Objective: We conducted a retrospective study to determine the relationship between gastroesophageal reflux (GER) and large airways malacia in infancy.
Methods: One hundred sixteen children referred for chronic respiratory problems who were between the ages of 3 and 28 months were investigated. All of them underwent flexible bronchoscopy and chest radiography. Eighteen children had laryngomalacia, 13 had tracheomalacia, and 23 had combined laryngotracheomalacia. During bronchoscopy, BAL was performed. An analysis of macrophages in the BAL fluid for lipid content was performed. Fifty-four children with laryngomalacia and tracheomalacia constituted the study group, and 62 children were in the control group. Reflux studies were obtained for 40 children from the study group and 41 from the control group.
Results: In the study group, 28 children (70%) had GER documented by reflux studies compared with 16 children (39%) in the control group (p [is less than] 0.01). In the control group, GER was found mainly among those with recurrent bilateral pneumonia. The lipid-laden macrophage score was correlated with the documented GER.
Conclusion: GER is prevalent among infants with large airways malacia, and treatment of this group with antireflux therapy should be considered. (CHEST 2001; 119:409-413)
Key words: gastroesophageal reflux; laryngomalacia; tracheomalacia
Abbreviations: GER = gastroesophageal reflux; LLM = lipid-laden macrophage
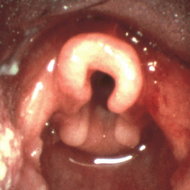
Laryngomalacia is the most common cause of noninfective stridor in infancy.[1,2] It is diagnosed endoscopically by observing an abnormal prolapse of supraglottic structures during inspiration.[3] Tracheomalacia as well causes chronic respiratory symptoms, mainly wheezing due to significant narrowing of the compliant trachea.[4,5] The most common finding in patients with tracheomalacia is a malformation of the cartilage rings, which causes the tracheal lumen to be oval rather than round. Laryngomalacia and tracheomalacia may coexist, but they have differences in etiology and pathophysiology.[2,5]
Gastroesophageal reflux (GER) resulting in the aspiration of gastric content can cause an inflammatory reaction in the airways of infants and young children. This phenomenon is a relatively common cause of chronic pulmonary disease.[6,7] This is particularly true in children with GER secondary to an immature swallowing mechanism, mental retardation, a seizure disorder, and neuromuscular disease. There is a relationship between GER and the chronic lung diseases cystic fibrosis, bronchopulmonary dysplasia, and asthma.[8-10] A causal relationship has been found between GER and laryngomalacia[11] and tracheomalacia.[12]
Our purpose was to determine the prevalence of GER among infants with chronic respiratory symptoms and to determine whether laryngomalacia and tracheomalacia were associated with an increase in the prevalence of GER.
MATERIALS AND METHODS
Medical records of 116 infants with chronic respiratory symptoms who underwent bronchoscopy between July 1996 and August 1998 were reviewed retrospectively. All infants underwent chest radiographs before flexible bronchoscopy. Data related to their clinical and laboratory findings before and after bronchoscopy were collected and analyzed.
The indications for bronchoscopy were the following: respiratory symptoms unresponsive to medical treatment; persistent infiltrates apparent on chest radiograph; or suspected foreign body in the airways. Twenty-two infants had early-onset stridorous cough, and 94 infants had a chronic cough.
Those infants who were suspected of having GER (ie, recurrent vomiting, feeding-related cough, or unexplained cough not responding to medical treatment) underwent reflux studies as well. Reflux studies included barium swallow and pH monitoring with a single-channel probe. GER was defined by the following:
1. documentation of barium reflux via the gastroesophageal sphincter to the upper esophagus during barium swallow; and
2. pH [is less than] 4.0 for [is greater than] 8% of the duration of the pH monitoring study.
Bronchoscopy was performed in the local pediatric department. Sedation for bronchoscopy was achieved with IV midazolam (0.05 to 0.3 mg/kg) with or without meperidine (0.5 to 2.0 mg/kg). For topical anesthesia of the airways, 1% or 2% lidocaine (Xylocaine; Astra-Zeneca; Wilmington, DE) was used. Bronchoscopy was performed with a fiberoptic pediatric bronchoscope (model [BF]3C4; Olympus; Hamburg, Germany) with video imaging of the procedure. The video recordings were reviewed by the bronchoscopist for confirmation of the diagnosis by measurement of the tracheal diameter on the recording.
Laryngomalacia was defined as the severe collapse of the epiglottis and arytenoids.[13] Tracheomalacia was defined as a narrowing of the trachea with a cartilaginous to membranous ratio of at most 3:1 (normal ratio, 4.5:1) as seen and documented by bronchoscopy.[4] The diagnosis was confirmed by a second review.
