Purpose: We investigated whether oral tetracyclines could produce an efficient and safe pleurodesis as does parenteral doxycycline, which is currently unavailable in many countries.
Methods: Parenteral doxycycline (10 mg/kg), oral tetracycline (35 mg/kg), or doxycycline (10 mg/kg) was injected intrapleurally through a right chest tube in rabbits. The oral forms were dissolved in saline solution and passed through a sterile membrane filter. When daily aspirated pleural fluid was < 5 mL/24 h, the chest tube was removed. Fluid WBC, lactate dehydrogenase (LDH), and protein levels were measured 24 h after the injection. After the death of the animals on day 14, pleurodesis was graded from 1 (none) to 8 (> 50% symphysis) by two observers blinded to treatment groups.
Results: The right pleurodesis score of the combined oral groups (median, 7.0; interquartile range [IQR], 4.0; n = 26) did not differ significantly (p = 0.349) from that of the parenteral group (median, 7.5; IQR, 6.0; n = 10). Oral tetracycline (capsule or tablet, n = 6 in each group) and doxycycline (capsule or tablet, n = 7 in each group) were as effective as parenteral doxycycline in producing pleurodesis: tetracycline capsule (median, 7.50; IQR, 6.00); tetracycline tablet (median, 6.50; IQR, 6.00); doxycyeline capsule (median, 4.00; IQR, 1.00); doxycycline tablet (median, 8.00; IQR, 5.00), and parenteral doxycycline (median, 7.50; IQR, 6.00) [p = 0.235]. The left pleurodesis scores were 1.00 in all 36 rabbits. Fluid total volume, WBC, LDH, and protein levels were comparable between each oral and parenteral group, excluding WBCs in the tetracycline tablet group (p = 0.047). The complications were nonfatal (right hemothorax: tetracycline capsule [n = 3]/tetracycline tablet [n = 2], doxycycline tablet [n = 2], parenteral doxycycline [n = 2]; left hemothorax: tetracycline capsule [n = 1]; ascites: parenteral doxycycline (n = 1]). There was no growth on all filtrate cultures. Oral forms cost less than parenteral doxycycline (< $1 vs $4.72 per rabbit). Filtering costs were $1.12 per rabbit.
Conclusion: Oral tetracycline or doxycycline is as effective and safe as parenteral doxycycline in producing pleurodesis in rabbits; thus, they may also be used in humans.
Key words: capsule; doxycycline; pleurodesis; tablet; tetracycline
Abbreviations: IQR = intraquatile range; LDH = lactate dehydrogenase; MMP = matrix degrading metalloproteinase
**********
Pleurodesis is the obliteration of the pleural space by fusion of the visceral and parietal pleurae with fibrous tissue Recurrent and symptomatic pleural effusions and pneumothoraces are indications for pleurodesis. Most of the agents used for pleurodesis injure the pleura and cause an inflammatory reaction together with a pleural effusion. Subsequently, the local activation of the coagulation system and the production of fibrogenic eytokines such as transforming growth factor [beta] lead to the production of collagen that can result in a pleurodesis. (1)
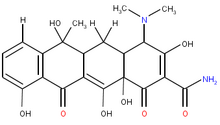
Currently talc, (2) tetracycline derivatives, (3-5) and bleomycin (5,6) are the most frequently used sclerosing agents. A survey study by Lee et al (7) questioning pleurodesis practice for malignant pleural effusions in five English-speaking countries reported that talc was the most commonly used agent, followed by tetracycline derivatives and bleomycin. Bleomycin is more expensive (1,6) and less efficient than tetracycline derivatives (1) or talc. (1,2) The intrapleural injection of talc can cause ARDS and death. (7,8) Although tetracycline (35 mg/kg) was the most effective compound when original animal experiments on producing pleurodesis in rabbits were performed, (9,10) the parenteral form of tetracycline is currently not available in most countries. Subsequently, parenteral doxycydine was used for pleurodesis at an approximate dose of 10 mg/kg and was shown to have comparable effectiveness with tetracycline at 35 mg/kg. (3,5,11,12) However, parenteral doxycycline is also not available currently in many parts of the world. In contrast, tetracycline and doxycycline capsules and tablets are available worldwide but (to our knowledge) have never been tested for their efficacy and safety in producing pleurodesis. We hypothesized that they can produce pleurodesis as efficaciously, safely, and cost effectively as their parenteral forms.
