More than 900,000 cases of nonmelanoma skin cancer are estimated to occur annually.[1] Basal cell carcinoma is the most common of these lesions. In the general population, the estimated lifetime risk of developing this malignancy is 28 to 33 percent.[1] Although basal cell carcinoma can occur in younger persons, it usually develops after the age of 50 years.[2] Up to 85 percent of basal cell carcinomas are found in the head and neck region 3 but the lesions occasionally occur in unusual and routinely photo-protected locations.[4-9]
Solar radiation exposure is the most important etiologic factor in the development of basal cell carcinoma.[10,11] Sun exposure from infancy to the age of 19 years may increase the risk of basal cell carcinoma in adulthood.[11,12] The risk factors for basal cell carcinoma are listed in Table 1.[10-12]
Information from references 10, 11 and 12.
Illustrative Case
A 69-year-old white man with a history of basal cell carcinoma of the head and neck presented for an annual skin examination. The examination revealed generalized evidence of long-term sun exposure. In addition, a firm, reddish-pink nodule with central erosion was found in his right axilla (Figure 1).
[Figure 1 ILLUSTRATION OMITTED]
Histopathologic examination of a shave biopsy specimen identified the lesion as a basal cell carcinoma. Curettage and desiccation were performed. The patient was instructed to use a sunscreen, to wear protective clothing and to perform periodic self-examinations of his skin. The importance of routine full cutaneous and mucocutaneous examinations performed by a physician was also emphasized.
Clinical Features
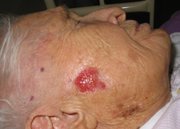
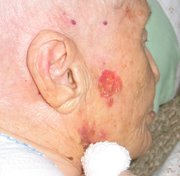
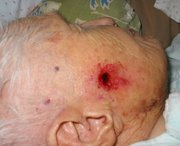
The recognized clinical types of basal cell carcinoma include nodular, cystic, superficial and sclerosing (morpheaform) lesions. The nodular type is the most common variant. Characteristically, this type of basal cell carcinoma has a pearly white to translucent skin-colored papule or nodule, usually with a rolled border, a central depression and an overlying telangiectatic vessel. The sclerosing basal cell carcinoma, which is the most aggressive variant, is an infiltrative, yellowish plaque-appearing lesion. Basal cell carcinomas are usually skin-colored, but they may be pigmented, particularly in dark-skinned persons. In such cases, the lesions may be confused with melanomas.[13]
While basal cell carcinomas usually occur in the head and neck region, they can also develop in unusual body sites. Relatively photo-protected areas in which basal cell carcinomas have been reported include the axilla, groin, penis, scrotum, vulva, breast, nipple, palm, foot, nail and mouth.[4-9]
Of the estimated 750,000 basal cell carcinomas that occur each year,[1] approximately 70,000 are located in covered anatomic sites.[14] In one series,[15] 72.7 percent of basal cell carcinomas were detected visually with the patients fully dressed. The remaining 27.3 percent of the lesions were detected only after the patients had removed all of their clothing and had undergone a total skin examination.
Etiology
While the relationship between solar radiation or artificial ultraviolet radiation and the development of basal cell carcinomas is well established, the occurrence of these lesions in unexposed areas suggests the existence of less recognized etiologic factors. These factors may include genetic predisposition, the systemic effect of ultraviolet radiation in decreasing cell-mediated immune system tumor surveillance,[8] trauma, chronic ulcers, burn scars, immunosuppression and genodermatoses such as xeroderma pigmentosum, albinism and the basal cell nevus syndrome.[10]
The basal cell nevus syndrome is an autosomal dominant syndrome that is caused by a mutation in a tumor suppressor gene localized to chromosome 9q.[16] The syndrome is characterized by multiple basal cell carcinomas, epidermal cysts, odontogenic keratocysts of the jaw, palmar and plantar pitting, calcified dural folds, neoplastic growths (medulloblastomas), hamartomatous growths (ovarian fibromas) and skeletal abnormalities (bifid ribs and frontal bossing).[17] In patients with this syndrome, basal cell carcinomas most often occur on the face and torso. The lesions appear at an early age and proliferate in puberty.[17]
Studies have shown that patients with a first basal cell carcinoma are at increased risk for developing a second basal cell carcinoma.[18] One study[19] found that another malignancy of this type developed in up to 45.2 percent of patients with basal cell carcinoma. The risk of a second basal cell carcinoma is greatest in the first year after treatment of an initial lesion.[19] The mean recurrence rate at five or more years after treatment of the initial malignancy is 18 percent.[20]
Compared with the general population, patients with a history of basal cell or squamous cell carcinoma are at increased risk for melanoma.[14,21] This malignancy currently occurs in one of every 87 persons in the United States.[22]
Periodic Skin Examination
For preventive purposes, physicians should perform complete cutaneous and mucocutaneous examinations, especially in patients with a history of skin cancer. The routine, full examination is inexpensive, painless and safe. Furthermore, it can be performed in a matter of minutes.
