Clinical experience indicates the beneficial effects of antibiotic-loaded bone cement. Although in vitro studies have shown the formation of a biofilm on its surface they have not considered the gap between the cement and the bone. We have investigated bacterial survival in that gap. Samples with gaps 200 µm wide were made of different bone cements. These were stored dry ('pre-elution') or submersed in phosphate-buffered saline to simulate the initial release of gentamicin ('post-elution'). The gaps were subsequently inoculated with bacteria, which had been isolated from infected orthopaedic prostheses and assessed for their sensitivity to gentamicin. Bacterial survival was measured 24 hours after inoculation. All the strains survived in plain cements. In the pre-elution gentamicin-loaded cements only the most gentamicin-resistant strain, CN5115, survived, but in post-elution samples more strains did so, depending on the cement tested. Although high concentrations of gentamicin were demonstrated in the gaps only the gentamicin-sensitive strains were killed. This could explain the increased prevalence of gentamicin-resistant infections which are seen clinically.
Gentamicin has been added to bone cement for more than three decades in order to reduce the risk of failure of an implant because of infection. It has been claimed that the initial release of antibiotic eliminates infra-operative contamination and that the release observed up to several months post-operatively is effective in preventing haematogenous infection.1 Despite the lack of detailed knowledge about the mechanism of release and therefore the long-term risks, such as the development of resistance among dormant bacteria resisiding in the biofilm or in the blood stream,2 gentamicin is being used widely as a prophylactic measure.3
The preferred mode of growth of bacteria in nature is in the biofilm,4 where they have increased resistance to antibiotic compared with organisms in suspension. This has often been ascribed to the protection offered by bacterial slime,^ but may also be due to the physiological changes of bacteria in an adhered state.6"8 This phenomenon has engendered a cautious view as to the routine prophylactic use of antibiotic-loaded bone cement because of the long-term slow release of antibiotic which has been associated with the development of resistance to gentamicin.9,10
Several in vitro studies have shown bacterial growth on antibiotic-loaded bone cements.11-16 The geometry of most of the models differs greatly from the situation in vivo. In the femur of a cadaver pig it has been shown that the boundary layer between the bone cement and bone was 50 to 500 µm wide along 15% of the interfacial circumference.17 The concentrations of antibiotic in these gaps have been shown to be much higher than those in the geometries studied so far.18
Our aim was to investigate bacterial survival in a simulated interfacial gap similar to the clinical situation. This was carried out in prepared bone-cement blocks before and after elution giving a circumstance analogous to the high initial release which occurs intra-operatively and with slow, long-term release.
Materials and Methods
Bone cements and method of preparation. Six commercially available bone cements were used: CMW 1 Radiopaque G, containing 1.7 w/w% gentamicin base, plain CMW 1 Radiopaque (DePuy CMW, Leeds, UK), Palacos R-G containing 0.84 w/w% gentamicin base, Palacos R (Schering-Plough, Maarssen, The Netherlands), Palamed G, containing 0.86 w/w% gentamicin base, and Palamed (Ortomed, Zwijndrecht, The Netherlands). The cements were prepared by mixing the powder with the liquid, according to the manufacturer's instructions in sterile conditions, manually with a spatula in a ceramic bowl, under atmospheric pressure and at ambient temperature. At the time specified for the start of application the doughy bone cements were spread over polytetrafluorethylene (PTFE) moulds containing stainlesssteel strips 200 µm thick.
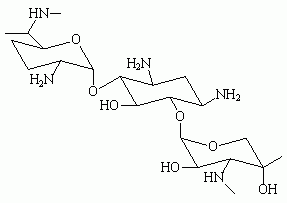
To facilitate removal of the glass plates after hardening of the bone cement, the mould was covered with a sterile overhead projector film (Océ, MC 110, 's Hertogenbosch, The Netherlands) and then compressed between two glass plates for the time specified for final hardening. The plates were then left in place for 24 hours without compression. The stainless-steel strips were removed and the bone-cement blocks were gently punched out of the mould. This produced blocks with a central gap as shown in Figure 1. The gap had a surface area of 0.61 cm^sup 2^ and a volume of 6 µl. The blocks were examined macroscopically and those with visibly entrapped gas bubbles or other defects in proximity to the surface were discarded.