MATERIALS AND METHODS
The study protocol was approved by the Vanderbilt University Institutional Animal Care and Use Committee. The methods used were similar to those described in our previous studies. (13,14)
Chest Tube Insertion
New Zealand white rabbits weighing 2.5 to 3 kg were anesthetized with an IM injection of 35 mg/kg of ketamine hydrochloride (Fort Dodge Animal Health Laboratories; Fort Dodge, IA) and 5 mg/kg of xylazine hydrochloride (Fermenta; Kansas City, MO). The chest was shaved, and the skin was sterilized with 10% povidone iodine (Baxter; Deerfield, IL). The rabbit was placed in the left lateral decubitus position, and a skin incision < 3 cm was made midway between the tip of the right scapula and the sternum, approximately 2 cm above the costal margin. A chest tube (silicone tube, 0.062-inch internal diameter and 0.125-inch outer diameter; Braintree Scientific; Braintree, MA) with three extra openings near the distal end to enhance drainage was then inserted by blunt dissection into the right pleural cavity and was secured at the muscle layers with purse-string sutures. The proximal end of the chest tube was tunneled underneath the skin and drawn out through the skin posteriorly and superiorly between the two scapulae. A three-way stopcock (Medex; Dublin, OH) was attached to the end of the chest tube via an adapter through which pleural air was evacuated from the pleural space. The three-way stopcock was then removed from the chest tube, and the exterior end of the chest tube was sealed with a one-way valve with a cap (Medex) via the adapter and was sutured to the skin. Any pleural fluid or air that accumulated could be aspirated through the one-way valve. A local anesthetic was applied into the incision area before it was sutured since the rabbits were not given full general anesthesia. The left pleural cavity was used for control.
Intrapleural Injections
Thirty-six rabbits were allocated into tetracycline capsule (n = 6), tetracycline tablet (n = 6), doxycycline capsule (n = 7), doxycycline tablet (n = 7), and parenteral doxycycline (n = 10) groups. Oral forms of tetracycline (35 mg/kg) or doxycycline (10 mg/kg), or parenteral doxycycline (10 mg/kg) dissolved in 2 mL 0.9% sodium chloride were injected intrapleurally immediately after chest tube placement. Injection of tetracycline or doxycycline was followed by an injection of 1 mL of 0.9% sodium chloride solution to clear the dead space of the chest tube.
The tetracycline and doxycycline capsule contents and all of the tetracycline and doxycycline tablets were previously dissolved in 0.9% normal saline solution using sterile methods. Subsequently, the solution was filtered through a 0.2-[micro]m sterile and nonpyrogenic polyethersulfone membrane (Puradise 25 AS, 25 mm in diameter; Whatman; Clifton, NJ) to remove infectious materials and other particulate matter. The sterility of the filtered solutions was assessed by bacterial culture. The total amount of inactive ingredients in each tablet or capsule was determined by weighing the tablet or the contents of the capsule and then subtracting the stated amount of the tetracycline derivative.
Measurements for Pleural Fluid and Follow-up
After the intrapleural injection, the chest tube was aspirated at 24-h intervals for any pleural fluid that had accumulated. The volumes of the aspirated fluids were recorded. The levels of protein, lactate dehydrogenase (LDH), and WBC were measured in the fluid collected 24 h after the injection. The protein and LDH levels were determined with an automated analyzer (Johnson & Johnson; Rochester, MY). The lower and upper limits of normal serum LDH are 120 IU/L and 200 IU/L, respectively, by this method. The total WBC count was measured using an automated counter (Coulter Electronics; Luton, UK) that was calibrated daily. The first reading was ignored, and the mean of the next three readings was recorded.