The bronchoscope was placed in the lobe that appeared most involved in the underlying disease as seen by chest radiograph or by direct vision, or was placed in the right lower lobe if the disease was not localized.
Bronchial lavage was performed with 10-mL aliquots of sterile normal saline solution, which was injected and manually suctioned using bronchoscopic suction. This procedure was repeated until a total volume of 3 mL/kg was achieved. The material from the bronchial washing was centrifuged, and the cellular pellet was resuspended to a density of approximately 1 x [10.sup.4] macrophages/ mL. One hundred microliters of the cell suspension was placed on a slide and was allowed to fix in formaldehyde vapor.
Standard oil red O stain and hematoxylin-eosin stain were performed.[14] We used a quantitation of lipid-laden macrophages (LLM), as described by Colombo and Hallberg.[15] The results of the macrophage analysis were compared with the clinical bronchoscopic and radiologic findings.
Statistical Analysis
Data were evaluated by the Student's t test, analysis of variance, and [chi square] analysis to compare the different groups.
RESULTS
One hundred sixteen children between the ages of 3 and 34 months were studied. The mean ([+ or -] SD) age was 16 [+ or -] 8 months. There were 76 male infants (mean age, 19 [+ or -] 12 months) and 40 female infants (16 [+ or -] 14 months). Fifty-eight infants had normal findings on chest radiographs, 37 had persistent unilateral lung infiltrates, and 21 had persistent bilateral infiltrates. Ten infants were suspected of having foreign-body aspiration.
The infants initially underwent flexible bronchoscopy in order to confirm the diagnosis. In four infants, a foreign body was found and extracted by rigid bronchoscopy under general anesthesia. Fifty-four infants were found to have large-airway malacia, and they constituted the study group. Laryngomalacia was found in 18 infants (13 male). Tracheomalacia was found in 13 infants (10 male), in 1 of them after a repair of a vascular ring. Twenty-three infants (16 male) had combined laryngotracheomalacia. Sixty-two children without malacia constituted the control group (35 male).
Reflux studies were performed in 11 infants with laryngomalacia, in 13 infants with tracheomalacia, in 16 infants with laryngotracheomalacia, and in 41 children from the control group.
GER was found in 7 of 11 infants (63%) with laryngomalacia, in 7 of 13 infants (53%) with tracheomalacia, in 14 of 16 infants (87%) with laryngotracheomalacia, as well as in 16 of 41 children (39%) from the control group (p [is less than] 0.01). The results of pH monitoring and barium swallow testing are shown in Table 1. The relationships among reflux studies, chest radiograph findings, and bronchoscopic findings are shown in Table 2.
[TABULAR DATA 1-2 NOT REPRODUCIBLE IN ASCII]
The results of the LLM study are shown in Table 3. The LLM score was correlated with the presence of either laryngomalacia or tracheomalacia and with recurrent bilateral pneumonia. The LLM score also was correlated with the presence of GER (Table 3).
[TABULAR DATA 3 NOT REPRODUCIBLE IN ASCII]
DISCUSSION
Some infants, especially those in the younger age group, suffer from chronic respiratory symptoms related to anatomic dysfunction of the airways due to flexible tissue of the large airways.[1] Laryngomalacia is the most prevalent malformation associated with recurrent respiratory symptoms in infancy and is occasionally accompanied by tracheomalacia. The combination of laryngomalacia and tracheomalacia can be related to malfunction of the upper airways. Visualization of the airways by a flexible bronchoscope has become prevalent and has resulted in more accurate diagnosis and better care.[1] Laryngomalacia and tracheomalacia represent a delay in the maturation of the supporting structures of the larynx and trachea. The diagnosis in most cases should be based on clinical history and physical examination, with bronchoscopy reserved for patients with severe or atypical symptoms, or for those with an unusual clinical course. Diagnosis with bronchoscopy provides strong reassurance, and support is often needed to allay parental anxiety. Laryngomalacia and tracheomalacia tend to improve with age in most cases.[16]
GER is prevalent among children mainly at infancy.[17,18] GER may be classified as physiologic or pathologic, depending on its degree and consequences. GER can cause failure to thrive, recurrent pneumonia, and asthma as well as apnea and life-threatening events.[8,17,19] GER can cause chronic pulmonary disease in the airways of infants and young children.[6,7,19] There is also a relationship between chronic lung disease and GER.[8-10]
In our study, the group of infants with laryngomalacia, tracheomalacia, and laryngotracheomalacia had a higher incidence of GER compared to the control group. Barium studies and pH monitoring documented the diagnosis of GER, with the presence of LLMs being supportive of the diagnosis.