The blocks were randomly divided into two groups. Half was kept in dry storage and designated 'pre-elution' cement. The other half was placed in a large volume of phosphate-buffered saline (PBS) for three weeks at 37°C using a Gyrotory Water Bath Shaker (Model G 76°; New Brunswick Scientific Co Inc, Edison, New Jersey). Finally, these blocks, designated 'post-elution' cement, were removed from the fluid and left to dry for three days in a sterile environment.
Bacterial strains, gentamicin sensitivity and growth conditions. We used six bacterial strains isolated from patients with infection of an orthopaedic prosthesis who had been treated in the University Hospital of Groningen (Tables I and II). The strains were chosen to reflect the spectrum of bacteria found in deep infections19 and to cover a range of gentamicin sensitivities. E-tests (AB Biodisk, Dàlvagen, Sweden) were used to establish the gentamicin sensitivity of the strains.
The bacteria were cultured from cryopreservative beads (Protect; Technical Service Consultants Ltd, Heywood, UK) onto blood-agar plates at 37°C in ambient air for three days. From these plates, one colony was subcultured on to blood-agar plates for another 24 hours to ensure the purity of the culture. One colony from the blood-agar plate was used to create a preculture in 10 ml of Tryptone Soy Broth (TSB) (Oxoid Ltd, Basingstoke, UK) under the same incubation conditions for 24 hours. The mean growth density was determined by counting the number of colony-forming units (CFU) after growth of serial dilutions on TSB agar plates. For all bacteria this was 2.1 x 10^sup 8^ cfu/ml after 24 hours.
This preculture was subsequently diluted in TSB, at 1:10, to provide new nutrients. The gaps in the cement were then filled with 6 µl of this dilution. These inoculated cement blocks were incubated for 24 hours in a water-vapour-saturated environment at 37°C before microbiological evaluation. This procedure was performed in triplicate with separately cultured strains both for the pre-elution and post-elution samples of cement.
Quantification of biofilms. The cement blocks were broken to expose the biofilm and the gap surface. Both sides were scraped with a stainless-steel blade to harvest the bacteria which were adhering to the surface. The blade was wiped with a cotton swab, soaked in 9 g/1 sodium chloride, which was then placed in 4.5 ml of 9 g/1 sodium chloride, vortexed and sonicated for 60 seconds in 35 KHz ultrasonic bath (Transsonic TP 690-A, Elma, Singen, Germany). Serial dilutions were made and poured on to TSB agar plates for overnight incubation at 37°C. The agar plates which had not shown any growth on the first day were left in the incubator for up to one week, after which they were checked again. This was done in order to detect possible slowly growing subpopulations which can be seen after exposure to a hostile environment such as a high concentration of antibiotic.20
All the results were expressed in ^sup 10^log cfu/cm^sup 2^ and experiments were carried out in triplicate with separately cultured strains, unless bacterial growth was completely absent on the gentamicin-loaded variant in the first experiment in which case the experiment was only performed once.
Statistical analysis. To determine the effects of the antibiotic on bacterial survival and growth, ^sup 10^log cfu/cm^sup 2^ values for gaps in gentamicin-loaded cements were compared with those for the plain cement, using a two-tailed Student ?-test for unpaired samples while assuming a normal distribution of the data, as shown before.18
Results
Gentamicin sensitivity. All the strains used are shown together with their sensitivity to gentamicin (Tables I and II). Two strains, a coagulase-negative stapbylococcus (CNS) 7334 and Staphylococcus aureus 7323, were sensitive to gentamicin. Two strains, Pseudomonas aeruginosa 5148 and CNS 7353, had intermediate sensitivities to gentamicin (1 to 4 mg/1), of which the latter was able to show resistant subpopulations with a gentamicin sensitivity of up to 32 mg/l, while two other strains, CNS 5234 and CNS 5115, were gentamicin-resistant with a sensitivity above 4 mg/1.21
Quantification of the biofilms on the cement samples. A summary of the mean number of the CFU harvested from the cement surfaces is given in Tables I and II for pre-elution and post-elution cements. The procedure applied to create so-called post-elution samples did not affect the formation of biofilm. Large numbers of bacteria were found on all plain cements (mean ^sup 10^log cfu/cm^sup 2^ range 4.8 for CNS 7334 on CMW 1 to 7.3 for P. aeruginosa 5148 on Palamed and Palacos R).