Gentamicin (2 mg/kg) was administered IM every 24 h until the removal of the chest tubes. The chest tube was removed under light sedation when the pleural fluid drainage was < 5 mL over the preceding 24 h.
Grading for Pleurodesis
Rabbits were killed on day 14 by carbon dioxide euthanasia following sedation. The thorax was removed en bloc. The lungs were expanded by the injection of 50 mL of 10% neutral-buffered formalin into the exposed trachea. The trachea was then ligated, and the entire thorax was submerged into 10% neutral-buffered formalin solution for at least 48 h. To assess the pleurodesis, the pleural cavity was carefully exposed as previously described. (13,14) Using a semiquantitative scheme, a consensus grading on the degree of macroscopic pleurodesis was reached by two investigators (K.B.L. and R.W.L.) blinded to the treatment group. The degree of pleurodesis was graded using the following scale: 1 = no adhesions between the visceral and parietal pleura; 2 = rare adhesions between the visceral and parietal pleura with no symphysis; 3 = a few scattered adhesions between the visceral and parietal pleura with no symphysis; 4 = many adhesions between the visceral and parietal pleura with no symphysis; 5 = many adhesions between the visceral and parietal pleura with symphysis involving < 5% of the hemithorax; 6 = many adhesions between the visceral and parietal pleura with symphysis involving 5 to 25% of the hemithorax; 7 = many adhesions between the visceral and parietal pleura with symphysis involving 25 to 50% of the hemithorax; and 8 = many adhesions between the visceral and parietal pleura with symphysis involving > 50% of the hemithorax. Adhesions were defined as fibrous connections between the visceral and parietal pleura. Symphysis was present if the visceral and parietal pleura were difficult to separate as a result of adhesions.
Statistical Analysis
The data were expressed as mean [+ or -] SD when they were normally distributed, and as median (intraquartile range [IQR]) when they were not normally distributed. One-way analysis of variance was used to compare the means, and the Kruskal-Wallis test was used to compare the medians between the groups. The correlations were assessed by the Pearson product moment correlation test. All data were analyzed with statistical software (Sigma Stat; SPSS; Chicago, IL). A p value < 0.05 was considered significant.
RESULTS
The intrapleural injection of each of the five preparations elicited an exudative pleural effusion (Table 1). Thirty of the effusions (83%) were bloody appearing.
The volumes of aspirated pleural fluids at 24 h, 48 h, and 72 h after pleural instillation and the total volumes were comparable between each group receiving an oral form and receiving parenteral doxycycline (p > 0.05). These volumes tended to be less in the combined oral group (n = 26) than in the parenteral doxycycline group (n = 10), but only the difference in the 24-h pleural fluid volume achieved statistical significance (p = 0.024). When the four oral groups were compared regarding all parameters, the only significant difference was that the 72-h pleural fluid volume was significantly higher in the tetracycline capsule group than in the doxycycline capsule group (p = 0.032) [Table 2].
There were no significant differences in the pleural fluid LDH or protein levels between the groups or when the four groups that received the oral preparations were combined and compared with the group that received the parenteral doxycycline. The pleural fluid WBC level was significantly lower in the group that received parenteral doxycycline than in the group that received tetracycline tablet (p = 0.047), and the combined oral group (p = 0.036) [Table 1]. However, pleural fluid WBC levels of the parenteral group (median, 2,564/[micro]L; IQR, 1,784/[micro]L; n = 10) and the combined oral group excluding the tetracycline tablet group (median, 9,077/[micro]L; IQR, 2,722/[micro]L; n = 20) were comparable (p = 0.144).
