CONTINUING EDUCATION
To earn CEUs, see test on page 20.
LEARNING OBJECTIVES
Upon completion of this article the reader will be able to:
1. Recognize genetic disorders that are currently approved for population-based carrier screening (PBCS) and why these have been chosen.
2. Recognize the carrier frequencies of common genetic disorders and the reduction of affected birth rates as a result of PBCS.
3. Recognize the populations that are at high risk for each genetic disorder.
4. Recognize the genetic mutations and mode of inheritance common for each disorder.
5. Recognize methods to reduce the rates of these disorders.
6. Recognize methods to identify each disorder and challenges associated with the testing methods.
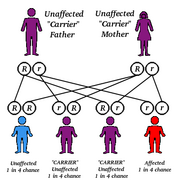
Population-based carrier screening (PBCS) for genetic diseases began in the United States with a pilot program for Hexoseaminidase A (HexA) deficiency, also known as Tay Sachs disease (TSD) in Ashkenazi Jewish (AJ) populations in Baltimore, MD, and Washington, DC, in 1970. TSD, an autosomal recessive (AR) disease, occurs with a frequency of 1:3600 births to AJ couples and has a carrier frequency of 1:30 in that population. This carrier frequency is 10 times that observed in non AJ populations. (1)
[ILLUSTRATION OMITTED]
Following widespread implementation, PBCS for TSD resulted in a >90% reduction in the births of affected children with TSD to AJ couples in the United States and Canada. (1) Another PBCS program, initiated by members of Dor Yeshorim, a group of Orthodox Jews, routinely screens adolescents in their community for TSD and several other prevalent AJ genetic diseases. Results are confidential, held in a centralized database, and are not disclosed to individuals. Rather, all marriages in this community are arranged, and couples are chosen to prevent the union of two carriers of the same genetic disease. This system has the advantage of avoiding any potential stigmatization of individuals, but is only practical in societies where all marriages are arranged.
Methods of TSD carrier detection
Initially, PBCS for TSD was performed exclusively by an enzyme assay for HexA. The HexA assay uses an artificial umbilliferyl N-acetylglucosamine substrate that creates a fluorescent product 4-methylumbelliferone when cleaved by HexA. (2) Unfortunately, a second enzyme, HexB, whose deficiency causes Sandhoff disease, also cleaves the same artificial substrate. Therefore, the HexA activity must be differentiated from the HexB activity. The fact that HexB is relatively resistant to heat inactivation with respect to HexA is exploited for this purpose. The assay is initially performed to obtain a total Hex activity (A+B), and then the assay is repeated after heat treatment of the clinical sample. The percentage of HexA activity is determined by the equation:
((Total Hex Activity-Heat Stable Hex Activity)/Total Hex Activity) X100.
Standard curves for percentages of HexA activity are generated for both noncarriers and carriers, and three standard deviations above the mean for carriers and three standard deviations below the mean for noncarriers are usually chosen as the cutoffs. The regions of overlap are called the "gray zone" or indeterminate level. For example, a theoretical range might be: noncarrier >55% HexA, inconclusive 40% to 55% HexA, carrier 20% to 40% HexA, and affected <10% HexA. In general, as ranges are set and as false-positive rates decrease, so do the percentages of patients whose results will be in the gray zone. In the scheme above, one might observe a specificity of 98%, with 10% of patients indeterminate. By increasing the indeterminate range to 35% to 60%, specificity may increase to >99%, but the percentage of patients with indeterminate results may increase to >20%. Generally, individuals whose results are indeterminate will continue to have gray-zone values upon repeated testing. DNA testing is appropriate for such individuals. (1)
HexA measurements can be performed on a variety of sample types including serum, leukocytes, platelets, amniocytes, chorionic villi, and cultured skin fibroblasts. For the purposes of PBCS, serum, leukocyte, and platelet screening are customarily offered. Serum testing is the least accurate in terms of specificity and sensitivity and suffers from interference by pregnancy and oral-contraceptive use. The sensitivity and specificity of the white cell and platelet assays vary among laboratories, as does the percentage of patients who will be "indeterminate."
