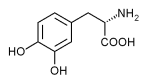
Study objectives: Upper-airway obstruction (UAO) may be present in patients with Parkinson's disease (PD), and its reversibility after levodopa therapy has been suggested. To investigate the effects of oral intake of levodopa on pulmonary function and UAO criteria in patients with PD, we studied 22 patients with PD.
Design: Pulmonary function tests were performed after a 12-h withdrawal of levodopa therapy, and 1 h after oral intake of placebo or levodopa, according to a double-blind, placebo-controlled, crossover study. Six UAO criteria were recorded to detect UAO in patients. UAO was found in 5 of 21 patients on baseline conditions (1 patient could not perform all tests).
Results: Among the patients with UAO, after levodopa therapy three of five patients did not meet the four of six required criteria for defining UAO. Levodopa produced its effects on UAO criteria by means of a saw-tooth pattern improvement and/or a decrease below the defined thresholds of the peak inspiratory flow and the [FEV.sub.1]/peak expiratory flow (PEF) and [FEV.sub.1]/forced expiratory flow after 50% of the FVC ([FEV.sub.0.5]) ratios. Levodopa PEF increased by 0.85 L/s in patients with UAO and by 0.24 L/s in patients without UAO, while after placebo it increased by 0.03 L/s in patients with UAO and decreased by 0.16 L/s in patients without UAO (p = 0.02). Whereas in patients without UAO an increase of the [FEV.sub.1]/PEF and [FEV.sub.1]/[FEV.sub.0.5] ratios was observed after placebo and levodopa intake, these ratios decreased after levodopa and increased after placebo in patients with UAO.
Conclusions: These results show that levodopa administration in patients with PD induces significant variations in PEF and UAO ratios ([FEV.sub.1]/PEF and [FEV.sub.1]/[FEV.sub.0.5]). (CHEST 2001; 119:387-393)
Key words: levodopa; Parkinson's disease; pulmonary function; upper-airway obstruction
Abbreviations: ANOVA = analysis of variance; [FEV.sub.0 5] = forced expiratory volume in 0.5 s; [FEF.sub.50] = forced expiratory flow after 50% of the FVC; [FIF.sub.50] = forced inspiratory flow after 50% of the FVC; PD = Parkinson's disease; PEF = peak expiratory flow; PIF = peak inspiratory flow; UAO = upper-airway obstruction
Respiratory abnormalities reported in patients with Parkinson's disease (PD) are associated with substantial morbidity and mortality.[1] Pulmonary function abnormalities were initially attributed to an obstructive defect[2,3] caused by an increased parasympathetic activity.[3] More recently, a dysfunction of the upper-airway muscles was suggested to be responsible for upper-airway obstruction (UAO).[4]
However, the effects of PD therapy on this respiratory dysfunction are unclear. Two studies report conflicting results. Obenour et al[3] did not find any improvement of the respiratory function after treatment with levodopa despite improvement of the neurologic features, while Nakane et al[5] found an improvement of respiratory function with levodopa compared with placebo. De Bruin et al[6] reported that apomorphine, a dopaminergic agonist, may improve the dysfunction of airway musculature in patients with PD. Finally, in one single patient with PD, a sequence of flow-volume curves suggested reversibility of a pattern of UAO after oral intake of levodopa.[7]
However, levodopa may induce respiratory dysfunction in PD patients.[8-12] Accordingly, it is of clinical interest to undertake more systematic studies on the effects of levodopa on pulmonary function in PD. We therefore performed a double-blind, placebo-controlled, crossover trial to study the effects of oral intake of levodopa on pulmonary function and UAO ratios in patients with PD.
MATERIALS AND METHODS
Patients
The study, which was approved by the local ethics committee, included 22 consecutive patients (age range, 52 to 89 years) drawn from general clinics, who met the criteria for idiopathic PD[13] and gave informed consent. Potential study subjects were identified in the patient population of one of the investigators. One patient was withdrawn after enrollment because she could not perform the whole study. Severity of PD was classified on a scale from I to V, as stage II (n = 9), stage III (n = 10), and stage IV (n = 2), according to Hoehn and Yahr.[14] No specific rating scale for disability in PD was performed during the study. Fifteen patients were treated long term with levodopa (100 to 600 mg/d for [is greater than] 3 months) and had no history of levodopa-induced dyskinesias or significant motor fluctuations. The other patients were naive to levodopa. None of the patients had a significant history of respiratory disease. Other exclusion criteria were as follows: use of medication that might result in pulmonary dysfunction, structural abnormalities of upper airways including oral cavity, and dementia. Two patients (both treated with levodopa) reported previous episodes of dyspnea, not chronologically related with levodopa intake.
