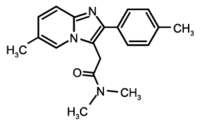
A case is reported in which a patient experienced somnambulistic episodes only after taking zolpidem tartrate for insomnia. Previous to the patient's use of zolpidem tartrate he had never experienced sleepwalking, and once the medication was discontinued the sleepwalking stopped. A search of the literature revealed only two other cases of zolpidem-induced sleepwalking, both involving individuals with a previous history of somnambulism in their youth.
Introduction As the use of zolpidem tartrate (Ambien) becomes increasingly more accepted in the United States as a considerably safe, nonbenzodiazepine hypnotic and the number of patients prescribed this medication increases, the number of reported side effects should also increase. A recent search of the psychopharmacological literature revealed only two reported cases of somnambulism occurring after the patient had taken 10 mg of zolpidem tartrate for insomnia. We describe a third, rather unusual case of sleepwalking that occurred with a patient who was recently prescribed zolpidem tartrate. Case Report The patient was a 46-year-old, married, white, male U.S. Army officer who was being seen for a complaint of insomnia felt to be caused by the inability to establish a regular sleeping pattern during the preceding months. He was not able to set a regular sleep schedule because of occupational demands placed on his unit, which was faced with an increased mission load. Other mental illness had been painstakingly ruled out by a battery of psychological tests (these tests included a computerassisted version of the Structured Clinical Interview for DSM-IV [SCID], a computer-assisted version of the SCID II [which evaluates for personality disorder], an in-depth biopsychosocial history, and a clinical interview). The patient denied any significant stressors occurring in his life. He was begun on a brief, temporary regimen of zolpidem tartrate 10 mg as needed for sleep. He had a previous history of major depression (single episode, moderate, without psychosis) 4 years earlier, which was successfully treated with a selective serotonin reuptake inhibitor for 1 year and then discontinued without recurrence of symptoms. Also, as a veteran of Operations Desert Shield and Desert Storm, in which the patient served as an Apache helicopter pilot, he had undergone a thorough screening for psychological and physical illness soon after being diagnosed with major depression in 1993. The patient remained in excellent health and continued to run 3 to 6 miles daily and denied the use of any illicit substances, tobacco products, or alcohol.
Before beginning the psychopharmacological treatment of his insomnia, the patient was given a routine screening polysomnogram, which was normal except for the decreased sleep time and the increased sleep latency. Recent routine laboratory tests were normal, and a urine drug screen was negative.
The first 3 nights that the patient took the 10 mg of zolpidem tartrate before sleep were uneventful, and the patient stated that he had total relief of the insomnia. On the 4th night, the patient stated that he was noted by the other members of his household to arise at approximately 1:00 a.m. and begin to prepare a meal. Upon completion of the preparation of the meal, he consumed it and returned to bed. Mild attempts by the household members to interact with the patient were completely ignored by the patient, and so they returned to bed, unaware that the patient was sleepwalking. When the patient was harassed the next morning for leaving the kitchen in a state of disarray, he denied any knowledge of the previous night's culinary activities. Both the patient and the other members of the household believed the other parties to be playing a practical joke on the other. The same scenario occurred repeatedly for several nights, and each party continued in its denial. It was only after the patient's spouse showed the patient a videotape of his previous night's culinary sleepwalking activities that the patient contacted J.H. with the complaint of somnambulism.
The patient brought the videotape to the appointment for viewing. (Because of this individual's current occupation in the military, he requested complete confidentiality.) During the recorded somnambulistic episode, the patient microwaved some leftovers from a previous meal, ate the reheated food, was engaged in a conversation in which his reply was mostly gibberish, and returned to bed. The entire episode took approximately 24 minutes. The decision to discontinue the zolpidem tartrate was agreed to immediately, and the patient's insomnia returned that night. After a literature search for articles describing zolpidem and sleepwalking, the patient was questioned regarding any previous history of somnambulism. He denied any childhood or adult history of sleepwalking. Mendelson' implies that a history of childhood somnambulism may be a factor in individuals who have a somnambulistic response to zolpidem. According to the observations of Nicholson and Pascoe2 regarding the age of subjects exposed to zolpidem and its effects on slow-wave (delta) sleep, only young adults (not middle-aged adults) are noted to have an increase in slow-wave sleep. Iruela3 hypothesizes that several criteria may be necessary for a subject to develop druginduced somnambulism. These criteria are a past history of sleepwalking, a medication increasing slow-wave sleep, and a precipitating external or internal stimulus. Based on the observations of Nicholson and Pascoe2 regarding zolpidem tartrate's lack of effect on the middle-aged adult's delta sleep, it would appear that these criteria are unnecessary to elicit a somnambulistic response to zolpidem tartrate. (The subject's slow-wave response to zolpidem tartrate is unknown, and this criterion may still be required.)
In summary, as the population of individuals using zolpidem tartrate for insomnia increases, the number of cases of zolpidem tartrate-induced sleepwalking will increase. With an increase in more reported instances of zolpidem tartrate-induced sleepwalking, more data can be gathered to determine exactly what criteria, if any, are required to bring about this very fascinating effect.
References
1. Mendelson WB: Sleepwalking associated with zolpidem. Letter.) J Clin Psychopharmacol 1994;14: 150.
2. Nicholson AN, Pascoe PA: Hypnotic activity of zolpidem: night-time and daytime studies in young and middle-aged adults. In Imidazopyridines in Sleep Disorders, pp 231-40. Edited by Sauvanet JP, Langer SZ, Morselli PL. New York, Raven Press, 1988.
3. Iruela LM: Zolpidem and sleepwalking. J Clin Psychopharmacol 1995; 15: 223.
Guarantor: CPT Jeff Harazin, MC USA
Contributors: CPT Jeff Harazin, MC USA; MAJ Timothy R. Berigan, MC USA
Division of Mental Health, 82d Airborne, Fort Bragg, NC 28307-5000.
The opinions and assertions contained herein are the private views of the authors and are not to be construed as official or as reflecting the views of the Department of the Army, the Department of Defense, or the U.S. Government.
This manuscript was received for review in January 1998. The revised manuscript was accepted for publication in December 1998.
Copyright Association of Military Surgeons of the United States Sep 1999
Provided by ProQuest Information and Learning Company. All rights Reserved