Problems with PBCS for TSD
Soon after widespread PBCS for TSD began, some asymptomatic adults were identified who appeared to have complete HexA deficiency in the standard assay. Such individuals were described as having a "pseudodeficiency." Pseudodeficiency alleles have normal in vivo activity against naturally occurring substrates, but lack activity against the artificial substrate used in the in vitro assay. With the advent of DNA-based testing, we now understand that the molecular basis for AJ pseudodeficiency alleles is the missense mutation R247W. (2) We observed this mutation to have a frequency of 1:473 AJ individuals; approximately 1:16 AJ individuals who appear to be TSD carriers by HexA screening are not carriers. (3) Other pseudodeficiency alleles have been identified in French Canadians and in Sephardic Jews.
For this reason, all individuals who are apparent TSD carriers by HexA analysis require reflex DNA testing to establish if they carry a pseudodeficiency allele and are not TSD carriers. Individuals who are compound heterozygotes for a pseudodeficiency allele and a true TSD mutation would appear to be affected using HexA testing, thereby explaining the unaffected adults with HexA deficiency results. Unfortunately, amniocytes and chorionic villi with the same genotype would be inaccurately diagnosed as affected. Prior to the advent of generally available molecular testing in the early 1990s, it is possible that some unaffected fetuses may have been misdiagnosed and terminated due to this phenomenon. The important message from this experience is: Whenever large-scale screening programs are introduced, unforeseen complications will arise that will require continuous re-evaluation and refining of screening recommendations. Most recently, cystic fibrosis (CF) carrier screening has required one major modification. (4)
Molecular testing for TSD
In 1988, the HexA gene was cloned by Myerowitz and colleagues. (5) Shortly thereafter, it was demonstrated that approximately 98% of AJ carriers for TSD carry one of three predominant mutations: 1278insTATC, IVS12+1G>C, or G269S. Several studies have examined the efficacy of PBCS for TSD using either DNA testing alone, HexA testing with reflex to DNA testing for all positives, and combined DNA and HexA testing. Not surprisingly, the greatest sensitivity and specificity is observed with a combination of both DNA and HexA testing for all specimens. Unfortunately, most AJ individuals can have only one test paid for by insurers.
Two large studies have compared the sensitivity and specificity of the three-mutation and pseudo-mutation DNA test alone vs. the HexA test with DNA follow-up of positive results. Both studies concluded that DNA testing is the preferred method if a single test must be chosen. (6,7) In the largest published study, more than 103,000 HexA tests performed in several centers were pooled and compared with results of leukocyte HexA, platelet HexA, and DNA testing. The best enzyme-testing results were obtained when leukocyte assays were performed, and inconclusive tests were scored as positive. This resulted in a sensitivity of 99.08% and specificity of 99.01%. DNA testing alone performed with a sensitivity of 98.82% and specificity of 100%. (7) Platelet Hex A testing performs similarly to leukocyte testing and better than serum testing.
DNA testing for TSD can be performed using four single-plex assays for each patient or a single multiplex assay as performed in our laboratory. We use a technology known as single-base primer extension. A multiplex PCR is performed, amplifying all relevant sequences. Following PCR, four sequencing primers are added (one for each mutation). These primers end one base pair 5-prime to the mutation site. Then a mixture of all four dideoxynucleotides, each coupled to a different fluorescent dye, are added and primer extension performed. Each sequencing primer differs in length from the others. Following the primer extension reaction, the products are separated on a capillary DNA sequencer. The color at the length of the appropriate primer determines the nucleotide at that position. The genotype is determined automatically, using commercially available software. (3)
Options for carrier couples
Carrier couples may elect to have preimplantation genetic analysis in order to assure an unaffected pregnancy, or prenatal diagnosis using chorionic villi sampling (CVS) in the first trimester, or amniocentesis in the second trimester. If DNA testing has identified mutations in both parents, DNA testing of the fetus is the preferred method for prenatal diagnosis. If one or both parental mutations have not been defined, a HexA assay can be performed on cultured CVS or amniocytes.