Pulmonary Function Testing
Pulmonary function tests included spirometry with standard spirometer and maximal inspiratory and expiratory flow-volume curves (Medical Graphics Corporation; St. Paul, MN). At least three reproducible flow-volume curves were obtained. Values of FVC, [FEV.sub.1], forced expiratory volume in 0.5 s ([FEV.sub.0.5]), peak expiratory flow (PEF), peak inspiratory flow (PIF), forced expiratory flow after 50% of FCV ([FEF.sub.50]), and forced inspiratory flow after 50% of the FVC ([FIF.sub.50) were obtained and were expressed as percent predicted, except for PIF, for which no reliable references were available.
Criteria for UAO
In order to detect an UAO, the following ratios were also calculated: [FEV.sub.1]/PEF, [FEV.sub.1]/[FEV.sub.0.5], [FEF.sub.50] and PEF/ [FEF.sub.50]. UAO was considered to be present if at least four of the six following criteria were present at all baseline studies: a flow-volume curve with a characteristic pattern of UAO (saw-tooth sign, ie, flow oscillations of the flow-volume loop tracing),[4] a PIF [is less than] 3 L/s,[4] a [FEV.sub.1]/PEF ratio [is greater than] 8.5 mL/L/min,[4] a [FEV.sub.1]/ [FEV.sub.0.5] ratio [is greater than] 1.5,[4] a [FEF.sub.50]/[FIF.sub.50] ratio [is greater than] 1,[4] and a PEF/[FEF.sub.50] ratio [is less than] 2.[5]
Protocol
Treatment with levodopa and other antiparkinsonian drugs was withdrawn 12 h before the study. Study design was a placebo-controlled, double-blind, crossover trial of levodopa. The first flow-volume curves were done without any antiparkinsonian therapy (baseline). Then placebo or levodopa-benserazide (100 mg/25 mg, 1 to 2.6 mg/kg of levodopa according to the weight of patients) was given to the patient, and flow-volume curves were done after 45 to 60 min. A second study with the drug or placebo was done at least 24 h after the first experiment, according to the same protocol in all patients. The order of both studies was randomly assigned, and the investigators and patient remained double blinded during the whole study. No GI or other side effects following the administration of medications were observed.
Statistical Analysis
Results are expressed as mean [+ or -] SD unless indicated. Category data were compared using [chi square] test. Analysis of variance (ANOVA) was used to test differences in continuous variables. ANOVA for repeated measurements was used to study the effects of the UAO status and medication on the variation of pulmonary function data. No effect of the order of medication was found. The usual p [is less than] 0.05 threshold for significance was retained for all statistics. Statistics were performed (BDMP v 7.0 Statistical Software Package; BDMP Statistical Software; BDMP; Los Angeles, CA).
RESULTS
Clinical Characteristics
The study population consisted of 7 women and 14 men. Five patients (two women, three men) had UAO according to our criteria. The demographic and clinical characteristics of the patients are expressed in Table 1, showing no significant difference between patients with and without UAO.
(*) Data are expressed as mean [+ or -] SD unless otherwise indicated.
Pulmonary Function and UAO Criteria at Baseline
Tables 2, 3 summarize spirometry data and UAO criteria at baseline evaluation and following oral intake of placebo and levodopa in patients with and without UAO. At baseline, patients with UAO exhibited the following characteristics when compared with patients without UAO: lower PEF (absolute and percent predicted values), higher [FEV.sub.1]/PEF, [FEF.sub.50]/[FIF.sub.50] and [FEV.sub.1]/[FEV.sub.0.5], and lower PEF/ [FEF.sub.50]. The differences in the UAO criteria between the two groups of patients were expected, since these criteria defined the UAO status in our patients. Baseline data did not differ according to the drug given thereafter.