The purpose of the complete cutaneous and mucocutaneous examination is to decrease the morbidity and mortality of skin cancer through early detection and treatment.[23] In one study,[24] 162 (15 percent) of 1,106 patients who agreed to be screened for skin cancer were found to have other important incidental findings, such as neurofibromatosis and Kaposi's sarcoma. Most studies suggest that full examinations need to be performed periodically.[25] In Australia, screening every six months is recommended for patients with a history of skin cancer.[26,27]
The American Academy of Dermatology (AAD) and the Centers for Disease Control and Prevention (CDC) encourage regular self-examinations to help in the early detection of skin cancer.[28] Although randomized, controlled trials are needed to determine the efficacy of self-examinations, one case-controlled study[29] reported that skin self-examinations led to a 63 percent reduction in melanoma mortality.
Treatment
The treatment of basal cell carcinoma must be tailored to the patient, to the size and location of the lesion and to the familiarity of the surgeon with particular techniques. Standard treatments include cryosurgery, radiotherapy, curettage and desiccation, surgical excision and Mohs micrographic surgery. One study[20] reported the following five-year recurrence rates for treated basal cell carcinomas: 1.0 percent after Mohs micrographic surgery; 7.5 percent after cryosurgery; 7.7 percent after curettage and desiccation; 8.7 percent after radiotherapy; and 10.1 percent after surgical excision.
Curettage and desiccation, radiotherapy and cryosurgery are used to treat low-risk tumors. An example might be a small primary basal cell carcinoma of nonaggressive histologic subtype that is located in a low-risk body site (extremity or torso).
Nonaggressive histologic subtypes of basal cell carcinoma are nodular and superficial, while aggressive histologic subtypes are morpheaform (infiltrative or sclerosing), micronodular and metatypical.[30] Surgical excision and Mohs micrographic surgery are used to treat more aggressive tumors. The five-year recurrence rate for surgical excision is higher than the recurrence rates for curettage and desiccation, radiotherapy and cryosurgery. The probable explanation is that surgical excision is used on more aggressive tumors, which are inherently more likely to recur.
Mohs micrographic surgery, which involves referral to a dermatologic surgeon, is used to treat high-risk tumors. Indications for this technique include the following: a recurrent nonmelanoma skin cancer; a tumor of aggressive histopathology (infiltrative); a large tumor (more than 2 cm in diameter and located on the face, more than 4 cm in diameter in other locations); a tumor in a high-risk location (e.g., central face); a tumor for which conservation of tissue or vital structures is necessary; and a radiation-induced tumor.[30,31]
In one five-year study[32] 1,455 (40.8 percent) of 3,560 basal cell carcinomas diagnosed were treated with Mohs micrographic surgery. This surgical technique involves horizontal or tangential excision of the lesion with immediate microscopic examination of the excised specimen.[32] If the margins of the tumor are positive, the procedure is repeated until all pathologic tissue has been removed. Compared with routine excision, Mohs micrographic surgery spares more normal tissue. Thus, more healthy tissue is available for reconstruction of the defect. If indicated, Mohs micrographic surgery is the most effective treatment for basal cell carcinoma. Of the standard treatment choices, it has the lowest recurrence rate.
Fortunately, basal cell carcinomas rarely metastasize.[33] Reported sites of metastases include the lymph nodes, lungs, bone, skin and liver.[33] Most of the morbidity associated with this malignancy, including disfigurement and loss of vital function (e.g., vision), is caused by local extension of a neglected tumor. The most important variable contributing to increased morbidity and mortality is the size of the primary tumor, and the most important factor contributing to the size of the tumor is patient neglect.[34]
Final Comment
Overall, basal cell carcinoma is easily treated when it is detected early. Thus, all skin sites must be examined carefully for both routine and atypical presentations of this malignancy.
