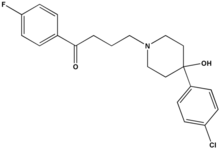
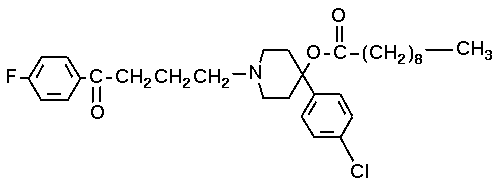
The long-term use of antipsychotic medication is an integral part of the therapeutic treatment of many persons with psychotic disorders. A recent meta-analysis of 81 studies confirmed the relationship between weight gain and antipsychotic agents and illustrated that some medications have a greater weight-promoting effect than others. For example, the average weight gain by persons taking the atypical antipsychotic medications clozapine and olanzapine over 10 weeks was 4.5 kg and 4.2 kg, respectively, whereas a weight gain of 1.1 kg was reported for haloperidol. Despite the methodologic limitations in many studies of the relationship between diabetes and atypical antipsychotic use, it also appears that persons treated with clozapine or olanzapine are at increased risk for developing hyperglycemia and diabetes. However, this may be independent of weight gain. Atypical antipsychotic medications are commonly prescribed in the clinical setting and over extended periods of time, which means that in some persons the weight-promoting effect of the pharmacologic agent may continue for years. Generalized equations cannot be used to predict resting energy expenditure (REE) in many clinical populations. REE prediction equations are ideally suited for participants/patients who are similar in terms of height, weight, age, race, and health status to the population on which the original formula was developed. There has been speculation regarding the effects of atypical antipsychotic medication on REE, specifically that drug-induced reduction in REE may contribute to weight gain. However, to date there have been no published guidelines to assist clinicians in choosing appropriate REE prediction equations. The objectives of this study were to measure REE via indirect calorimetry in a group of men taking clozapine and to determine whether REE can be accurately predicted using previously published regression equations for this population.
Men diagnosed with schizophrenia who had been taking clozapine for at least 6 months were recruited from the Park-Centre for Mental Health, a tertiary mental health facility in Brisbane, Australia. Body composition was determined by measuring total body water via the deuterium dilution method and REE was measured using a ventilated hood system. The equations of Harris-Benedict, Schofield, Movahedi, Owen and colleagues, and Jensen and colleagues were used to predict REE and are referred to as REE(HB), REE(S), REE(M), REE(O), and REE(J), respectively. The Harris-Benedict and Schofield equations were chosen because they are commonly used by practitioners. The predictive capability of the Movahedi equation was evaluated because it has been recommended in recently published guidelines on the treatment of obesity. The Owen and Jensen equations were chosen because they were derived using fat-free mass (FFM). The BMI of the reference group used to generate the equations was similar to that of the group studied.
Results from this study suggest that the commonly used Harris-Benedict, and Schofield equations can be used to predict REE in men who have been taking clozapine for 6 months, provided the predicted REE values are reduced by 280 kcal/day. Further efforts to develop specific prediction equations for persons taking clozapine and other atypical antipsychotic medications may assist with the clinical management of medication-induced weight gain.
J. Sharpe, N. Byrne, T. Stedman, A. Hills. Resting energy expenditure is lower than predicted in people taking atypical antipsychotic medication. JADA; 105(4):612-615 (April 2005) [Correspondence: Jenny-Kay Sharpe, MHSc, General Health Services, Park-Centre for Mental Health, Treatment, Research and Education, Locked Bag 500, Richlands 4077, Queensland, Australia. E-mail: JennyKay_Sharpe@health.qld.gov.au]
COPYRIGHT 2005 Frost & Sullivan
COPYRIGHT 2005 Gale Group