[TABULAR DATA 2-3 NOT REPRODUCIBLE IN ASCII]
Effects of Levodopa on Pulmonary Function and UAO
All patients were responsive to levodopa, with a minimum 50% response to levodopa. The improvement in motor disability with levodopa was similar in patients with and without UAO, mainly through a not formally rated reduction of akinesia (improvement in walking, standing, sitting, speaking, mimic and bradykinesia). Among the patients with UAO, after levodopa therapy, three of five patients did not meet the four of six required criteria for defining UAO; this was the case in only one patient after placebo treatment. In patients with UAO, levodopa produced its effects on UAO criteria by means of a saw-tooth pattern improvement (two patients) and/or a decrease below the defined thresholds of the [FEV.sub.1]/PEF ratio (two patients), the [FEV.sub.1]/[FEV.sub.0.5] ratio (one patient), and the PIF (one patient). Figures 1, 2 show representative examples of the effects of levodopa on flow-volume loops in patients with UAO. Figure 1 shows a typical flow-volume loop with evidence of a positive effect of levodopa on saw-tooth pattern and inspiratory and expiratory flow rates. Figure 2 shows a significant improvement in saw-tooth pattern following levodopa, with only slight improvement in inspiratory flow rate. The effects of the medication on spirometry and flow-volume curves data are presented in Tables 2, 3. After levodopa, PEF increased by 0.85 L/s in patients with UAO and by 0.24 L/s in patients without UAO, while after placebo it increased by 0.03 L/s in patients with UAO and decreased by 0.16 L/s in patients without UAO (p = 0.02). Whereas in patients without UAO an increase of the [FEV.sub.1]/PEF and [FEV.sub.1]/[FEV.sub.0.5] ratios was observed after placebo and levodopa intake, these ratios decreased after levodopa and increased after placebo treatment in patients with UAO (Table 3). In these patients as a whole, however, there was not a complete resolution of the five spirometric UAO criteria, including the [FEV.sub.1]/PEF and [FEV.sub.1]/ [FEV.sub.0.5] ratios for which an individual improvement had been noted. The two-way ANOVA for repeated measurements identified that differences between the means could be attributed to the UAO status for the variations of the percent predicted PEF (p = 0.01) and [FEV.sub.1]/[FEV.sub.0.5] ratio (p = 0.03), and to a medication effect for the variations of PEF (absolute and percent predicted values of p = 0.02 and p = 0.01, respectively) and the [FEV.sub.1]/PEF (p = 0.01) and [FEV.sub.1]/[FEV.sub.0.5] (p = 0.005) ratios (Tables 2, 3). In addition, an interactive effect of both the UAO status and the medication was found to be significant on the variations of the percent predicted PEF (p = 0.04) and the [FEV.sub.1]/PEF (p = 0.03) and [FEV.sub.1]/[FEV.sub.0.5] (p = 0.002) ratios.
[Figures 1-2 ILLUSTRATION OMITTED]
DISCUSSION
Our data show that in PD patients, levodopa administration induces significant variations in PEF and UAO ratios ([FEV.sub.1]/PEF and [FEV.sub.1]/[FEV.sub.0.5]).
The involvement of the upper airways in the respiratory dysfunction found in patients with PD has been shown in previous reports.[4,16,17] Our patients were classified as having UAO if they fulfilled at least four of six UAO criteria on both baseline studies, and the thresholds for the UAO ratios were chosen according to several studies.[4,15-17] Patients with UAO had a lower PEF than patients without UAO. Since the decrease in PEF was not associated with a decrease in [FEV.sub.1] or forced expiratory flow, midexpiratory phase, lower airway obstruction is unlikely. Bogaard et al[17] have shown that a progression in the severity of PD was associated with a PEF decrease. These authors also suggested that PEF is the most affected variable in the pattern of flow-volume curve.[16] In addition, Izquierdo-Alonso et al[18] found lower PEFs in patients with severe PD as compared with moderate or mild PD. Indeed, Bateman et al[19] have postulated that low values of PEF in PD patients may reflect more severe muscle disorders. The mechanism of UAO is poorly understood. PD symptoms affect various group of muscles, including upper-airway muscles. A saw-tooth pattern in flow-volume curve may correspond to upper-airway muscle tremor.[20] Moreover, the muscle dysfunction is often asymmetrical, and the lack of coordination between the numerous pharyngeal and laryngeal muscles may lead to distortion of upper-airway geometry.
