A Case Report
The diagnostic approach, clinical evolution, and treatment of a patient with primary pulmonary lymphangioleiomyomatosis are reported. This patient presented a restrictive respiratory syndrome resistant to conventional glucocorticoid therapy. The diagnosis, based on clinical and histologic examinations, was confirmed by immunohistochemical localization of one of the desmins, the smooth muscle cell actin, and HMB45 antigen. The patient received treatment with an anti-estrogenic agent (tamoxifen citrate) and high doses of medroxyprogesterone acetate, an antigonadotropic progestin. Respiratory function improved rapidly with clinical relief. (CHEST 1999; 115:276-279)
Key words: anti-estrogen; progestin; pulmonary lymphangioleiomyomatosis
Abbreviation: LLM = lymphangioleiomyomatosis
Lymphangioleiomyomatosis (LLM) is a rare pathologic finding, for fewer than 100 cases have been described. It consists of a smooth muscle cell proliferation into the perilymphatic pulmonary interstitium. LLM occurs primarily in women of childbearing age. One third of the cases arise in women less than 20 years of age.[1-6]
LLM also may appear as a forme fruste of tuberous sclerosis (Bourneville's disease)[5] or may belong to the syndrome described by Carney et al,[4] which includes the triad of pulmonary chondroma, extra-adrenal paraganglioma, and gastric leiomyosarcoma. The past histories of women with LLM frequently include hysterectomy or myomectomy,[3] and a premenopausal state[5] associated with tobacco use.[3] Finally, benign leiomyomas represent fewer than 2% of the benign tumors of the lower respiratory tract.[6]
The onset of the disease is paucisymptomatic. Although the diagnosis is most often evoked by a routine chest x-ray film, a histologic examination of a surgically or transbronchially[1,3] obtained pulmonary biopsy specimen is essential to confirm it. LLM must be differentiated from other pathologic states, including smooth muscle cell hyperplasia. The main differential diagnosis is benign metastasizing leiomyoma associated with a uterine leiomyoma. This last entity responds favorably to corticosteroids in contrast to LLM which is resistant to glucocorticoids but is favorably influenced by oophorectomy, progestins, anti-estrogenic agents, or luteinizing hormone-releasing hormone agonists.[2-4]
The histologic, clinical, and therapeutic characteristics of an LLM case successfully treated with an anti-estrogenic agent and a progestin are described.
CASE REPORT
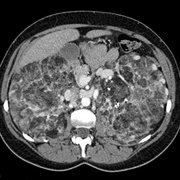
In May 1987, a 36-year-old patient was admitted to the emergency department with a dry cough, a morning fever, and diffuse sweating. Her general condition was satisfactory. Clinical examination detected a temperature of 36.5 [degrees] C; heart rate, 80 beats per minute; BP, 135/70 mm Hg. She complained of profound asthenia and considerable anxiety. Digital clubbing was noted as well as rales on the right side of the chest and severe dyspnea (grade III, Sadoul scale[7] equivalent to grade II [MRC.sup.8]). The chest x-ray film revealed pneumonia of the median right lobe (anterior segment), bronchopneumonia in the lower lobes of both right and left lungs (Fig 1). Clinical biology was characterized by an acute inflammatory syndrome. Antibiotics (doxycyline, 200 mg/d) and anxiolytics (alprazolam) were prescribed.
[Figure 1 ILLUSTRATION OMITTED]
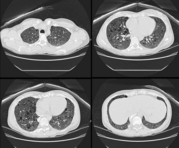
Six weeks later, the patient was readmitted with increased dyspnea. The chest x-ray film was abnormal, suggesting a possible interstitial alveolar pneumopathy and fibrous sequelae of the median right lobe pneumonia. Bronchofibroscopy was performed with BAL. A liver biopsy was obtained in order to exclude sarcoidosis. No Mycoplasma organisms were detected in the BAL fluid. Pulmonary biopsy specimens suggested the possibility of idiopathic pulmonary fibrosis or LLM or interstitial desquamative pneumopathy (Fig 2). Treatment with glucocorticoids (methylprednisolone, 16 mg/d) was initiated. No attenuation of the symptoms was obtained after 4 weeks, at which time the dosage was adjusted to 8 mg/d; again, there was no clinical improvement.
[Figure 2 ILLUSTRATION OMITTED]
In the meantime, electron microscopical examination of the lung biopsy specimen indicated the presence of ectopic smooth muscle cells infiltrating the alveolar septae. These cells were immunohistochemically labeled by anti-smooth muscle cell actin and anti-vimentin antibodies; they did not contain factor VIII, HMB45, protein S-100, or the estradiol receptor, but some did express the progesterone receptor. A diagnosis of incipient LLM or benign metastasizing leiomyoma was then advanced. The clinical gynecologic examinations and pelvic echography excluded the presence of uterine fibromas.