The barium esophagogram often has been used to diagnose reflux, but, due to its lack of sensitivity and specificity, it is not the "gold standard" for this purpose.[20] It is best used to observe anatomy and function and to eliminate other GI and vascular disorders from consideration; therefore, in our study a relatively large number of children had barium studies. Of the methods used to detect acid reflux, 24-h pH monitoring has become the "gold standard" because of its reported 87% sensitivity, 93% specificity, and 92% accuracy.[14] A negative pH result, however, is not sufficient for ruling out GER.[21,22]
In our study, 43 children were studied using both methods (eg, barium esophagogram and pH monitoring) for the detection of GER. From this group, 14 children had positive pH monitoring results with normal barium studies, 13 children had positive pH monitoring results with positive barium studies, and 16 children had negative results in both studies.
The finding of alveolar LLM is a nonspecific marker for parenchymal pulmonary disease.[23] In pediatric patients, it was found that the quantitation of LLMs may serve as a useful tool as an adjunct to other studies in the diagnosis of recurrent aspiration.[15] It was found that BAL fluid from children with respiratory symptoms and documented GER contains significantly more LLMs compared to BAL fluid from children without GER.[24] It was suggested that milk and its products aspirated into the bronchial tree accumulate in the alveolar macrophages and become lipids, which can be detected by oil red O stain.[25] In the study of Colombo and Hallberg.[15] an LLM score [is greater than] 70 was associated with aspiration. In our study, the children with malacia and with bilateral infiltrates on chest radiographs had scores that were above the aspirator score according to Colombo and Hallberg.[15] This score evidently correlates either with lung injury or with the high rate of GER among these children. The lower LLM score among children without GER, compared to those with GER, supports the connection between the presence of GER and the LLM score. In our study, we found that GER was more prevalent among children with malacia of the large airways. The diagnosis was based on reflux studies and LLMs that were found in BAL fluid. This finding is supported by other studies.[11,26]
The high prevalence of GER among infants with large-airways malacia can be related to the negative intrapleural pressure induced by airflow obstruction during respiration. The high negative pressure leads to a malfunction of the lower gastroesophageal sphincter and causes GER. As shown in an animal model,[27] the possible mechanism that associates laryngomalacia or tracheomalacia with GER is that airway obstruction in these infants induces wide swings in intrathoracic and abdominal pressures that overcome the antireflux barrier, resulting in GER. All the children who were suspected of having GER were treated. They responded well to antireflux therapy, which included eating thicker food and assuming the upright position, and to drug therapy, which included prokinetic agents such as cisapride or metoclopramide. In the severe cases, H-2 receptor blockers such as cimetidine or ranitidine were used. The response was defined as decreases in respiratory symptoms as well as in the need for medical treatment and care. The improvement can be related to the natural history of malacia, but the importance of antireflux therapy may be to prevent chronic injury to the lung.
In view of the complications that can result from GER into the respiratory tract[6,7] and the relationship between chronic lung disease and GER,[8-10] our results suggest that treatment with antireflux positioning and, possibly, medication to prevent chronic respiratory symptoms should be considered among infants with large-airways malacia.
In conclusion, we have shown that GER is common among infants with laryngomalacia, tracheomalacia, and laryngotracheomalacia, and that, therefore, treatment of this group of infants with antireflux therapy should be considered.