Motor disability in PD patients can be partly reversed with levodopa therapy, resulting in decreased tremor and muscle rigidity, coordination of muscles, and facilitation of movement. These effects should alleviate UAO. Indeed, we observed a significant reduction in UAO after levodopa intake. Obenour et al,[3] in an open study of 31 patients with PD, did not find any change in expiratory flow or lung volumes after levodopa treatment. Nakane et al[5] found that a significant improvement occurred in PEF, maximal voluntary ventilation, [FEV.sub.1], FVC, and total lung capacity in patients receiving levodopa. We also found an increased PEF and a trend toward an increased [FEV.sub.1] following levodopa in our patients, but no effects were noted on lung volumes. However, our study cannot help to distinguish whether the increase in PEF is related to a direct effect on UAO or is linked with the improvement of motor disability in PD patients. Reports concerning the effects of antiparkinsonian drugs on pulmonary function yielded inhomogenous results, since lung volumes and expiratory flows were found either increased[5-7] or unchanged.[3] We found that levodopa produced its effects on UAO criteria by means of a saw-tooth pattern improvement and/or a decrease below the defined thresholds of the PIF and the [FEV.sub.1]/PEF and [FEV.sub.1]/[FEV.sub.0.5] ratios. Of note, these last ratios have been found to increase in UAO associated with extrapyramidal disorders.[4,16,17] The other ratios that we studied (ie, [FEF.sub.50]/[FIF.sub.50] and PEF/[FEF.sub.50]) were not modified after levodopa administration. These discrepancies could be explained by several reasons: (1) UAO in PD patients does not result from a lesion but from a dysfunction of upper airways; (2) the variations of the caliber of upper airways may be different in expiration and inspiration; (3) these abnormalities may also depend on the motor status of the patient, demonstrating partial or complete benefit of levodopa.[1] Thus, the usual criteria proposed for the study of UAO that is caused by a lesion may not apply in this dysfunctional UAO.[20] The observed abnormality of PEF could result from the combination of UAO and lack of coordination of respiratory muscles.
Several limitations of our study must be taken into account. First, the magnitude of the changes that we observed in several spirometry and UAO variables may be attributable to test-to-test variations, even if they met statistical significance. Attentive care was taken to obtain full understanding and cooperation from the patients, and in all but one enrolled subject, at least three reproducible volume curves were obtained. Indeed, PEF measurements are subject to variability,[21] especially in patients in whom motor disability may preclude satisfactory pulmonary function testing.[6] However, no specific reproducibility criterion for PEF measurement has been advocated.[21] Finally, our study design (a placebo-controlled, double-blind, crossover trial of levodopa) strengthens our confidence in the significance of the variations that we observed. Second, we studied the effects of drugs on pulmonary function tests 45 to 60 min after oral intake of drug, which is the usual levodopa plasma concentration peak.[22] However, the optimal improvement in motor function may be delayed, especially in the six patients who were naive to levodopa. Furthermore, the 100-mg levodopa trial dose was standardized and may have been insufficient in four patients who used higher morning doses. This may explain why levodopa has alleviated but not totally reversed UAO in some patients. Finally, we chose to make it possible to study patients after 45 min, which may have been too early to begin testing in some patients, but allowed to reduce fatigue at the end of session.[6]
We conclude that in PD patients, levodopa administration induces significant variations in PEF and UAO ratios ([FEV.sub.1]/PEF and [FEV.sub.1]/[FEV.sub.0.5]). The fact that these effects are found independently from the UAO status is in favor of a beneficial effect of levodopa on pulmonary function. Future trials are therefore warranted to investigate the clinical relevance of our findings.