Carrier screening for additional diseases: official recommendations
Generalized TSD screening was implemented without any official recommendations from professional organizations. In 1998, the American College of Medical Genetics (ACMG) recommended adding Canavan disease (CD) testing by DNA analysis to TSD screening for AJ individuals. Cystic fibrosis was added to the list in 2001. DNA testing for three common CD mutations (E285A, A305E, and Y213X) will identify 98% of AJ CD carriers. Our laboratory utilizes the same single-base primer extension for CD that is used for TSD. Currently, carrier screening for several other diseases of high prevalence in the AJ population are available, but no official recommendations have been made regarding the appropriateness of population screening. In addition to CF, CD, and TSD, our laboratory currently offers DNA-based testing for Gaucher disease, Bloom syndrome, familial dysautonomia, Fanconi anemia complement group C, Niemann-Pick disease, Mucolipidosis IV, and atypical congenital adrenal hyperplasia. (3)
PBCS in other populations
In the United States, population screening for hemoglobinopathies has been offered for decades among individuals of sub-Saharan African descent (sickle cell anemia [SCA]), individuals of Southeast Asian descent (alpha thalassemia), and people of Mediterranean descent (beta thalassemia) with varying levels of success. Carrier testing for SCA may be performed by several techniques of hemoglobin analysis, including hemoglobin electrophoresis, isoelectric focusing, and high-performance liquid chromatography, or HPLC. All of these techniques are able to reliably identify hemoglobin S. Perhaps the most striking examples of successful PBCS programs are these of the island nations of Sardinia and Cyprus, where the incidence of thalassemia has been reduced >99%. (8)
Carrier testing for beta thalassemia is more complicated and the interpretation more nuanced. In general, carriers of beta thalassemia will have a mild hypochromic microcytic anemia (reduced mean corpuscular volume and reduced mean corpuscular hemoglobin concentration) and increased hemoglobin A2 on a hemoglobin chain analysis. A small percentage of carriers of carriers of beta thalassemia, however, can have normal hemoglobin A2. Gene sequencing of the entire coding region of the globin gene is capable of identifying most carriers of beta thalassemia and can resolve ambiguities of hemoglobin analysis. Some forms of beta thalassemia due to gene deletions will not be detected by DNA sequence analysis. Since compound heterozygotes for hemoglobin S and beta thalassemia will have symptoms of SCA, it is essential to investigate both the sickle cell status and the thalassemia status of spouses of known heterozygotes for hemoglobin S.
The evaluation of patients for alpha thalassemia carrier status is complicated by the fact that there are two different functional alpha globin genes--alpha 1 and alpha 2. Therefore, wild type individuals have four copies of the alpha globin gene in each cell--two copies each of alpha 1 and alpha 2. Most cases of alpha thalassemia are due to gene deletions. Table 1 is a listing of commonly observed alpha thalassemia genotypes and their functional significance.
Notably, there are two genetic forms of alpha thalassemia minor (severe). Both alpha globin genes can be deleted from a single chromosome, or one alpha globin gene can be deleted from each chromosome. Although both these genotypes result in the presence of two alpha globin genes, they have very different implications for inheritance risk. Two parents who carry both genes deleted on the same chromosome are at a 25% risk of having a child with alpha thalassemia major, a 50% risk of having a child with alpha thalassemia minor (severe), and a 25% chance of having a normal child. All off-spring of parents who are homozygous for chromosomes with one deleted alpha globin gene will have alpha thalassemia minor (severe). It is, therefore, imperative to distinguish the genetic structure present in patients with alpha thalassemia minor (severe).
Tests based on the polymerase chain reaction (PCR) are capable of identifying the most common variants of such chromosomes. In the rare instances when this testing is uninformative, other types of DNA testing may be required, such as multiple Southern Blots. (8) The most common alpha thalassemia mutation, occurring in more than 1% of individuals of sub-Saharan ancestry, is known as alpha 3.7 and is a deletion of one of the two genes on a single chromosome. The mutations deleting both globin genes on a single chromosome are more common in Southeast Asian populations.
