If electrical impulses are taking the fast track and bypassing the atrioventricular node, trouble's afoot.
Although preexcitation syndromes such as Wolff-Parkinson-White (WPW) syndrome are uncommon, they can have life-threatening implications. Rapid, potentially fatal arrhythmias are far more common in patients with these conduction defects than in the general population.
Correctly identifying a preexcitation syndrome is essential because the wrong treatment can worsen the problem: Traditional therapy for supraventricular tachycardia (SVT), which can be confused with a preexcitation syndrome, can accelerate the heart rate and lead to death. In this article, I'll discuss preexcitation syndromes, with emphasis on WPW, the most common one.
On the fast track
Normally, electrical impulses in the heart pass through the atrioventricular (AV) node on their way to depolarize the ventricles. The AV node acts as a moderator, slowing the impulse before it reaches the ventricles to allow for proper coordination between the upper and lower heart chambers. This brief pause lets blood travel through the AV valves into the ventricles before ventricular contraction occurs.
The AV node also limits the number of electrical impulses that reach the ventricles. In some arrhythmias, the atria can deliver more than 200 impulses to the AV node every minute. The AV node doesn't let them all pass because at that rate, the ventricles have no time to fill and limited time to contract, resulting in a precipitous drop in cardiac output and blood pressure.
However, in a patient with a preexcitation syndrome, the electrical impulses bypass the AV node. Instead, they travel on an alternative pathway (also called a bypass tract or accessory pathway) that has no delays or rate limits. Consequently, the atrial impulse reaches the ventricles sooner than if it had gone via the AV node. Under certain circumstances, these bypass tracts can allow dangerously fast rhythms to occur.
A bypass tract can occur in any area of the heart that permits the depolarization wave to reach the ventricles prematurely. Bypass tracts-connections made up of strands of atrial-like muscle-can occur in any number of combinations involving the atria, ventricles, AV node, bundle of His, or fascicles and can occur on either side of the heart. Each anomalous connection produces specific changes on the 12-lead electrocardiogram (ECG) that will provide clues to the location and type of bypass tract.
Trouble in the delta
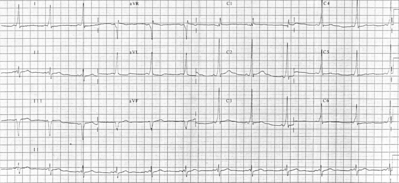
In WPW, the most common preexcitation syndrome, the bypass tract is the bundle of Kent, a direct connection between an atrium and ventricle. This congenital condition was first described in otherwise-healthy young people with recurrent paroxysmal supraventricular tachycardia (PSVT) whose baseline ECG rhythm showed a short PR interval and a bundle-branch block pattern (findings common in many preexcitation syndromes). However, WPW syndrome has these distinct hallmarks:
* a short PR interval (less than 0.12 second). The absence of the normal AV nodal delay causes the QRS to occur earlier and therefore closer to the P wave. Remember that the PR interval also shortens with an increased heart rate and in some atrial and junctional rhythms.
* a delta wave. This small segment of the QRS, which is frequently referred to as an abnormal "slurring of the QRS" (see Conduction in Wolff-Parkinson-White Syndrome), represents early, abnormal depolarization of the ventricles. Negative delta waves in lead V^sub 1^ indicate a right-sided bypass tract and can mimic the pathologic Q-waves of myocardial infarction.
* a wide QRS complex. The QRS complex is greater than 0.1 second (normal is 0.06 to 0.1 second) because the electrical impulse depolarizes the ventricles earlier than usual. A small amount of ventricular tissue begins to depolarize near the bypass tract, activating before the rest of the ventricle, but because it doesn't use the fast bundle-branch pathway, the QRS complex is wider than normal.
Measuring preexcitation
You can determine the amount of preexcitation from the distortion present in the PR interval. The amount of ventricular tissue depolarized from the bypass tract can vary from none to all, depending on several factors, including the distance from the sinus node to the bypass tract and the difference in the conduction times between the AV node and the bundle of Kent.
A small delta wave results if AV node conduction is enhanced and the bypass tract has less time to conduct the impulse. A small delta wave also occurs if the bypass tract is in the left lateral wall (and therefore farther away from the sinus node) because the AV node has more time to conduct normally.
To determine the role the bypass tract plays in ventricular depolarization, look at the PR interval. If the QRS complex begins before the P wave ends, then the ventricles are completely depolarized (or nearly so) from the bypass tract. This can occur with a prolonged first-degree AV block or a third-degree AV block; both can allow the electrical impulses using the bypass tract time to reach both ventricles first. As a result, the entire QRS complex appears as a delta wave, which can make diagnosis from the 12-lead ECG difficult if not impossible.
If some of the PR segment is present, then the QRS complex represents a fusion beat or some degree of depolarization from both the bypass tract and the AV node.
A large delta wave means that more tissue is being activated by electrical impulses conducted to the ventricles via the bundle of Kent.