In an effort to reduce the incidence and mortality of skin cancer, the AAD and CDC have made several recommendations.[28] First, patients should be informed that their physician should examine any sore that does not heal or any skin growth that is enlarging. Second, the need to use sunscreens with a sun protection factor (SPF) of 15 or more should be stressed to all patients. In addition, patients should be encouraged to wear sun-protective clothing and should be urged to avoid excessive sun exposure (especially from 10 a.m. to 4 p.m.). Patients should also avoid using artificial tanning booths. Finally, patients should be instructed to perform skin self-examinations and to routinely see their physician for full cutaneous and mucocutaneous examinations.
The opinions and assertions contained herein are the private views of the authors and are not to be construed as official or as representing the views of the Department of Defense, the Department of the Army or the Department of the Air Force.
REFERENCES
[1.] Miller DL, Weinstock MA. Nonmelanoma skin cancer in the United States: incidence. J Am Acad Dermatol 1994;30(5 Pt 1):774-8.
[2.] Cox NH. Basal cell carcinoma in young adults. Br J Dermatol 1992-127:26-9.
[3.] Reizner GT, Chuang T, Elpern DJ, Stone JL, Farmer ER. Basal cell carcinoma in Kauai, Hawaii: the highest documented incidence in the United States. J Am Acad Dermatol 1993;29(2 Pt 1):184-9.
[4.] Robins F, Rabinovitz HS, Rigel D. Basal-cell carcinomas on covered or unusual sites of the body. J Dermatol Surg Oncol 1981;7:803-6.
[5.] Johnson TM, Tschen J, Ho C, Lowe L, Nelson BR. Unusual basal cell carcinomas. Cutis 1994;54:85-92.
[6.] Rahbari H, Mehregan AH. Basal cell epitheliomas in usual and unusual sites. J Cutan Pathol 1979;6: 425-31.
[7.] Piro GF, Collier DU. Basal cell carcinoma of the palm. J Am Acad Dermatol 1995;33(5 Pt 1):823-4.
[8.] Goldminz D, Scott G, Klaus S. Penile basal cell carcinoma. Report of a case and review of the literature. J Am Acad Dermatol 1989;20:1094-7.
[9.] Nahass GT, Blauvelt A, Leonardi CL, Penneys NS. Basal cell carcinoma of the scrotum. Report of three cases and review of the literature. J Am Acad Dermatol 1992;26:574-8.
[10.] Preston DS, Stern IRS. Nonmelanoma cancers of the skin. N Engl J Med 1992;327:1649-62.
[11.] Gallagher RR Hill GB, Bajdik CD, Fincham S, Coldman AJ, McLean DI, et al. Sunlight exposure, pigmentary factors, and risk of nonmelanocytic skin cancer. I. Basal cell carcinoma. Arch Dermatol 1995;131:157-63.
[12.] Stern RS, Weinstein MC, Baker SG. Risk reduction for nonmelanoma skin cancer with childhood sunscreen use. Arch Dermatol 1986;122:537-45.
[13.] Bigler C, Feldman J, Hall E, Padilla RS. Pigmented basal cell carcinoma in Hispanics. J Am Acad Dermatol 1996;34(5 Pt 1):751-2.
[14.] Marghoob AA, Slade J, Salopek TG, Kopf AW, Bart IRS, Rigel IDS. Basal cell and squamous cell carcinomas are important risk factors for cutaneous malignant melanoma. Screening implications. Cancer 19951-75:707-14.
[15.] Lee G, Massa MC, Welykyj S, Choo J, Greaney V Yield from total skin examination and effectiveness of skin cancer awareness program. Findings in 874 new dermatology patients. Cancer 1991;67:202-5.
[16.] Compton JG, Goldstein AM, Turner M, Bale AE, Kearns KS, McBride OW, et al. Fine mapping of the locus for nevoid basal cell carcinoma syndrome on chromosome 9q. J Invest Dermatol 1994; 103:178-81.
[17.] Gorlin RJ. Nevoid basal cell carcinoma syndrome. Dermatol Clin 1995;13:113-25.
[18.] Karagas MR, Stukel TA, Greenberg ER, Baron JA, Mott LA, Stern IRS. Risk of subsequent basal cell carcinoma and squamous cell carcinoma of the skin among patients with prior skin cancer. Skin Cancer Prevention Study Group. JAMA 1992-1 267:3305-10.
[19.] Marghoob A, Kopf AW, Bart IRS, Sanfilippo L, Silverman MK, Lee P, et al. Risk of another basal cell carcinoma developing after treatment of a basal cell carcinoma. J Am Acad Dermatol 1993;28:22-8.