Once the parental mutations have been identified, prenatal and preimplantation diagnoses are available for at-risk couples for these hemoglobinopathies. If the mutations cannot be identified, prenatal diagnosis may still be accomplished by obtaining a fetal blood sample in a procedure known as percutaneous umbilical cord blood sampling, or PUBS. Fetal blood can then be subjected to chain analysis for the purposes of prenatal diagnosis.
PBCS for cystic fibrosis
In October 2001, the American College of Obstetrics and Gynecology (ACOG), in collaboration with the ACMG, published recommendations for population-based carrier screening for CF in Caucasian and AJ couples. (9) The initial ACOG recommendations involved testing Caucasian couples (including AJ) for 25 of the >1,000 CF mutations. The ACOG statement focused attention on PBCS because, unlike prior screening programs that were limited to minority populations, the CF recommendation impacts the majority of pregnancies in the United States. It is important to keep in mind that PBCS is not a new phenomenon: rather, it is the scale of testing that is new.
The recommendation that 25 mutations and six polymorphisms be tested for each patient required the development of reagents capable of performing such testing. There are now many analyte-specific reagent (ASR) systems available: line-probe reverse-dot blot, allele-specific PCR with gel electrophoresis, Luminex-based, Cleavase-based (Invader), mass spectrometry, and other laboratory-developed tests.
The most glaring error in the ACOG recommendation was the inclusion of I148T, one of the 25 mutations initially recommended for testing; I148T was found to be a polymorphism and will be removed from the panel. (4)
Discussion and future directions
Without exception, all diseases for which PBCS has been recommended or is generally accepted are autosomal recessive diseases with carrier frequencies in their target populations of >1:50. Table 2 is a list of these diseases with the carrier frequencies, disease frequencies, symptoms and method of PBCS listed.
In our laboratory, disorders requiring three or more mutations to be analyzed are performed using the single-base primer extension described above. Single-mutation analysis is performed using the Promega ReadIT luciferase-based system. An oligonucleotide specific for the normal allele is added to one well, and a primer complementary to the mutant allele is added to the second well. The mismatched base is placed at the 5-prime penultimate base of the primer. The ReadITreaction is performed by adding excess pyrophosphate to drive the DNA polymerase backward. If there is a perfect match, an ATP (adenosine triphosphate) is generated; if there is a mismatch, an AMP (adenosine monophosphate) is produced. A luciferase reaction produces light in the presence of ATP but not AMP. Genotype assignment is made by examining the ratio of the lumens in the wild type vs. the mutant probe well in an automated luminometer. This process is automated using software provided by the manufacturer. (3) Several other platforms are easily capable of performing single-base analysis.
Mucolipidosis IV requires two mutations to be analyzed. Our laboratory exploited the phenomenon that one of the mutations is a 6.4 kb deletion to enable us to design a realtime PCR assay using Taqman chemistry capable of detecting both mutations simultaneously. When duplex PCR is performed, one reaction amplifies the fragment containing the SNP IVS3-2 A>G, and the second amplifies a junctional fragment only present when the deletion has occurred. PCR is performed in an ABI 7900 instrument capable of simultaneously detecting four dyes. The first dye is used as an internal standard, the second dye is linked to the wild type SNP (single nucleotide polymorphism), the third dye is linked to the mutant SNP, and the fourth dye is linked to the junctional fragment.
Prenatal and preimplantation diagnoses are available using amniocentesis or CVS for any couples when both parents are found to be carriers of the same AR disorder.
A new disease for PBCS: Fragile X syndrome
The decision whether or not to recommend PBCS has historically depended on several considerations: the frequency of the disease (and therefore carriers), the ability to and cost of detecting carriers, the severity of the disease, the cost of treating the disease, identifying a target population, and other demographic and socioeconomic factors.