However, neither of the two gentamicin-sensitive strains nor the two strains with intermediate gentamicin sensitivity were able to survive in gaps in pre-elution gentamicinloaded cements (Table I). Also the resistant subpopulation of CNS 7353 and the resistant CNS 5234 did not survive after 24 hours of incubation. The two gentamicin-sensitive strains and the intermediately sensitive P. aeruginosa 5148 were also unable to survive in gaps made in post-elution gentamicin-loaded cements (Table II), despite the absence of the high initial release of antibiotics. The other intermediately sensitive strain, CNS 7353, showed significantly (p
The highly gentamicin-resistant strain CNS 5115 showed reduced survival in gaps made in pre-elution gentamicin-loaded cements compared with plain cements, but in post-elution cement there was no significant effect of gentamicin-loading. CNS 5234, being less resistant than CNS 5115, did not survive in pre-elution gentamicinloaded cements, but growth in gaps made in post-elution blocks was not significantly reduced as compared with plain cements (p values 0.083, 0.001 and 0.075 for CMW 1 Radiopaque G, Palacos R-G and Palamed G, respectively).
Discussion
In this study, the differences in bacterial survival in gentamicin-loaded and plain cements were studied in an in vitro simulation of a prosthesis-related interfacial gap. Earlier in vitro studies have shown statistically significant reductions in bacterial growth on antibiotic-loaded cements compared with plain cements, but bacterial survival and growth were generally not below detection as in our study.11-16 Since antibiotic-loaded cements remain in situ for many years and show long-term release of small amounts of antibiotic, experiments were also done with cements after the initial release of antibiotics. In gaps in pre-elution cements only the highly resistant CNS 5115 strain survived but in post-elution cement survival was also possible for less resistant strains.
The difference between the earlier in vitro studies and our study centres around a different ratio of exposed area of cement (A) over the volume (V) involved. Table III gives a summary of A/V ratios published in the literature. For studies with cement samples in a test tube, the area over volume ratio (A/V) is between 0.30 and 3.0 cm-1,11,12 while in a modified Robbins device it ranges from 0.030 to 0.00041 cm-1.13-16 In a clinical situation a rough estimate22 indicated that the surface area of the cement is in excess of 150 cm^sup 2^ and that the volume of an interface tissue layer 1 mm thick is approximately 15 cm^sup 3^, which would yield a conservative estimate for the A/V ratio of 10. In the gap model as applied here, the A/V ratio is much higher (100 cm^sup -1^). Previously,18 we have shown that these A/V ratios yield concentrations of gentamicin of up to 4000 mg/1 in gaps in pre-elution cements. This exceeds the minimum inhibitory concentrations (MIC) of the five non-surviving strains in our study by at least two orders of magnitude, but is only about 15 times higher than the MIC of the surviving CNS 5115.
Bacterial killing in gaps in post-elution cement occurred in sensitive strains and to a less extent in progressively more resistant strains. Concentrations of gentamicin in post-elution gaps were measured using identical methods previously employed for the pre-elution cements.18 After two hours concentrations of gentamicin for CMW 1 Radiopaque G, Palacos R-G and Palamed G of 75, 180 and 650 µg/ml were measurable. This approaches, or even exceeds, the MIC values of the strains involved in our study which has shown that gentamicin-sensitive strains were able to survive in earlier models with a lower A/V ratio and were eliminated in a simulated prosthesis-related interfacial gap, both in pre-elution and post-elution cements.
From a clinical point of view it is important to discuss, on the basis of the present results, whether gentamicin-loaded cement is useful in primary and in revision arthroplasty. Examinations in animal models of primary arthroplasty have indicated that the prophylactic use of antibiotic-loaded cement protects against infection after intra-operative challenge with gentamicin-sensitive bacteria.23-26 Randomised, controlled prospective clinical trials have also shown that antibiotic-loaded cement provides a protective effect against deep infection.27-29 This is further supported by recommendations to use gentamicin-loaded cement in combination with systemic antibiotics in primary arthroplasty, based on a cost-effectiveness and empirical analyses.30,31 Our study confirms the efficacy of gentamicin-loaded cement. However, it also points out that gentamicin-loaded cement may select for gentamicin-resistant strains. Similar observations have been made in clinical practice.9,23
Revision arthroplasty is known to have a higher risk of infection than primary arthroplasty." If gentamicin-loaded cement has been used in primary surgery, bacteria involved in these infections may already have survived a high concentration of gentamicin inside a prosthesis-related gap and are probably gentamicin-resistant. Subsequent use of gentamicin-loaded cement is therefore less efficacious, while still carrying the risk of further selecting resistance.
