Definition
Thalassemia is an inherited disorder that affects the production of hemoglobin and causes anemia. Hemoglobin is the substance in red blood cells that enables them to transport oxygen throughout the body. It is composed of a heme molecule and protein molecules called globins. Owing to an inherited genetic trait, lower-than-normal amounts of globins are manufactured in the bone marrow. If the trait is inherited from both parents, a globin may be entirely absent. Thalassemia causes varying degrees of anemia, which can range from insignificant to life threatening. People of Mediterranean, Middle Eastern, African, and Asian descent are at higher risk of carrying the genes for thalassemia.
Description
Hemoglobin molecules are vital for transporting oxygen throughout the body. Hemoglobin and red blood cell production is carried out in the bone marrow, a spongy tissue found within certain bones, such as the hips, skull, and breast bone. The hemoglobin molecule is actually made up of five smaller component molecules: a heme molecule and four protein molecules called globins. Normal hemoglobin molecules contain two pairs of different globin molecules.
Humans have the genes to construct six types of globins, but do not use all six at once. Different globins are produced depending on the stage of development: embryonic, fetal, or adult. During embryonic development, hemoglobin contains two zeta-globin molecules and two epsilon-globin molecules. At the fetal stage, the body switches to alpha-globin and gamma-globin production. Within weeks after birth, an infant continues to produce alpha-globin, but gamma-globin is replaced by beta-globin and a very minor amount of delta-globin. After the first two to three months of life, most hemoglobin in the body is composed of two alpha-globins and two beta-globins; approximately 0.5% is composed of two alpha-globins and two delta-globins.
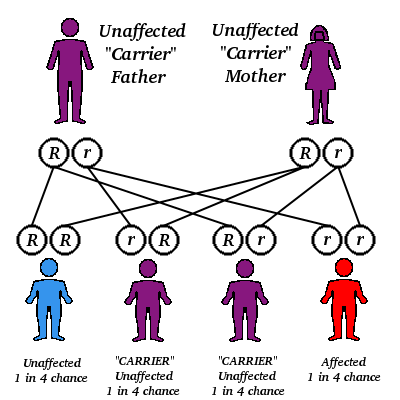
There is a different gene for each type of globin, with the exception of alpha-globin, which has two genes. (Genes are inherited in pairs, one copy from each parent.) A gene mutation may lead to inadequate levels of the related globin, reduced hemoglobin formation, and anemia. Such mutations are the underlying cause of thalassemia.
Thalassemia is classified according to the globin that is affected. The most common types of thalassemia are beta-thalassemia and alpha-thalassemia. Beta-thalassemia is caused by a mutation in the gene responsible for beta-globin. If a mutated beta-globin gene is inherited from both parents, the result is beta-thalassemia major, a severe, potentially life-threatening anemia. Beta-thalassemia major may also be referred to as Cooley's anemia or erythroblastic anemia. If only one mutated copy of the beta-globin gene is inherited, mild-to-nonexistent symptoms may appear; this condition is called beta-thalassemia minor. A person with one mutated copy of the beta-globin gene is referred to as a carrier of the beta-thalassemia trait.
The alpha-thalassemias are more complex because a person inherits two alpha-globin genes from each parent, yielding a total of two pairs of alpha-globin genes. Mutations in these genes can give rise to a range of symptoms. As long as adequate levels of alpha-globins are produced, the person--otherwise, called the carrier of the alpha-thalassemia trait--will have few, if any symptoms. In cases in which alpha-globin is severely reduced, or not produced at all, the consequences can be fatal during fetal development or shortly after birth.
People with Mediterranean (including North African), Middle Eastern, or southeast Asian ancestry are at higher risk of being carriers of or developing beta-thalassemia than are other populations. Alpha-thalassemia also is more likely to affect people of Mediterranean, African, Middle Eastern, and southeast Asian descent. In some areas, 1 in 150-200 children are born with thalassemia major. It has been estimated that 2 million people in America carry the thalassemia trait. When two carriers of the same type thalassemia produce a child, there is a 25% possibility that the child will inherit moderate or severe thalassemia.
Causes & symptoms
Thalassemia arises from mutations in one or more globin genes, leading to a reduction or absence of the associated globin. The severity of symptoms is directly related to whether one or both genes in a pair is mutated. However, symptoms may be modified by other genetic or environmental factors.