Each of the oral forms of tetracycline or doxycycline was as efficacious as parenteral doxycycline in producing pleurodesis in rabbits (p = 0.235) [Fig 1]. Furthermore, the right pleurodesis score of the four oral groups combined (median, 7.0; IQR, 4.0; n = 26) did not differ significantly (p = 0.349) from that of the parenteral doxycycline group (median, 7.5; IQR, 6.0; n = 10). The combined oral group excluding the tetracycline tablet group (median, 7.0; IQR, 3.0; n = 20) was also comparable (p = 0.415) to the parenteral group regarding the right pleurodesis score. The left pleural score in each rabbit in all five groups was 1.
A significantly positive correlation existed between the right pleurodesis score and total, 24-h, 48-h, or 72-h pleural fluid volume, and the fluid protein level. Thus, efficiency of pleurodesis tended to be better in the presence of larger fluid volume or higher fluid protein level. However, no significant correlation was found for fluid WBC or LDH levels and the pleurodesis scores (Table 3).
All 36 rabbits survived without any major distress until death. Right hemothoraces occupying 20 to 90% of the hemithorax were observed at autopsy in three rabbits in the tetracycline capsule group, and in two rabbits in each of the tetracycline tablet, doxycycline tablet, and parenteral doxycycline groups, while one 5% left hemothorax was observed in the tetracycline capsule group. This left hemothorax accompanied one of the above-mentioned right hemothoraces. In the rabbits with hemothoraces, the pleurodesis scores ranged between 6 and 8. One rabbit from the parenteral doxycycline group was noted to have moderate ascites at death but no hemothorax. This rabbit had a pleurodesis score of 6.
There was no growth on microbiological culture of the filtered oral tetracycline or doxycycline solutions. When a 100-mg doxycycline capsule was dissolved in 10 mL of saline solution and then filtered, the prefiltration and postfiltration concentrations of the doxycycline capsule solution did not differ significantly (10 mg/mL vs 11.8 mg/mL, respectively).
Based on the per-tablet costs of a tetracycline 250-mg capsule ($0.25) or tablet ($0.23), doxycycline 100-mg capsule ($1.44) or tablet ($3.16), and per vial cost of doxycycline 100 mg injectable ($15.73), the per -rabbit cost of tetracycline capsule ($0.10) and tablet ($0.10), and doxycycline capsule ($0.48) and tablet ($0.95) were lower than that of parenteral doxycycline ($4.72). The cost of each filter used in filtering the solutions was $1.12.
DISCUSSION
Our study demonstrates that oral forms of tetracycline or doxycycline are as effective and safe as parenteral doxycycline in producing pleurodesis in rabbits. The oral forms are less expensive and more widely available than the parenteral forms. It is likely that these results can be extrapolated to human subjects since parenteral tetracycline (4,6,15) and doxycycline (3,5,15) have been extensively studied and shown to be effective and safe in producing pleurodesis in patients.
The ingredients other than tetracycline or doxycycline in tablets or capsules are in small amounts. There is 10-mg filling in a 250-mg tetracycline tablet, 100-mg filling in a 500-mg tetracycline capsule, 25- to 50-mg filling in a 100-mg doxycycline tablet, and 100- to 350-mg filling in a 100-mg doxycycline capsule. Depending on the form (capsule/tablet) and also the product from different producers, these inactive ingredients may include anhydrous lactose, colloidal silicone dioxide, magnesium stearate, methylcellulose, microcrystalline cellulose, polyethylene glycol, sodium starch glycolate, stearic acid, titanium dioxide, FD&C Yellow No. 6, FD&C Red No. 40, hydroxypropyl methylcellulose, propylene glycol, povidone, talc, triacetin, aluminum lake, polyparaben, methylparaben sodium citrate, potassium sorbate, xanthan gum, progyl paraben, and polyethylene glycol. (16)
One might question whether one of these other substances might be responsible for the pleurodesis. The only two substances in the above list that have been shown to produce pleurodesis are talc (2,8,11) and povidone. (17) However, in the current study, we did not use any product containing talc, and povidone is present only in the tetracycline tablets we used. Povidone is not likely to have a role in the pleurodesis score of the tetracycline tablet group considering the minute amount administered (< 10 mg per 250-mg tetracycline tablet that equates to < 4.2 mg per rabbit). Moreover, the tetracycline capsule group that did not receive any povidone had a higher pleurodesis score than the tetracycline tablet group. Previous studies have shown that only larger doses of povidone in humans (20 mg/kg) (17) or rabbits (40 mg in total) produce an effective pleurodesis (Yubiao Guo, MD; personal communication; December 2003). In addition to all of the above, the pleurodesis score of the tetracycline tablet group, or the combined oral group with or without tetracycline tablet group was comparable to that of the parenteral doxycycline group.