No delta wave, a normal PR interval, and a normal-width QRS may mean that a bypass tract, despite being capable of conducting electrical impulses, doesn't. More on this later.
Around and around
Circus movement tachycardias (CMTs) in WPW syndrome, also known as AV reentrant tachycardias, are caused by differing refractory periods of the bypass tract and the AV node. One pathway is capable of accepting an electrical impulse when the other is not. Therefore, during PSVT in WPW syndrome, the impulse usually travels in an antegrade fashion, from the atria through the AV node and His-Purkinje system to the ventricles, and then retrograde through the bypass tract (which is no longer refractory) back to the atrium, where the process repeats itself. This is called orthodromic CMT. A circular pathway is now formed, thus the term circus. Any event or medication that blocks or slows only one pathway can precipitate a CMT.
Premature beats, either atrial or ventricular, can precipitate a CMT. The premature beat reaches both the AV node and the bypass tract and finds one of them still refractory. The electrical impulse is then conducted down to the ventricles using one pathway, leaving the other available to conduct the impulse in the opposite direction.
Sinus tachycardia at a trigger rate (which varies from person to person) can also initiate a CMT. Above this rate, one pathway is refractory and the other isn't. This disparity causes the development of a circular pathway as described above.
As mentioned previously, in an orthodromic CMT, conduction is antegrade through the AV node and His-Purkinje system and retrograde to the atria through the bypass tract. This results in a normalwidth QRS complex without a delta wave because the ventricles are depolarized normally through the bundle branches. Because the impulses travel through the AV node, ventricular response rates typically aren't as fast.
In antidromic CMTs, which occur in about 5% of WPW patients with PSVT, the pattern is reversed. Conduction is antegrade through the bypass tract and retrograde through the normal AV system. This results in a tachycardia with a wide QRS complex because the ventricles are activated by the bypass tract.
Antidromic CMTs can be triggered by atrial flutter and atrial fibrillation (AF). Normally, the AV node prevents many of the atrial impulses from reaching the ventricles, but the bypass tract doesn't have these rate-limiting properties. As a result, impulses travel rapidly down the bypass tract and through the ventricular tissue, triggering rapid ventricular response rates.
Atrial fibrillation and flutter
Atrial fibrillation and atrial flutter are extremely dangerous in patients with a bypass tract that can conduct impulses in an antegrade fashion. The sheer number of impulses the atria can generate in these arrhythmias causes the problem because the bypass tracts aren't as rate-discriminating as the AV node. They can conduct more than 200 impulses/minute to the ventricles, resulting in symptoms such as palpitations and signs of low cardiac output such as hypotension.
The resultant tachycardia resembles ventricular tachycardia (VT), except that it's irregular. This is an important diagnostic aid: VT is regular more than 75% of the time.
The QRS configuration will also be dramatically different from that during sinus rhythm because of the abnormal antegrade impulse conduction through the bypass tract.
Unlike the CMTs discussed earlier, this rapid arrhythmia isn't due to a reentry phenomenon, but rather from direct conduction of atrial impulses through the bypass tract to the ventricles.
Hidden bypass tracts
Sometimes, during a normal sinus rhythm, no impulse is conducted down the bypass tract, and the patient's ECG looks perfectly normal. However, after a precipitating event (such as a premature atrial contraction triggering PSVT at a rate that exceeds the AV node's refractory period), the bypass tract begins conducting electrical impulses from the atria and an orthodromic or antidromic CMT is initiated, depending on the direction the bypass tract is conducting the electrical impulse. This is called a latent bypass tract.
A concealed bypass tract is similar to a latent bypass tract in that during normal rates and rhythms, the ECG shows no signs of the underlying bypass tract. This is because the bypass tract can transmit electrical impulses in the retrograde direction only, and thus no ventricular preexcitation occurs.
This anomaly is difficult to detect. Careful evaluation and comparison of the 12-lead ECGs both during sinus rhythm and during PSVT is vital to diagnosis. Even with intense scrutiny, electrophysiology studies may be needed for diagnosis.
Be careful not to confuse latent and concealed bypass tracts. The inherent risks of the tachycardias, particularly those associated with AF, are less severe with a concealed bypass tract than with a latent one. This is because in AF, the concealed bypass tract isn't capable of transmitting the atrial impulses directly to the ventricles, and the AV node can provide some measure of protection from excessively high ventricular rates.
SELECTED REFERENCES
ECG Interpretation Made Incredibly Easy!, 2nd edition. Springhouse, Pa., Springhouse Corp., 2001.
Lewis, K., and Handal, K.: Sensible ECG Analysis. Albany, N.Y., Delmar Learning, 1999.
By Henry B. Geiter, Jr., RN, CCRN
Henry B. Geiter, Jr., is a pool nurse at Bayfront Medical Center in St. Petersburg, Fla.; a critical care transport nurse for Sunstar-EMS in Clearwater, Fla.; and an adjunct instructor at St Petersburg College.
Copyright Springhouse Corporation Nov 2003
Provided by ProQuest Information and Learning Company. All rights Reserved