New avenues for LNS gene transfer
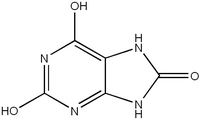
Lesch-Nyhan syndrome (LNS) sitshigh on the heap of most likely candidates for the first human gene transfer therapy. A neurologic disorder, LNS is the result of a single mutated gene that codes for a protein known as hypoxanthine phosphoribosyltransferase (HPRT). And restoring just 1 percent of normal HPRT levels may be enough to reduce a patient's LNS symptoms from compulsive self-mutilation to those of but a simple case of gout.
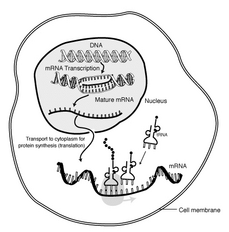
Gene therapy involves inserting a normalgene that will synthesize the needed product in order to correct a genetic or acquired disease. Of the two types of gene therapy, most research is concentrated on somatic-cell transfer, which targets the synthesis of the cell and affects only the patient. The other type, germ-cell transfer, enables the patient to pass the new gene to offspring.
Much of the work toward achievingsomatic-cell transfer in humans with LNS has been done using retroviruses, which are viruses that use RNA and synthesize it into DNA, in a process called reverse transcription. Through this method, an RNA gene coded with HPRT is transcribed into DNA and integrated into the host chromosomal DNA.
But one team of researchers now thinkswhat will be needed to correct LNS is a viral transfer that uses DNA rather than RNA. Led by Thomas D. Palella and William N. Kelley, researchers at the University of Michigan in Ann Arbor suggest that using the herpes simplex virus type 1 (HSV-1) will aid the transfer and expression of HPRT in deficient neuronal cells. Palella presented his findigns last week at the American Rheumatism Association meeting in Washington, D.C.
Part of the reason the team chose thiscarrier virus, or vector, says Palella, is because it will go directly to the central nervous system, whereas retroviruses cannot be directed there. In addition, retroviruses require replicating cells for integration, and cells in the central nervous system do not replicate. HSV-1, which is the type of herpes associated with cold sores, infects a cell by crawling up the cell's axon and harboring in the cell until activated. Because HSV-1 is incurable, researchers would somehow have to rid it of its disease effects before using it in gene transfer therapy.
The group, which has submitted itsfindings to a scientific journal for review, reports success in survival of human HPRT genes cloned in HSV-1 and transferred into rat neuronal cells. To make the recombinant virus, researchers took the thymidine kinase gene from the HSV-1 genome and cloned it with HPRT. The recombinant virus then successfully infected all cells receiving it. After 20 and 30 hours, the infected rat cells showed HPRT activity at levels comparable to those seen in a naturally occurring HPRT cell line.
Because herpes is a disease-causingvirus, rat cells responded by killing 80 percent of the naturally occurring herpes viral vectors infecting them. In contrast, 80 percent of the recombinant viral vectors were still alive in the rat cells after 30 hours, although researchers had done nothing to alter the disease effects of HSV-1. Part of this, Palella says, may be because interrupting the thymidine kinase chain automatically limits the disease effects of the HSV-1 genome.
Palella and his colleagues would likenext to insert HPRT into other viral genes in the HSV-1 genome to see how well the genes express the HPRT. The researchers also are looking into integrating promoters into a gene and then administering certain metals or steriods to help regulate levels of HPRT expression.
Palella emphasizes that his team's reportis preliminary and that there are many hurdles to overcome before HPRT gene transfer in humans can be realized. "In the gene transfer world, we're swimming upstream,' says Palella, "or at least against the current.'
COPYRIGHT 1987 Science Service, Inc.
COPYRIGHT 2004 Gale Group