REFERENCES
[1] Wood RE. Spelunking in the pediatric airway: exploration with the flexible fiberoptic bronchoscope. Pediatr Clin North Am 1984; 31:785-799
[2] Solomons NB, Prescot CAJ. Laryngomalacia: a review and the surgical management for severe cases. Int J Pediatr Otorhinolaryngol 1987; 13:31-39
[3] McSwiney PF, Cavanagh NPC, Languth P. Outcome in congenital stridor (laryngomalacia). Arch Dis Child 1977; 52:215-218
[4] Benjamin B. Tracheomalacia in infants and children. Ann Otol Rhinol Laryngol 1984; 93:438-442
[5] Mair EA, Parsons DS. Pediatric tracheobronchomalacia and major airway collapse. Ann Otol Rhinol Laryngol 1992; 101:300-309
[6] Euler AR, Byrne WJ, Ament ME, et al. Recurrent pulmonary disease in children: a complication of gastroesophageal reflux. Pediatrics 1979; 63:47-51
[7] Spaulding HS Jr, Mansfield LE, Stein MR, et al. Further investigation of association between gastroesophageal reflux and bronchoconstriction. J Allergy Clin Immunol 1982; 69: 516-521
[8] Andze GO, Brandt ML, St. Vil D, et al. Diagnosis and treatment of gastroesophageal reflux in 500 children with respiratory symptoms: the value of pH monitoring. J Pediatr Surg 1991; 29:299-300
[9] Christie DI, O'Grady LR, Mack DV. Incompetent lower esophageal sphincter and gastroesophageal reflux in recurrent and acute pulmonary disease of infancy and childhood. J Pediatr 1978; 93:23-27
[10] Malfroot A, Vandenplass Y, Verlinden M, et al. Gastroesophageal reflux and unexplained chronic respiratory disease in infants and children. Pediatr Pulmonol 1987; 3:208-213
[11] Belmont JR, Grundfast K. Congenital laryngeal stridor (laryngomalacia): etiologic factors and associated disorders. Ann Otol Rhinol Laryngol 1984; 93:430-437
[12] Callahan CW. Primary tracheomalacia and gastroesophageal reflux in infants with cough. Clin Pediatr (Phila) 1998; 37:725-732
[13] Richardson MA, Cotton RT. Anatomic abnormalities of the pediatric airway. Pediatr Clin North Am 1984; 31:821-833
[14] Lillie RD, Fullmer HM. Histopathologic technique and practical histochemistry. 4th ed. New York, NY: McGraw-Hill, 1976; 559-610
[15] Colombo JL, Hallberg TK. Recurrent aspiration in children: lipid-laden alveolar macrophages quantitation. Pediatr Pulmonol 1987; 3:86-89
[16] Vandenplas Y, Govayvaerts H. Gastroesophageal reflux, as measured by 24 hour pH monitoring, in 509 healthy infants screened for risk of sudden infant death syndrome. Pediatrics 1991; 88:834-840
[17] Friedman EM, Vastola AP, McGill TJ, et al. Chronic pediatric stridor: etiology and outcome. Laryngoscope 1990; 100:277-280
[18] Balistreri WF, Farrel MK. Gastroesophageal reflux in infants. N Engl J Med 1983; 309:790-792
[19] Burton DM, Pransky SM, Katz RM, et al. Pediatric airway manifestations of gastroesophageal reflux. Ann Otol Rhinol Laryngol 1992; 101:742-749
[20] Leonidas JC. Gastroesophageal reflux in infants: role of the upper gastrointestinal series. AJR Am J Roentgenol 1984; 143:1350-1351
[21] Van den Abbeele T, Bruhier N, Narcy P. Severe laryngeal manifestations of gastro-esophageal reflux in children. Pediatr Pulmonol 1997; 16(suppl): 237-238
[22] Contencin P, Narcy P. Gastroesophageal reflux in infants and children: a pharyngeal pH monitoring study. Arch Otolaryngol Head Neck Surg 1992; 118:1028-1030
[23] Corwin RW, Irwin RS. The lipid-laden alveolar macrophages as a marker of aspiration in parenchymal lung disease. Am Rev Respir Dis 1985; 132:576-581
[24] Nussbaum E, Maggi C, Mathis R, et al. Association of lipid-laden alveolar macrophages and gastroesophageal reflux in children. J Pediatr 1987; 110:190-194
[25] Moran JR, Nlock SR, Lyerly AD, et al. Lipid-laden macrophages and lactose assay as markers of aspiration in neonates with lung disease. J Pediatr 1988; 112:643-645
[26] Halstead LA. Role of gastroesophageal reflux in pediatric upper airway disorders. Otolaryngol Head Neck Surg 1999; 120:208-214
[27] Wang W, Tovar JA, Eizaguirre I, et al. Airway obstruction and gastroesophageal reflux: an experimental study on the pathogenesis of the association. J Pediatr Surg 1993; 28:995-998
(*) From the Department of Pediatrics (Drs. Bibi, Khvolis, Ohaly, and Ater), the Department of Pathology (Dr. Ben Dor), and the Department of Radiology (Dr. London), Barzilai Medical Center, Ashkelon, Israel; and the Pediatric Respirology Unit (Dr. Shoseyov), Bikur Holim Hospital, Jerusalem, Israel.
Manuscript received October 29, 1999; revision accepted July 10, 2000.
Correspondence to: Haim Bibi, MD, Barzilai Medical Center, Ashkelon, Israel 78306
COPYRIGHT 2001 American College of Chest Physicians
COPYRIGHT 2001 Gale Group