In conclusion, our study confirms that in a simulated prosthesis-related interfacial gap, high concentrations of gentamicin can be attained. In both pre-elution and in postelution preparations these concentrations appear to be successful in killing commonly infecting bacterial strains with MICs which are more than 100-fold lower, but a selection of resistant strains can be detected.
No benefits in any form have been received or will be received from a commercial party related directly or indirectly to the subject of this article.
References
1. Wahlig H, Dingeldein E. Antibiotics and bone cements: experimental and clinical long-term observations. Acta Orthop Scand 1980;51:49-56.
2. Wahlig H, Buchholz HW. Experimental and clinical studies on the release of gentamicm from bone cement. Chirurg 1972;43:441-5.
3. Heck D, Rosenberg A, Schink-Ascani M, Garbus S, Kiewitt T. Use of antibioticimpregnated cement during hip and knee arthroplasty in the United States. J Arthroplasty 1995;10:470-5
4. Costerton JW, Lewandowski Z, Caldwell D, Korber D, Lappin-Scott H. Microbial biofilms. Annu Rev Microbiol 1995;49:711-45
5. Arizono T, Oga M, Sugioka Y. Increased resistance of bacteria after adherence to polymethylmethacrvlate: an in vitro study. Acta Orthop Scand 1992;63:661-4.
6. Naylor PT, Myrvik QN, Gristina A. Antibiotic resistance of biomaterial-adherent coagulase-negative and coagulase-positive staphylococci. Clin Orthop 1990;261: 126-33
7. Brown MR, Allison DG, Gilbert P. Resistance of bacterial biofilms to antibiotics: a growth-rate related effect? J Antimicmb Chemother 1988;22:777-80.
8. Mah TF, O'Toole GA. Mechanisms of biofilm resistance to antimicrobial agents. Trends Microbiol 2001;9:34-9.
9. Tunney MM, Ramage G, Patrick S, et al. Antimicrobial susceptibility of bacteria isolated from orthopaedic implants following revision hip surgery. Antimicrob Agents Chemother 1998;42:3002-5.
10. Van de Belt H, Neut D, Van Horn JR, et al. ....or not to treat? Nat Med 1999;5: 358-9
11. Kendall RW, Duncan CP, Smith JA, Ngui YJ. Persistence of bacteria on antibiotic loaded acrylic depots: a reason for caution. Clin Orthop 1996;329:273-80.
12. Chang CC, Merritt K. Microbial adherence on poly(methylmethacrylate) (PMMA) surfaces. J Biomed Mater Res 1992;26:197-207.
13. Oga M, Arizono T, Sugioka Y. Inhibition of bacterial adhesion by tobramycinimpregnated PMMA bone cement. Acta Orthop Scand 1992;63:301-4.
14. Poelstra KA, Busscher HJ, Schenk W, Van Horn JR, Van der Mei HC. Effect of gentamicin loaded PMMA bone cement on Staphylococcus aureus biofilm formation. Biofouling 1999;14:249-54
15. Van de Belt H, Neut D, Schenk W, et al. Staphylococcus aureus biofilm formation on different gentamicin-loaded polymethylmethacrylate bone cements. Biomaterials 2001;22:1607-11.
16. Van de Belt H, Neut D, Schenk W, et al. Gentamicin release from polymethylmethacrvlate bone cements and Staphylococcus aureus biofilm formation. Acta Orthop Scand 2000;71:625-9.
17. Wang JS, Franzen H, Lidgren L. Interface gap after implantation of a cemented femoral stem in pigs. Acts Orthop Scand 1999;70:234-9.
18. Hendriks JGE, Neut D, Van Horn JR, Van der Mei HC, Busscher HJ. The release of gentamicin from acrylic bone cements in a simulated prosthesis-related interfacial gap. J Biomed Mater Res 2003;64-6:1-5.
19. Tsukayama DT, Estrada R, Gustilo RB. Infection after total hip arthroplasty: a study of the treatment of one hundred and six infections. J Bone Joint Surg [Am] 1996;78-A:512-23.