The median right pleurodesis scores obtained with the oral forms of tetracycline and doxycycline ranged from 4.0 to 8.0, being [greater than or equal to] 6.5 in three of the four groups. Thus, 70% (18 of 26 animals) of the oral group had a pleurodesis with many adhesions and accompanying symphysis involving [greater than or equal to] 5% of the hemithorax. The doxycycline capsule group had the lowest mean score, which was 4.0. It can be seen in Figure 1 that the pleurodesis score was 1.0 in three of the rabbits in this group. Furthermore, in these three cases, pleural fluid total volumes ([less than or equal to] 1.5 mL), and protein ([less than or equal to] 2.82 g/dL), LDH ([less than or equal to] 3,102 IU/L), and WBC ([less than or equal to] 10,370/[micro]L) levels tended to be lower than those of the other rabbits with higher pleurodesis scores in the same and other groups. We believe that technical problems with the pleurodesis explain these three low scores. Although we cannot prove it, we believe that the chest tubes were misplaced in these three rabbits such that the doxycycline partly or never entered the pleural space. Another possible explanation for the lower score in the doxycycline capsule group could be that the doxycycline was incompletely dissolved or that it was removed by the filter. However, when we dissolved the contents of one 100-mg doxycycline capsule in 10 mL of saline solution and then filtered, the measured concentration of doxycycline in the solution was 11.8 mg/mL with an expected concentration of 10.0 rag/mL. Accordingly, the poorer results with the doxycycline capsule cannot be blamed on lower concentrations of doxycycline in the filtrate.
To create a pleurodesis, usually the pleura is injured with a sclerosing agent that leads to an inflammatory response that can result in pleurodesis. After instillation of a sclerosing agent, the pleural inflammation is indicated by the accumulating exudative pleural fluid with high WBC, protein, and LDH levels. (12,18-20) In all of the 36 rabbits in the current study, the aspirated pleural fluid was an exudate with total volumes of 9.21 to 26.10 mL, and high LDH (> 1,900 U/L), WBC (> 2,500/[micro]L), and protein levels (> 2.90 g/dL). It also was bloody appearing in 83% of the rabbits. The positive correlation between the pleural score and fluid volume or protein level in the current study supports the importance of inflammation in pleurodesis. A positive correlation would also be expected for fluid LDH and WBC levels since they also are surrogates of inflammation. However, in the present study, pleural fluid LDH and WBC were not significantly correlated with the pleurodesis score.
There were some differences between the groups regarding the features of the pleural fluid. The pleural fluid WBC in the parenteral doxycycline group was significantly lower than that in the combined oral group with the highest pleural fluid WBC occurring in the tetracycline tablet group. One possible explanation for the higher WBC (8,623 to 17,998 cells/[micro]L) in the oral groups than the parenteral group (2,564/[micro]L) could be additional inflammation due to the fillers present in the oral forms. It is noteworthy that the group with the highest WBC level, the tetracycline tablet group, was the only group that received povidone as a filler. However, if this were the explanation, one would also expect the fluid volume, LDH, and protein levels to be significantly higher; however, they were all comparable. Furthermore, tetracycline tablet did not induce the highest pleurodesis score. In fact, it induced the second lowest among the other scores. Thus, the highest WBC level in the tetracycline tablet group did not result in a significantly higher pleurodesis score in this group.
