DANBURY, Conn., Dec. 22 /PRNewswire-FirstCall/ -- Penwest Pharmaceuticals Co. today announced that Endo Pharmaceuticals Inc., Penwest's partner in the development and commercialization of oxymorphone extended- release tablets (oxymorphone ER), reported that it has filed what Endo believes is a complete response to the U.S. Food and Drug Administration's (FDA) approvable letter on its New Drug Application (NDA) for the investigational product oxymorphone ER.
Endo also said that, under the Prescription Drug User Fee Act (PDUFA) guidelines, the FDA has 60 days to decide if Endo's submission meets the FDA's criteria for a complete response and, if so, Endo expects that the FDA will issue an action letter for the NDA no later than 180 days from today.
As previously disclosed, on October 20, 2003, the FDA issued an approvable letter for oxymorphone ER but had requested that Endo address certain questions and provide more clarification and information, including data from an additional clinical trial, to further confirm the safety and efficacy of oxymorphone ER.
Endo stated in its release that if oxymorphone ER is approved within 180 days from today, Endo will be prepared to launch the product in the second half of 2006, and that oxymorphone ER would compete in the market for long- acting, strong opioids, which was a $4.2 billion market in 2004.
Jennifer L. Good, President and Chief Operating Officer of Penwest, said, "With Endo's filing of its response with the FDA, we have reached another important milestone in the process toward final approval for oxymorphone ER. We look forward to the approval and launch of this product, which we believe will offer physicians and patients who require continuous opioid therapy for an extended period of time a valuable new option for managing chronic, moderate-to-severe pain."
In order to meet the FDA's request for more clinical information for oxymorphone ER, Endo conducted two separate multi-center, randomized, double- blind, parallel group trials evaluating this product in two distinct groups of patients with chronic low back pain: opioid-naive and opioid-experienced. The trial involving opioid-naA[macron]ve patients was conducted under the FDA's Special Protocol Assessment (SPA) process. These trials were both 12-week trials and demonstrated statistically (p<0.0001) and clinically significant efficacy in these patient populations.
The data from the two new oxymorphone ER Phase III studies will supplement the previously submitted Phase III trials for oxymorphone ER that Endo believes the FDA has already accepted as demonstrating efficacy in the intended patient population.
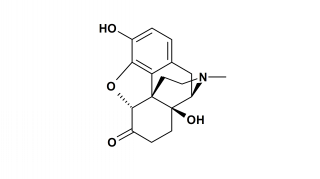
About Oxymorphone ER
Oxymorphone ER is a semi-synthetic u-opioid agonist that has been formulated as a 12-hour extended-release tablet using Penwest Pharmaceuticals' proprietary time-release technology, TIMERx(R) delivery system. Oxymorphone ER has been studied in a wide range of chronic pain conditions, including low back pain, osteoarthritis pain, post-surgical pain and cancer pain. Oxymorphone ER is currently under review by the FDA for the treatment of moderate to severe chronic pain. The safety and efficacy of oxymorphone ER have not been established by the FDA.
Oxymorphone ER is a Schedule II controlled substance.
Respiratory depression is the chief hazard from all opioid agonists including oxymorphone preparations. Respiratory depression is a particular potential problem in elderly or debilitated patients as well as in those suffering from conditions accompanied by hypoxia or hypercapnia when even moderate therapeutic doses may dangerously decrease pulmonary ventilation. The most common adverse reactions reported by patients treated with oxymorphone ER during clinical trials were nausea, constipation, dizziness, pruritus (itching), vomiting, somnolence, increased sweating, sedation and headache.
Penwest Pharmaceuticals
Penwest develops pharmaceutical products based on innovative oral drug delivery technologies. We are focusing our development efforts principally on products that address diseases of the central nervous system. The foundation of our technology platform is TIMERx(R), an extended release delivery system that is adaptable to soluble and insoluble drugs and that is flexible for a variety of controlled release profiles. We have also developed two additional oral drug delivery systems, Geminex(R) and SyncroDose(TM). Geminex is a dual release rate drug delivery system that is designed to provide independent release of different active ingredients contained in a drug, and SyncroDose is a drug delivery system that is designed to release the active ingredient of a drug at the desired site and time in the digestive tract.
The matters discussed herein contain forward-looking statements that involve risks and uncertainties, which may cause Penwest's actual results in future periods to be materially different from any future performance suggested herein. For this purpose, any statements contained herein that are not statements of historical fact may be deemed to be forward-looking statements. Without limiting the foregoing, the words, "believes," "anticipates," "plans," "expects," "intends," "potential," and similar expressions are intended to identify forward-looking statements. Important factors that could cause results to differ materially include; dependence on collaborators such as Endo Pharmaceuticals to, among other things, sell products for which the Company receives royalties, file for regulatory approvals, and/or to advance clinical development and commercialization of products; regulatory risks relating to drugs in development such as oxymorphone ER, including the timing and outcome of regulatory action; uncertainty of success of collaborations; the timing of clinical trials and whether the results of clinical trials will be indicative of results obtained in future trials, will warrant further clinical trials, warrant submission of an application for regulatory approval of, or the regulatory approval of, the product that is the subject of the trial; actual and potential competition; the need for capital; and other risks as set forth under the caption Certain Factors That May Affect Future Results in Penwest's Quarterly Report on Form 10-Q filed with the Securities and Exchange Commission on November 9, 2005, which risk factors are incorporated herein by reference. Penwest disclaims any intention or obligation to update any forward-looking statements.
CONTACT: Investors - Diane D'Alessandro of Penwest Pharmaceuticals Co., +1-203-796-3706, 1-877-736-9378; or Media - Caroline Gentile or Jim Fingeroth, both of Kekst and Company, +1-212-521-4800
Web site: http://www.penw.com/
COPYRIGHT 2005 PR Newswire Association LLC
COPYRIGHT 2005 Gale Group