20. Neut D, Van de Belt H, Stokroos I, et al. Biomaterial-associated infection of gentamicin-loaded PMMA beads in orthopaedic revision surgery. J Antimicrob Chemofner 2001;47:885-91.
21. De Neeling AJ, Van Leeuwen WJ, Schouls LM, et al. Resistance of staphylococci in The Netherlands: surveillance by an electronic network during 1989-1995. J Antimicrob Chemother 1998;41:93-101.
22. Rodeheaver GT, Rukstalis D, Bono M, Bellamy W. A new model of bone infec tion used to evaluate the efficacy of antibiotic-impregnated polymethylmethacrylate cement. Clin Orthop 1983:178:303-11.
23. Schurman DJ, Trindade C, Hirshman HP, et al. Antibiotic-acrylic bone cement composites: studies of gentamicin and Palacos. J Bone Joint Surg [Am] 1978;60-A: 978-84.
24. Nijhof MW, Shert WJA, Fleer A, Vogely HC, Verbout AJ. Prophylaxis of implant-related staphylococcal infections using tobramycin-containing bone cement. J Biomed Mater Res 2000;52:754-61.
25. Petty W, Spanier S, Shuster JJ. Prevention of infection after total joint replacement: experiments with a canine model. J Bone Joint Surg [Am] 1988;70-A:536-9.
26. Josefsson G, Gudmundsson G, Kolmert L, Wijkstrom S. Prophylaxis with systemic antibiotics versus gentamicin bone cement in total hip arthroplasty: a five-year survey of 1688 hips. Clin Orthop 1990;253:173-8.
27. Josefsson G, Kolmert L Prophylaxis with systematic antibiotics versus gentamicin bone cement in total hip arthroplasty: a ten-year survey of 1,688 hips. Clin Orthop 1993;292:210-14.
28. Chiu FY, Chen CM, Lin CF, Lo WH. Cefuroxime-impregnated cement in primary total knee arthroplasty: a prospective, randomized study of three hundred and forty knees. J Bone Joint Surg [Am] 2002;84-A:759-62.
29. Espehaug B, Engesaeter LB, Vollset SE, Havelin LI, Langeland N. Antibiotic prophylaxis in total hip arthroplasty: review of 10,905 primary cemented total hip replacements reported to the Norwegian arthroplasty register, 1987 to 1995. J Bone Joint Surg [Br] 1997:79-8:590-5.
30. Persson U, Persson M, Malchau H. The economics of preventing revisions in total hip replacement Acta Orthop Scand 1999:70:163-9.
31. Hope PG, Kristinsson KG, Norman P, Elson RA. Deep infection of cemented total hip arthroplasties caused by coagulase-negative staphylococci. J Bone Joint Surg [Br] 1989;71-8:851-5.
32. Beeching NJ, Thomas MG, Roberts S, Lang SD. Comparative in-vitro activity of antibiotics incorporated in acrylic bone cement. J Antimicrob Chemother 1986;17: 173-84.
33. Berbari EF, Hanssen AD, Dufty MC, et al. Risk factors for prosthetic joint infection: case control study. Clin Infect Dis 1988;27:1247-54.
J. G. E. Hendriks, D. Neut, J. R. van Horn, H. C. van der Mei, H. J. Busscher
From the University of Groningen, The Netherlands
* J. G. E. Hendriks, MD PhD
* D. Neut, PhD
* H. C. van der Mei, PhD
* H. J. Busscher, PhD
Department of Biomedical Engineering, University of Groningen, Antonius Deusinglaan 1, 9713 AV Groningen, The Netherlands.
* J. R. van Horn, MD, PhD
Department of Orthopaedic Surgery, University Hospital Groningen, Hanzeplein 1, 9713 EZ Groningen, The Netherlands.
Correspondence should be sent to Dr H. J. Busscher; e-mail: h.j.busscher@med.rug.nl
©2005 British Editorial Society of Bone and Joint Surgery
doi:10.1302/0301-620X.87B2. 14781 $2.00
J Bone Joint Surg [Br] 2005;87-B:272-6.
Received 77 July 2003; Accepted after revision 29 April 2004
Copyright British Editorial Society of Bone & Joint Surgery Feb 2005
Provided by ProQuest Information and Learning Company. All rights Reserved