Tetracycline and doxycycline are dose-dependent effective inhibitors of matrix-degrading metalloproteinase (MMP) activity in pleural fluid. They may also reduce the synthesis of MMP via nonspecific cell injury. At a concentration of [greater than or equal to] 1 mg/mL, both drugs reduce MMP activity by > 75%. These data suggest one possible mechanism for tetracycline or doxycycline pleurodesis. (21) Thus, the pleural fibrotic process needed for pleurodesis is augmented by the reduction of MMP activity that favors remodeling through normal repair of the tissue rather than fibrosis. (22) Another proposed mechanism is that tetracyclines stimulate mesothelial cells to release a growth factor-like activity for fibroblasts. (23) Furthermore, both tetracycline (24) and doxycycline (12) pleurodesis is effective at neutral pH, suggesting a mechanism of action independent of acid injury to the mesothelium.
That most of the aspirated pleural fluids (83%) after intrapleural injections of tetracyclines were bloody appearing might have produced a positive effect on pleurodesis since bloody fluids may lead to formation of adhesions and, thus, augment pleurodesis. Previously, tetracyclines have been shown to cause bloody effusions and hemothoraces in animal studies. (10,12,18) These bloody effusions may be a component in the natural course of pleurodesis by tetracyclines (intense pleural inflammation [right arrow] exudative pleural effusion + neovascularization + increasing negative intrapleural pressure due to fibrosis [right arrow] rupture of newly formed fragile capillaries [right arrow] bloody effusions). The critical point is that excess of these fluids should be aspirated to bring visceral and parietal pleurae to apposition for an effective pleurodesis. In the current study, appropriate daily aspirations of the pleural fluids were performed.
Right hemothoraces occurred in a total of nine rabbits receiving intrapleural tetracycline derivatives. Although a high incidence of hemothorax (10,12) and substantial mortality (10) was reported in rabbits administered intrapleural tetracycline derivatives without chest tube drainage, in a more recent study, (18) insertion of a chest tube and daily aspirations of the accumulating fluid have prevented hemothorax and led to a better pleurodesis in rabbits. Similarly, hemothorax or mortality has not been reported in human subjects receiving intrapleural tetracycline or its derivatives and also being drained with chest tube. (2-6,15,24-28) In the current study, the complications including right or bilateral hemothorax and ascites did not have a significantly adverse effect on the pleurodesis efficiency since the pleurodesis scores ranged between 6 and 8 in these rabbits daily aspirated for pleural fluid. Furthermore, there was no major morbidity or mortality due to these complications. No infectious complication was observed in any of the rabbits. This may be at least partly due to the fact that the solutions prepared with the oral forms were filtered to eliminate any infectious agents. Moreover, antibiotic treatment until removal of the chest tube, removal of the chest tube right after pleural fluid is < 5 mL, meticulous care for sterility during and after surgery, and the antibiotic nature of the sclerosing agents used might have prevented infection. As evidenced by animal (9-12,18,19) and human studies, (2-6,15,24-28) infectious complications due to use of tetracycline and its derivatives in pleurodesis are not as frequent as those with other agents such as talc, (29) Corynebacterium parvum, (30) and cytotoxic agents (30) (almost none vs 3%).
Intense pleural pain caused by the intrapleural injection of tetracycline or its derivatives is a well-known complication of pleurodesis. Usually, conscious sedation with benzodiazepines or analgesia with a narcotic drug is recommended before starting pleurodesis with tetracycline and its derivatives. (3-6,25-28) In our study, we did not observe signs of pain in any rabbit, but this is probably due to the fact that the tetracycline compounds were injected while the rabbit was still under anesthesia.
The cost of oral forms of tetracycline or doxycycline for each rabbit is approximately 1/50 to 1/5 of parenteral doxycycline: < $1 vs nearly $5. When proportioned to a 60-kg human, these costs are < $20 vs nearly $100. The cost of the filter we used for each rabbit in filtering oral tetracycline and doxycycline is $1.12. Thus, the total expense of pleurodesis increases to < $2.2 per rabbit and $21.2 per human. However, the cost of oral forms of tetracycline or doxycycline combined with that of filter still remains considerably lower than the cost of parenteral doxycycline.
