arezzo, M B Brown, "Effect of aldose reductase inhibition on nerve conduction and morphometry in diabetic neuropathy," Neurology 53 (Aug 11, 1999) 580-591.
(51.) Ibid.
(52.) D F Horrobin, "Essential fatty acids in the management of impaired nerve function in diabetes," Diabetes 46 suppl (September 1997) S90-S93.
(53.) D Ziegler, F A Gries, "Alpha-lipoic acid in the treatment of diabetic peripheral and cardiac autonomic neuropathy," Diabetes 46 suppl (September 1997) S62-S66.
(54.) W S Kingery, "A critical review of controlled clinical trials for peripheral neuropathic pain and complex regional pain syndromes," Pain 73 (November 1997) 123-139.
(55.) W R Robbins et al, "Treatment of intractable pain with topical large-dose capsaicin: Preliminary report," Anesthesia & Analgesia 86 (March 1998) 579-583.
(56.) B Nicholson, "Gabapentin use in neuropathic pain syndromes," Acta Neurologica Scandinavica 101 (June 2000) 359-371.
(57.) M R Jackson, G P Clagett, "Antithrombotic therapy in peripheral arterial occlusive disease," Chest 119 suppl (January 2001) 283S-299S; M A Creager, "Medical management of peripheral arterial disease," Cardiology in Review 9 (July/August 2001) 238-245; American Diabetes Association, "Position statements: Aspirin therapy in diabetes," Diabetes Care 24, no 1 suppl (2001) S62-S63.
(58.) Jackson, Clagett, "Antithromboric therapy in peripheral arterial occlusive disease," 283S-299S.
(59.) Ibid.
(60.) Zoorob, Hagen, "Guidelines on the care of diabetic nephropathy, retinopathy, and foot disease," 2021-2128.
(61.) Stock, "Type 2 diabetes: Strategies for patient care and education," S6-S13; Rao, "Treating to target: A rational approach based on pathophysiology," S14-S21; American Diabetes Association, "Position statement: Smoking and diabetes," Diabetes Care 24 suppl (January 2001) S64-S65; C S Levetan, M F Magee, "Hospital management of diabetes," Endocrinology and Metabolism Clinics of North America 29 (December 2000) 745-770.
(62.) S Mudalier, S V Edelman, "Insulin therapy in type 2 diabetes," Endocrinology and Metabolism Clinics of North America 30 (December 2001) 935-982; J R White, R K Campbell, "Dangerous and common drug interactions in patients with diabetes mellitus," Endocrinology and Metabolism Clinics of North America 29 (December 2000) 789-802; American Diabetes Association, "Position statements: Standards of medical care for patients with diabetes mellitus," S33-S49.
(63.) Mudaliar, Edelman, "Insulin therapy in type 2 diabetes," 935-982; Physicians' Desk Reference Online, http://www.pdr.net (accessed 29 April 2002); American Diabetes Association, "Position statements: Insulin administration," Diabetes Care 25, no 1 suppl (2002) S112-S115.
(64.) J A Florence, B Yeager, "Treatment of type 2 diabetes mellitus," American Family Physician 59 (May 1999) 2849-2850; M C Riddle, "Oral pharmacologic management of type 2 diabetes," American Family Physician 60 (December 1999) 2613-2620; Physicians' Desk Reference Online, http://www.pdr.net.org (accessed 29 April, 2002); Levetan, Magee, "Hospital management of diabetes," 745-770; "Inpatient management guidelines for people with diabetes," National Guideline Clearinghouse, http://www.guidelines.gov/ FRAMESETS/guideline_fs. asp?guideline=000929&sSearch_strin g=Diabetes (accessed 14 June 2002).
(65.) G K McFarland, E A McFarlane, Nursing Diagnosis and Intervention: Planning for Patient Care, third ed (St Louis: Mosby, 1997); Levetan, Magee, "Hospital management of diabetes," 745-770.
(66.) Levetan, Magee, "Hospital management of diabetes," 745-770; "Inpatient management guidelines for people with diabetes."
Examination
DIABETES MELLITUS AND ITS CHRONIC COMPLICATIONS
AORN, Association of periOperative Registered Nurses, is accredited as a provider of continuing education in nursing by the American Nurses Credentialing Center's (ANCC's) Commission on Accreditation. AORN recognizes this activity as continuing education for registered nurses. This recognition does not imply that AORN or the ANCC's Commission on Accreditation approves or endorses any product included in the activity. AORN maintains the following state board of nursing provider numbers: Alabama ABNP0075, California CEP13019, and Florida FBN 2296. Check with your state board of nursing for acceptability of education activity for relicensure.
Professional nurses are invited to submit manuscripts for the Home Study Program. Manuscripts or queries should be sent to Editor, AORN Journal, 2170 S Parker Rd, Suite 300, Denver, CO 80231-5711. As with all manuscripts sent to the Journal, papers submitted for Home Study Programs should not have been previously published or submitted simultaneously to any other publication.
[ILLUSTRATIONS OMITTED]
Barbara K. Bailes, RN, EdD, CS, NP-C, is an associate professor at the University of Texas, Houston Health Science Center, School of Nursing, Houston.
COPYRIGHT 2002 Association of Operating Room Nurses, Inc.
COPYRIGHT 2002 Gale Group