Given all these variables, the next obvious candidate for PBCS must be Fragile X Syndrome (FraX), the most frequently occurring inherited form of mental retardation (MR) worldwide. Affected males with FraX suffer from moderate to severe MR with profound speech delays, socialization problems, and attention deficit disorder (ADD). One quarter to 35% of FraX males fulfill the diagnostic criteria for autism. Heterozygous females often suffer from ADD, learning disabilities, and social disabilities. (10)
Fragile X syndrome is seen in all races and ethnic groups with similar frequencies. Unlike the other diseases listed in Table 1 that are all AR diseases, FraX is X-linked. In AR diseases, both parents must be carriers in order to be at risk for having an affected child. Therefore, once a carrier female is identified, her spouse must be tested, and only if her spouse is also a carrier is their fetus at risk for the disease. In contrast, identifying a female carrier immediately puts her pregnancy at risk for having an affected fetus. This simplifies the logistics of screening and allows identification of many more at-risk couples when extended family members of identified carriers are screened.
Fourteen international studies, including populations in Canada (three studies), the United States (seven studies), Israel (two studies), Finland (one study), and Greece and Cyprus (one study), have demonstrated carrier frequencies of between 1:250 to 1:468 for FraX (for review, see reference 10). Using the lowest estimate, in a PBCS program for FraX, one in every 468 pregnancies will be identified at risk for FraX. This is greater than the 1:784 Caucasian couples predicted to be at risk for having a fetus with CF and the 1:900 AJ couples at risk for having a fetus with TSD. A recent study reported that parents of children diagnosed with FraX were overwhelmingly in favor of both newborn screening and carrier detection. (11)
The reasons preventing the recommendation of PBCS for FraX are most likely technical and financial. Carrier screening for FraX requires both a PCR-based assay and a Southern Blot due to the complexities of the mutations, triplet-repeat expansions. When the ACOG and ACMG made their recommendation for CF screening with 25 mutations, there were no available ASR reagents or FDA-approved test kits capable of analyzing this recommended panel of mutations. Laboratories and IVD companies developed and produced tests and reagents capable of performing these tests.
PBCS provides a way for couples to avoid the birth of children affected with genetic diseases prior to birth. As the fruits of the Human Genome Project become even more bountiful, an increasing number of genetic diseases will become appropriate for PBCS. This will inevitably put a strain on genetic-counseling resources. As multiplexing technology enables us to assay increasing numbers of mutations in a single individual at a reasonable cost, we may experience increasing pressure from patients and physicians to offer more tests to more individuals. Bead-based and microarray-based platforms are already capable of such multiplex analysis. This raises new ethical dilemmas; as more diseases are tested for, more individuals will be discovered to be carriers of at least one disease. In our experience with the eight-disease AJ panel, one in seven individuals was a carrier of at least one disorder. (3) It is estimated that each individual carries between six and 10 deleterious recessive genes. Do we need or want to know about each one? The answer may be "yes," but we need to ask the question.
CE test on POPULATION-BASED CARRIER SCREENING AND PRENATAL DIAGNOSIS
MLO and Northern Illinois University (NIU), DeKalb, IL, are co-sponsors in offering continuing education units (CEUs) for this issue's article on POPULATION-BASED CARRIER SCREENING AND PRENATAL DIAGNOSIS. CEUs or contact hours are granted by the College of Health and Human Sciences at NIU, which has been approved as a provider of continuing education programs in the clinical laboratory sciences by the ASCLS P.A.C.E.[R] program (Provider No. 0001) and by the American Medical Technologists Institute for Education (Provider No. 121019; Registry No. 0061). Approval as a provider of continuing education programs has been granted by the state of Florida (Provider No. JP0000496), and for licensed clinical laboratory scientists and personnel in the state of California (Provider No. 351). Continuing education credits awarded for successful completion of this test are acceptable for the ASCP Board of Registry Continuing Competence Recognition Program. After reading the article on page 12, answer the following test questions and send your completed test form to NIU along with the nominal fee of $20. Readers who pass the test successfully (scoring 70% or higher) will receive a certificate for 1 contact hour of P.A.C.E.[R] credit. Participants should allow four to six weeks for receipt of certificates.