REFERENCES
[1] Brown LK. Respiratory dysfunction in Parkinson's disease. Clin Chest Med 1994; 15:715-727
[2] Neu HC, Connolly JJ, Schwerley FW, et al. Obstructive respiratory dysfunction in parkinsonian patients. Am Rev Respir Dis 1967; 93:33-47
[3] Obenour WH, Stevens PM, Cohen AA, et al. The causes of abnormal pulmonary function in Parkinson's disease. Am Rev Respir Dis 1972; 105:382-387
[4] Vincken WG, Gauthier SG, Dollfuss RE, et al. Involvement of upper-airway muscles in extrapyramidal disorders. N Engl J Med 1984; 311:438-442
[5] Nakane KK, Bass H, Tyler HR. Levodopa in Parkinson's disease. Arch Intern Med 1972; 130:346-348
[6] De Bruin PFC, De Bruin VMS, Lees A, et al. Effects of treatment on airway dynamics and respiratory muscle strength in Parkinson's disease. Am Rev Respir Dis 1993; 148:176-180
[7] Vincken WG, Darauay CM, Cosio MG. Reversibility of upper airway obstruction after levodopa therapy in Parkinson's disease. Chest 1989; 96:210-212
[8] Granerus AK, Jagenburg R, Nillsson NJ, et al. Respiratory disturbance during L-DOPA treatment of Parkinson's syndrome. Acta Med Scand 1974; 195:39-43
[9] Weiner WJ, Goetz CG, Nausieda PA, et al. Respiratory dyskinesias: extrapyramidal dysfunction and dyspnea. Ann Intern Med 1978; 327:327-331
[10] De Keyser J, Vincken W. L-dopa induced respiratory disturbance in Parkinson's disease suppressed by tiapride. Neurology 1985; 35:235-236
[11] Jankovic J, Nour F. Respiratory dyskinesia in Parkinson's disease [letter]. Neurology 1985; 36:303-304
[12] Zupnick HM, Brown LK, Miller A, et al. Respiratory dysfunction due to L-dopa therapy for parkinsonism: diagnosis using serial pulmonary function tests and respiratory inductive plethysmography. Am J Med 1990; 89:109-114
[13] Hughes AJ, Daniel SE, Kilford L, et al. Accuracy of clinical diagnosis of idiopathic Parkinson's disease: a clinico-pathologieal study of 100 cases. J Neurol Neurosurg Psychiatry 1992; 55:181-184
[14] Hoehn MM, Yahr MD. Parkinsonism onset, progression and mortality. Neurology 1967; 17:427-442
[15] Mellisant AC, Van Noord JA, Van de Woestijne KP, et Comparison of dynamic lung function indices during forced and quiet breathing in upper airway obstruction, asthma, and emphysema. Chest 1990; 98:77-83
[16] Hovestadt A, Bogaard JM, Meerwaldt JD, et al. Pulmonary function in Parkinson's disease. J Neurol Neurosurg Psychiatry 1989; 52:329-333
[17] Bogaard JM, Hovestadt A, Meerwaldt JD, et al. Maximal expiratory and inspiratory flow-volume curves in Parkinson's disease. Am Rev Respir Dis 1989; 139:610-614
[18] Izquierdo-Alonso JL, Jimenez-Jimenez FJ, Cabrera-Valdivia F, et al. Airway dysfunction in Parkinson's disease. Lung 1994; 172:47-55
[19] Bateman DN, Cooper RG, Gibson GJ, et al. Levodopa dosage and ventilatory function in Parkinson's disease. BMJ 1981; 283:190-191
[20] Vincken WG, Cosio MG. Flow oscillations on the flow-volume loop: clinical and physiological implications. Eur Respir J 1989; 2:543-549
[21] American Thoracic Society. Standardization of spirometry: 1994 update. Am J Respir Crit Care Med 1995; 152:1107-1136
[22] Gancher ST, Nutt JG, Woodward WR. Peripheral pharmacokinetics of levodopa in untreated, stable, and fluctuating parkinsonian patients. Neurology 1987; 37:940-944
(*) From Service de Pneumologie (Dr. Herer), Centre Medical de Forcilles, Ferolles-Attilly; Service de Pneumologie (Dr. Housset), Centre Hospitalier Intercommunal de Creteil, Creteil; and Federation de Neurologie (Dr. Arnulf), UPRES EA 2397 Hopital Pitie-Salpetriere, Paris France.
Supported in part by the association "Compliance."
Manuscript received October 1, 1999; revision accepted August 2, 2000.
Correspondence to: Bertrand Herer, MD, Centre Medical de Forcilles, F-77170 Ferolles-Attilly, France; e-mail: HBherer@aol.com
COPYRIGHT 2001 American College of Chest Physicians
COPYRIGHT 2001 Gale Group