In conclusion, the present study demonstrates that the intrapleural injection of the oral forms of tetracycline and doxycycline dissolved in sterile saline solution produces a pleurodesis that is comparable to that produced by the intrapleural injection of parenteral doxycycline in rabbits. Thus, in countries where parenteral tetracycline and doxycycline are not available, oral forms of these drugs may also be used in humans for pleurodesis provided that meticulous care for avoiding infection is exercised.
REFERENCES
(1) Light RW. Pleural diseases, 4th ed. Philadelphia, PA: Lippincott, Williams and Wilkins, 2001; 120-123
(2) Hartman DL, Gaither JM, Kesler KA, et al. Comparison of insufflated talc under thoracoscopic guidance with standard tetracycline and bleomycin pleurodesis for control of malignant pleura/effusions. J Thorac Cardiovasc Surg 1993; 105: 743-747; discussion 747-748
(3) Heffner JE, Standerfer RJ, Torstveit J, et al. Clinical efficacy of doxycycline pleurodesis. Chest 1994; 105:1743-1747
(4) Alfageme I, Moreno L, Huertas C, et al. Spontaneous pneumothorax: long-term results with tetracycline pleurodesis. Chest 1994; 106:347-350
(5) Patz EF Jr, McAdams HP, Erasmus JJ, et al. Sclerotherapy for malignant pleural effusions: a prospective randomized trial of bleomycin vs doxycycline with small-bore catheter drainage. Chest 1998; 113:1305-1311
(6) Martinez-Moragon E, Aparicio J, Rogado MC, et al. Pleurodesis in malignant pleural effusions: a randomized study of tetracycline versus bleomycin. Eur Respir J 1997; 10:2380-2383
(7) Lee YCG, Baumann MH, Maskell NA, et al. Pleurodesis practice for malignant pleural effusions in five English-speaking countries: survey of pulmonologists. Chest 2003; 124:2229-2238
(8) Rehse DH, Aye RW, Florence ME. Respiratory failure following talc pleurodesis. Am J Surg 1999; 177:437-440
(9) Vargas FS, Teixeira LR, Silva LM, et al. Comparison of silver nitrate and tetracyeline as pleural sclerosing agents in rabbits. Chest 1995; 108:1080-1083
(10) Light RW, Wang NS, Sassoon CS, et al. Comparison of the effectiveness of tetracycline and minocycline as pleural sclerosing agents in rabbits. Chest 1994; 106:577-582
(11) Mitchem RE, Herndon BL, Fiorella RM, et al. Pleurodesis by autologous blood, doxycycline, and talc in a rabbit model. Ann Thorac Surg 1999; 67:917-921
(12) Hurewitz AN, Lidonicci K, Wu CL, et al. Histologic changes of doxycycline pleurodesis in rabbits: effect of concentration and pH. Chest 1994; 106:1241-1245
(13) Light RW, Cheng DS, Lee YC, et al. A single intrapleural injection of transforming growth factor-[beta](2) produces an excellent pleurodesis in rabbits. Am J Respir Crit Care Med 2000; 162:98-104
(14) Gary Lee YC, Teixeira LR, Devin CJ, et al. Transforming growth factor-[beta]2 induces pleurodesis significantly faster than talc. Am J Respir Crit Care Med 2001; 163:640-644
(15) Dryzer SR, Allen ML, Strange C, et al. A comparison of rotation and nonrotation in tetracycline pleurodesis. Chest 1993; 104:1763-1766
(16) Medical Economics Staff, ed. Physicians' desk reference 2003, 57th ed. Montvale, NJ: Thomson PDR, 2003; 2194, 2196
(17) Olivares-Torres CA, Laniado-Laborin R, Chavez-Garcia C, et al. Iodopovidone pleurodesis for recurrent pleural effusions. Chest 2002; 122:581-583