The fee for each continuing education test will be $20.
All feature articles published in MLO are peer-reviewed.
Objectives and CE questions prepared by Gail S. Williams, PhD, MT(ASCP)SBB, CLS(NCA), Northern Illinois University, College of Health and Human Sciences, Clinical Laboratory Sciences Program, DeKalb, IL.
1. Tay Sachs disease is also known as
a. AJ disease.
b. Hexoseaminidase A deficiency.
c. Hexoseaminidase B deficiency.
d. Canavan disease.
2. Which of the following has the highest carrier frequency?
a. Beta thalassemia.
b. Sickle cell anemia.
c. Tay Sachs disease.
d. Cystic fibrosis.
3. One method of reducing the incidence of several disorders found in the Ashkanazi Jewish community is to test teenagers and
a. abort all children from carriers.
b. arrange marriages so that no child with these disorders is likely to be born.
c. test their fetuses.
d. create special homes for carriers.
4. Tay Sachs disease is only found in the Ashkanazi Jewish population.
a. True
b. False
5. The enzyme assay for Tay Sachs disease is performed heated and unheated because
a. two different enzymes cleave the substrate, and one is enhanced by heating.
b. relative enzyme activity is being evaluated.
c. two different enzymes cleave the same substrate, and one is heat labile.
d. Sandhoff disease is caused by the same deficiency.
6. To confirm indeterminant results by enzyme assay, what is done?
a. DNA testing to confirm specific mutation.
b. Repeat testing.
c. Counseling.
d. Testing by a different EIA.
7. Which samples are the least reliable for Tay Sachs disease?
a. Leukocytes.
b. Serum.
c. Skin fibroblasts.
d. Chorionic villi.
8. A pseudodeficiency of the enzyme causing Tay Sachs disease is due to the following mutation (polymorphism):
a. Nonsense R247W.
b. Deleted R247W.
c. Missense R247W.
d. G269S.
9. When comparing the sensitivity and specificity of enzyme assays and DNA testing for Tay Sachs disease
a. the enzyme assay is most sensitive and specific.
b. the DNA assay is most sensitive and specific.
c. the enzyme assay is more specific but less sensitive.
d. the enzyme assay is more sensitive but less specific.
10. For couples who are carriers, what options are available for prenatal testing?
a. Preimplantation analysis.
b. Chorionic villi sampling during first trimester.
c. Amniocentesis during second trimester.
d. All of the above.
11. Which of the following is a mutation seen in Canavan disease?
a. 1148T.
b. E285A.
c. G269S.
d. R247W.
12. Alpha thalassemia is seen primarily in which population?
a. Mediterranean
b. African-American.
c. Caucasian.
d. Asian.
13. What is required to resolve ambiguous test results for beta thalassemia?
a. Hemoglobin electrophoresis.
b. Gene sequencing.
c. Isoelectric focusing.
d. Increased hemoglobin A2 levels.
14. Alpha thalassemia diagnosis is complicated because
a. there are variations of combinations of the two alpha globin gene deletions.
b. thalassemia major results in macrocytes rather than microcytes.
c. it is a autosomal recessive disease.
d. All of the above.
15. The deletion of two alpha thalassemia genes on the same chromosome is seen primarily in which population?
a. African-American.
b. Ashkanazi Jew.
c. Mediterranean.
d. Asian.
16. How many mutations have been identified for cystic fibrosis?
a. 25.
b. >100.
c. >500.
d. >1,000.
17. Which of the following is not an autosomal recessive trait?
a. Cystic fibrosis.
b. Fragile X syndrome.
c. Tay Sachs disease.
d. Gaucher disease.
18. What is considered when deciding to do population-based carrier testing?
a. Frequency of disease.
b. Ability and cost of detecting carriers.
c. Severity of disease.
d. All of the above.