[20.] Rowe DE, Carroll RJ, Day CL Jr. Long-term recurrence rates in previously untreated (primary) basal cell carcinoma: implications for patient follow-up. J Dermatol Surg Oncol 1989; 15:315-28.
[21.] Evans RD, Kopf AW, Lew RA, Rigel IDS, Bart RS, Friedman RJ, et al. Risk factors for the development of malignant melanoma--I: Review of case-control studies. J Dermatol Surg Oncol 1988;14:393-408.
[22.] Rigel DS, Friedman RJ, Kopf AW. The incidence of malignant melanoma in the United States: issues as we approach the 21st century. J Am Acad Dermatol 1996;34(5 Pt 1):839-47.
[23.] Shenefelt PD. Skin cancer prevention and screening. Prim Care 1992;19:557-74.
[24.] Lookingbill DP. Yield from a complete skin examination. Findings in 1157 new dermatology patients. J Am Acad Dermatol 1988;18(1 Pt 1):31-7.
[25.] Friedman RJ, Rigel IDS, Silverman MK, Kopf AW, Vossaert KA. Malignant melanoma in the 1990s: the continued importance of early detection and the role of the physician examination and self-examination of the skin. CA Cancer J Clin 1991; 41:201-26.
[26.] Girgis A, Campbell EM, Redman S, Sanson-Fisher RW. Screening for melanoma: a community survey of prevalence and predictors. Med J Aust 1991;154:338-43.
[27.] Fleming WB. The cancer-related health check-up. A guide for medical practitioners. Med J Aust 1985;143(6 Suppl):S33-40.
[28.] Goldsmith LA, Koh HK, Bewerse BA, Reilley B, Wyatt SW, Bergfeld WF, et al. Full proceedings from the National Conference to Develop a National Skin Cancer Agenda. American Academy of Dermatology and Centers for Disease Control and Prevention, Washington, D.C., April 8-10, 1995. J Am Acad Dermatol 1996;35(5 Pt 1):748-56.
[29.] Berwick M, Begg CB, Fine JA, Roush GC, Barnhill RL. Screening for cutaneous melanoma by skin self-examination. J Natl Cancer Inst 19961-88:17-23.
[30.] Randle HW. Basal cell carcinoma. Identification and treatment of the high-risk patient. Dermatol Surg 1996;22:255-61.
[31.] Welch ML, Anderson LL, Grabski WJ. How many nonmelanoma skin cancers require Mohs micrographic surgery? Dermatol Surg 1996-122:711-3.
[32.] Bennett RG. Current concepts in Mohs micrographic surgery. Dermatol Clin 1991;9:777-88.
[33.] Miller SJ. Biology of basal cell carcinoma (Part I). J Am Acad Dermatol 1991;24:1-13.
[34.] Sahl WJ. Basal cell carcinoma: influence of tumor size on mortality and morbidity. Int J Dermatol 1995; 34:319-21,
JOSEPH C. ENGLISH III, M.D., is currently a staff dermatologist at Evans Army Community Hospital, Ft. Carson, Colo. Previously he was chief resident in the Department of Dermatology at Brooke Army Medical Center/Wilford Hall Air Force Medical Center, San Antonio. Dr. English graduated from the Pennsylvania State University College of Medicine, Hershey, and completed a categorical medicine internship and a dermatology residency at Brooke Army Medical Center.
DANIEL R. CANCHOLA, M.D., is currently serving an internship in family practice at John Peterson Smith Hospital, Ft. Worth, Tex. Dr. Canchola received his medical degree from the University of Texas Health Science Center, San Antonio.
ERIC M. FINLEY, M.D., is currently a dermatology surgeon at Keesler Air Force Base, Biloxi, Miss. Previously he served in the Department of Dermatology at Brooke Army Medical Center/Wilford Hall Air Force Medical Center. Dr. Finley graduated from the University of Missouri-Columbia Medical School. He completed a dermatology residency at Rush-Presbyterian-St. John's Medical Center, Chicago, and a surgical fellowship at the Ochsner Clinic, New Orleans.
Address correspondence to Joseph C. English Ill, M.D., 1940 Avalon Ct., Colorado Springs, CO 80919. Reprints are not available from the authors.
COPYRIGHT 1998 American Academy of Family Physicians
COPYRIGHT 2000 Gale Group