(18) Wu W, Teixeira LR, Light RW. Doxycycline pleurodesis in rabbits: comparison of results with and without chest tube. Chest 1998; 114:563-568
(19) Vargas FS, Wang NS, Lee HM, et al. Effectiveness of bleomycin in comparison to tetracycline as pleural sclerosing agent in rabbits. Chest 1993; 104:1582-1584
(20) Kennedy L, Harley LA, Sahn SA, et al. Tale slurry pleurodesis: pleural fluid and histologic analysis. Chest 1995; 107: 1707-1712
(21) Hurewitz AN, Wu CL, Mancuso P, et al. Tetracycline and doxycycline inhibit pleural fluid metalloproteinases: a possible mechanism for chemical pleurodesis. Chest 1993; 103:1113-1117
(22) Marshall BC, Santana A, Xu QP, et al. Metalloproteinases and tissue inhibitor of metalloproteinases in mesothelial cells: cellular differentiation influences expression. J Clin Invest 1993; 91:1792-1799
(23) Antony VB, Rothfuss KJ, Godbey SW, et al. Mechanism of tetracycline-hydrochloride-induced pleurodesis: tetracycline-hydrochloride-stimulated mesothelial cells produce a growth-factor-like activity for fibroblasts. Am Rev Respir Dis 1992; 146:1009-1013
(24) Varela G, de Pablo P, Ruiz MJ. Pleurodesis with tetracycline in neoplastic effusions: is an acid pH necessary? Rev Med Univ Navarra 1988; 32:143-146
(25) Salomaa ER, Pulkki K, Helenius H. Pleurodesis with doxycycline or Corynebacterium parvum in malignant pleural effusion. Acta Oncol 1995; 34:117-121
(26) Herrington JD, Gora-Harper ML, Salley RK. Chemical pleurodesis with doxycycline 1 gr. Pharmacotherapy 1996; 16: 280-285
(27) Pulsiripunya C, Youngchaiyud P, Pushpakom R, et al. The efficacy of doxycycline as a pleural sclerosing agent in malignant pleural effusion: a prospective study. Respirology 1996; 1:69-72
(28) Prevost A, Nazeyrollas P, Milosevic D, et al. Malignant pleural effusions treated with high dose intrapleural doxycycline: clinical efficacy and tolerance. Oncol Rep 1998; 5:363-366
(29) Viallat JR, Rey F, Astoul P, et al. Thoracoscopic talc poudrage pleurodesis for malignant effusions: a review of 360 cases. Chest 1996; 110:1387-1393
(30) Chella A, Ribechini A, Dini P, et al. Treatment of malignant pleural effusion by percutaneous catheter drainage and chemical pleurodesis. Minerva Chir 1994; 49:1077-1082
* From the Department of Pulmonary Medicine (Dr. Bilaceroglu), Izmir Training and Research Hospital for Thoracic Medicine and Surgery, Izmir, Turkey. Department of Pulmonary Medicine (Drs. Hawthorne and Light), Vanderbilt University and St. Thomas Hospital, Nashville, TN; Department of Respiratory and Critical Care Medicine (Drs. Guo and Zhu), First Affiliated Hospital of Sun Yatsen University, Guangzhou, PR China; and Department of Pulmonary Medicine (Drs. Stathopoulos and Lane), Vanderbilt University, Nashville, TN.
This study was presented as a poster at the ACCP Congress, October 23-28, 2004, Seattle, WA.
Tiffs study was supported by the Saint Thomas Foundation, Nashville, TN.
Manuscript received March 31, 2005; revision accepted June 24, 2005.
Reproduction of this article is prohibited without written permission from the American College of Chest Physicians (www.chestjournal. org/mise/reprints.shtml).
Correspondence to: Semra Bilaceroglu, MD, FCCP, Associate Professor of Pulmonology, 6026 Sokak, No. 197/22, 35560 Bostanli, Izmir, Turkey; e-mail: semrab@superonline.com
COPYRIGHT 2005 American College of Chest Physicians
COPYRIGHT 2005 Gale Group