Beta-thalassemia
The inheritance of one mutated beta-globin gene is accompanied by few, if any symptoms. A person may have mild anemia, but may not even be aware of being a carrier unless tested. Anemia may only appear during pregnancy or following severe infections.
An inheritance of two mutated beta-globin genes causes a potential cascade of symptoms. Some individuals develop mild symptoms that do not require treatment, but others experience life-threatening anemia and complications. At the earliest, symptoms appear several weeks after birth; that is, when fetal gamma-globin production gives way to beta-globin production. Alpha-globin is produced normally and, relative to beta-globin, it is over-produced. This excess alpha-globin precipitates in the immature red blood cells, destroying them.
The resulting anemia triggers a surge in red blood cell production, but the new cells are also defective. The bone marrow expands as it attempts to keep pace with the perceived need for new red blood cells, setting the stage for bone deformity and pain. Jaundice, indicated by yellowed skin, can result from high levels of bilirubin (the end product of hemoglobin degradation). The spleen can become abnormally large and effects on the immune system increase the vulnerability to infection. At the same time, a lot of energy is invested in red blood cell production, stunting development and growth. Individuals who produce high levels of delta-globin, or continue to produce gamma-globin, have less severe symptoms. Delta- or gamma-globin can compensate, at least in part, for the missing beta-globin.
Alpha-thalassemia
Similar to beta-thalassemia, if enough alpha-globin is being produced, symptoms may be nonexistent. If the alpha-globin levels are low enough to produce symptoms, onset may begin early in life as alpha-globin production starts during the fetal stage.
Symptoms vary widely. Some individuals with alpha-thalassemia have symptoms similar to those of beta-thalassemia major; others develop mild anemia, a variably enlarged spleen, and potential bone changes. If alpha-globin is not produced, the risk of death is very high. Gamma- or beta-globin production is normal, but instead of precipitating, these proteins form abnormal hemoglobin molecules that contain four copies of the same globin. These abnormal hemoglobins can carry oxygen, but they do not release it easily. Tissues remain starved for oxygen and the fetus is extremely anemic.
Diagnosis
Thalassemia may be diagnosed from the symptoms; however, with proper medical treatment, a diagnosis may be made before symptoms become life- or health-threatening. Basic information that is used in diagnosis includes race and ethnic background, family history, and age. Unexpectedly slow development, along with pallor, jaundice, enlarged spleen or liver, or deformed bones can be common signs of thalassemia.
Laboratory tests are used to confirm a diagnosis and determine the type of thalassemia. These tests can also be used to identify carriers. A blood count is done in which the numbers of red blood cells are calculated. The size of the blood cells and the ratio of mature to immature cells are also determined. A higher-than-normal presence of unusually small or immature red blood cells indicate a problem in red blood cell production. Further tests measure the amount of hemoglobin; a low concentration indicates anemia. Hemoglobin molecules can be separated based on the component globin molecules, which aids in diagnosing the type of thalassemia. This process is known as hemoglobin electrophoresis.
Treatment
Thalassemia cannot be cured; therapy focuses on managing symptoms. Treatment is not necessary for individuals who are unaffected or only develop mild symptoms. The mainstays of thalassemia management are blood transfusions and iron chelation therapy. Blood transfusions are typically given every 6-8 weeks, but may be more frequent in some cases. These transfusions have two purposes: to keep hemoglobin at or near normal levels and to prevent the bone marrow from producing ineffective red blood cells.
Repeated transfusions carry the risk of iron loading, a condition in which the body accumulates too much iron. As the iron stores become too large, iron deposits form in the liver, heart, and endocrine glands. These iron deposits cause organ damage and, left unchecked, death. Iron chelation therapy begins between ages three to five with desferrioxamine. The desferrioxamine is administered under the skin of the abdomen or via an implanted venous (vein) port. Once the appropriate dose for an individual is determined, the drug must be administered on a daily or near-daily basis.
Thalassemia has been treated with bone marrow transplantation. However, bone marrow transplants are strictly limited by several factors, including the general health of the marrow recipient and whether a donor with compatible marrow can be found. Bone marrow transplants are risky--fatality rates range between 10-30%--and success cannot be guaranteed.
Prognosis
An individual's outlook depends on the exact type of thalassemia and the associated genes. In the carrier state, a person may never develop symptoms. Other carriers develop mild anemia at times of extreme stress, such as pregnancy or illness. Some forms of alpha-thalassemia, in which alpha-globin is severely reduced or absent, are nearly always fatal. Affected fetuses often die during weeks 34-40 of a pregnancy; some infants are born alive, but die within hours of birth.