19. Fragile X is the most common cause of inherited mental retardation worldwide.
a. True.
b. False.
20. Fragile X carrier detection and confirmation may be achieved by
a. PCR alone.
b. enzyme assays alone.
c. PCR to screen and Southern Blot to confirm.
d. analysis of the physical characteristics of the patient.
TEST ANSWER FORM
DIAGNOSTIC ASSAYS FOR HIV-1 INFECTION--July 2004
(This form may be photocopied; it is no longer valid for CEUs after Dec. 31, 2005.)
Note: Please use MLO's online version of the July 2004 CE test to send answers to Northern Illinois University. In Questions #16 and #19 in the print version of that test, selection circles for answers C and D on both were missing. Not using the online version of the July 2004 test may delay your test results. MLO apologizes for any inconvenience this error may have caused its readers.
[GRAPHIC OMITTED]
References
1. Kayback M, Lim-Steele J, Dabholkar D, Brown D, Levy N, Zeiger K. Tay-Sachs Disease--Carrier screening, prenatal diagnosis, and the molecular era. JAMA 1993; 270:2307-2315.
2. Gravel RA, Kaback M, Proia RL, Sandhoff K, Suzuki K, Suzuki K. The GM2 Gangliosidosis. In: Scriver CR, Deaudet AL, Sly W, Valle D, Childs B, Kinzler KW, Vogelstein B, eds. The metabolic and molecular basis of inherited disease. 8th ed. New York, NY: McGraw Hill; 2001:3827-3877.
3. Strom CM, Crossley B, Redman JB, Quan F, Buller A, McGinniss M, Sun W. Molecular screening for diseases frequent in Ashkenazi Jews: Lessons learned from more than 100,000 tests performed in a commercial laboratory. Genet Med. 2004;6:145-152.
4. Watson MS, Desnick RJ, Grody W, Mennuti MT, Popovich BW, Richards S. Cystic fibrosis carrier screening: Issues in implementation. Genet Med. 2002;4:407-409.
5. Myerowitz R, Costigan FC. The major defect in Ashkenazi Jews with Tay Sachs Disease is an insertion in the gene for the alpha-chain of beta hexosaminidase. J Biol Chem. 1988;263:185670-185679.
6. Triggs-Raine BL, Feigenbaum ASJ, Natowicz M, Skomorowski MA, Schuster SM, Clarke JTR, Mahuran DJ, Kolodny EH, Gravel RA. Screening for carriers of Tay-Sachs disease among Ashkenazi Jews; A comparison of DNA-based and enzyme based tests. New Eng J Med. 1990:323;6-12.
7. Bach G, Tomczak J, Risch N, Ekstein J. Tay-Sachs screening in the Jewish Ashkenazi population: DNA testing is the preferred procedure. Am J of Medical Genetics. 2001;99:70-77.
8. Weatherall DJ, Clegg JB, Higgs DR, Wood WG. The Hemoglobinopathies, In: Scriver CR, Deaudet AL, Sly W, Valle D, Childs B, Kinzler KW, Vogelstein B, eds. The metabolic and molecular basis of inherited disease. 8th ed. New York, NY: McGraw Hill; 2001:4571-4636.
9. American College of Obstetrics and Gynecology and American College of Medical Genetics. Preconception and prenatal carrier screening for cystic fibrosis. Clinical and laboratory guidelines. October 2001. Washington, DC. ACOG.
10. Crawford DC, Acuna, JM, Sherman S. FMR1 and the fragile X syndrome; Human genome epidemiology review. Genet Med. 2001;3:360-371.
11. Skinner D, Sparkman KL, Baily DB Jr. Screening for Fragile X Syndrome: Parent attitudes and perspectives. Genet Med. 2003;5:378-384.
By Charles M. Strom, MD, PhD
Charles M. Strom, MD, PhD, is the medical director, Genetic Testing Center, Quest Diagnostics, Nichols Institute in San Juan Capistrano, CA.
COPYRIGHT 2004 Nelson Publishing
COPYRIGHT 2004 Gale Group