Persons who are at risk of severe anemia and other complications may do well on a regimen of blood transfusions and iron chelation therapy. For this treatment to be effective, it must be followed assiduously. Blood transfusions and iron chelation therapy were begun in the 1960s. Survival into middle age is possible for some transfusion-dependent individuals; however, predicting longer-term survival is not possible because of the relatively short history of the treatment regimen.
Problems such as an enlarged spleen and bone deformation are unusual with blood transfusion therapy. Transfusion side effects, such as sensitization to blood antigens, occur in 5-15% of individuals. Rarely, a viral infection may be passed on to the blood transfusion recipient. Iron chelation therapy has been linked to nerve damage in about 30% of individuals on long-term iron chelation therapy. Damage is limited to vision and hearing, and it is recommended that individuals have their hearing and vision assessed regularly.
Prevention
Once the genes that determine thalassemia are inherited, the disease cannot be prevented. Screening offers the opportunity of identifying thalassemia carriers. Carriers may decide to undergo genetic counseling to assess potential risks to their children. Finally, prenatal testing, usually chorionic villus sampling or amniotic fluid testing, allows identification of thalassemia in unborn children.
Key Terms
- Anemia
- A medical condition in which blood hemoglobin falls to abnormally low levels. Major symptoms include pallor, weakness, and a lack of energy.
- Bilirubin
- A yellow pigment that is the end result of hemoglobin breakdown. This pigment is metabolized in the liver and excreted from the body through the bile. Bloodstream levels are normally low; however, extensive red cell destruction leads to excessive bilirubin formation and jaundice.
- Bone marrow
- The spongy tissue found in the center of certain bones. This tissue is the site of blood cell generation.
- Bone marrow transplantation
- A medical procedure used to treat some diseases that arise from defective blood cell formation in the bone marrow. Healthy bone marrow is extracted from a donor to replace the marrow in an ailing individual. Proteins on the surface of bone marrow cells must be identical or very closely matched between a donor and the recipient.
- Desferrioxamine
- The primary drug used in iron chelation therapy. It aids in counteracting the life-threatening buildup of iron in the body that is associated with long-term blood transfusions.
- Globin
- One of the six proteins associated with the hemoglobin molecule. Different globins are produced in the body depending on the stage of development: zeta- and epsilon-globins are produced during the embryonic stage; alpha- and gamma-globin are produced during fetal life; and alpha-, beta-, and delta-globins are produced after birth.
- Heme
- The component molecule of hemoglobin that serves as the oxygen-binding site. It enables hemoglobin to transport and deliver oxygen throughout the body.
- Hemoglobin
- The pigment found in red blood cells that is made up of a heme molecule and two pairs of different globin molecules. Most hemoglobin in adults contains two alpha-globin molecules and two beta-globin molecules. Hemoglobin enables red blood cells to transport oxygen throughout the body.
- Iron chelation therapy
- A medical treatment to remove excess amounts of iron from the body.
- Iron loading
- A side effect of numerous blood transfusions in which iron levels in the body accumulate to dangerous levels. As levels become excessive, iron deposits form in the heart, liver, and other organs. These deposits cause organ damage and are fatal if untreated.
- Jaundice
- The yellowing of the skin caused by high bilirubin levels in the bloodstream. Jaundice can cause liver damage.
Further Reading
For Your Information
Books
- Weatherall, D.J. "The Thalassemias." In Williams Hematology, 5th ed., edited by Ernest Beutler, et al. New York: McGraw-Hill, Inc., 1995.
- Weatherall, D.J., J.B. Clegg, D.R. Higgs, and W.G. Wood. "The Hemoglobinopathies." In The Metabolic and Molecular Bases of Inherited Disease, 7th ed, edited by Charles R. Scriver, et al. New York: McGraw-Hill, Inc., 1995.
Periodicals
- Giardini, Claudio. "Treatment of β-Thalassemia." Current Opinion in Hematology 4 (1997): 79.
- Weatherall, D.J. "The Thalassemias." British Medical Journal 314 (June 7, 1997): 1675.
Organizations
- Cooley's Anemia Foundation, Inc. 129-09 26th Ave. #203, Flushing, NY 11354. (800) 522-7222 or (718) 321-2873. http://www.thalassemia.org/.
Gale Encyclopedia of Medicine. Gale Research, 1999.