Oral contraception (30 [micro]g of ethynyl estradiol and 150 [micro]g of desogestrel) was stopped due to the possible effect of sex steroids on the proliferation of smooth muscle cells. Glucocorticosteroid therapy also was discontinued. A regimen specifically designed to suppress the proliferation of the smooth muscle cells was then established: daily administration of an anti-estrogenic agent (tamoxifen citrate, 20 mg/d) in combination with high doses of an antigonadotropic progestin (medroxyprogesterone acetate), 300 mg intramuscularly every 15 days for 2 months and then 500 mg/mo intramuscularly.
The objective parameters of the pulmonary function improved rapidly (Table 1) as well as her general health status. The classic side effects of this type of treatment were noted: artificial menopause and a weight gain of 15 kg over a period of 8 months. Presently, the patient remains asymptomatic with stable cardiorespiratory functions. After 2 years of receiving the initial regimen, medroxyprogesterone acetate was replaced with norethinderone acetate (10 mg/d) and tamoxifen therapy was maintained at the same dose. The gynecological and breast status have remained normal.
DISCUSSION
The diagnosis of LLM in this patient was suggested by the clinical symptoms and by the histologic examination (conventional histologic findings and immunohistochemical search for cytoskeletal proteins). The most specific criterion was the identification of ectopic smooth muscle cells by electron microscopy.[1,6] The tissue required for all pathologic examinations was obtained by surgical biopsy, although the amount required for all examinations could have been provided by transbronchial biopsy.
In this case, the study showed all smooth muscle cells consistently reacted with anti-smooth muscle actin, antivimentin, and desmin antibodies but were not recognized by anti-HMB45 or anti-protein S-100 antibodies.[1]
Bonetti et al[1] detected HMB45 in 2 of their 3 cases, thereby confirming the cellular heterogeneity of LLM proliferations. This heterogeneity also has been noted by immunohistochemistry and classic light and electron microscopic observations. It may reflect different degrees of differentiation of the same cell types or the coexistence of several cell types.
The presence of cytoplasmic progesterone receptors (but not estradiol receptors) in 10% of the cells suggested a possible benefit of high doses of the progestin medroxyprogesterone acetate. Uchida et al[9] described the existence of estradiol and progesterone receptors in smooth muscle cells in one case of benign metastasizing leiomyoma. They think that, even if normal lung tissue possesses hormone receptor, it is rational to consider that multiple leiomyomatous lesions in the lung should be diagnosed as benign metastasizing leiomyoma when they possess positive estrogen and progesterone receptors. The presence of such functional receptors might explain the efficacy of this type of treatment in the administration of a luteinizing hormone-releasing hormone agonist.[3,4]
Progestin therapy for LLM has been reported in at least 11 patients[2,5] It seems that most women have either improved or remained stable while taking medroxyprogesterone acetate whereas the condition of a few worsened despite the use of this drug or an luteinizing hormone-releasing hormone agonist. Tamoxifen therapy and oophorectomy were also recommended.[2,5]
The rationale for combining medroxyprogesterone acetate and tamoxifen, despite the absence of a demonstrable estradiol receptor, was that no correlation between the status of progesterone and estrogen receptors and the response to hormonal therapy has been observed.[5] It is also known that estrogen aggravated this condition.[2]
Ten years after the initial event, this woman remains asymptomatic, thus further eon firming the hypothesis that progestins are highly effective and should be prescribed as the first-line treatment. According to the meta-analysis conducted by Eliasson et al,[2] oophorectomy represents a valuable alternative.
Because LLM is a very rare disease, it is likely that the best way to resolve questions of optimal treatment and prognosis will be through multicenter trials. Adequate documentation of diagnosis, treatment, and pulmonary function is, however, essential for meta-analysis evaluation of treatment.
CONCLUSION
In one case of LLM, diagnosed on the basis of light and electron microscopic histologic and immunohistochemical findings that confirmed the presence of actin, desmin, and vimentin in the smooth muscle cell proliferation and detected progesterone receptors in some of the cells, dramatic clinical improvements of the subjective and objective respiratory function parameters were obtained by prescribing a regimen combining an anti-estrogenic agent (tamoxifen) and a progestin (medroxyprogesterone acetate).
REFERENCES
[1] Bonetti F, Chiodera PL, Pea M, et al. Transbronchial biopsy in lymphangiomyomatosis of the lung: HMB45 for diagnosis. Am J Surg Pathol 1993; 17:1092-1102
[2] Eliasson AH, Phillips YY, Tenholder MF. Treatment of lymphangioleiomyomatosis: a meta-analysis. Chest 1989; 196: 1352-1355
[3] Jacobson TZ, Rainey EJ, Turton CWG. Pulmonary benign metastasising leiomyoma: response to treatment with goserelin. Thorax 1995; 50:1225-1226
[4] Carney JA, Sheps SG, Go VLW, et al. The triad of gastric leiomyosarcoma, functioning extra-adrenal paraganglioma and pulmonary chondroma. N Engl J Med 1977; 296:1517-1518
[5] Taylor JR, Ryu J, Colby TV, et al. Lymphangioleiomyomatosis: clinical course in 32 patients. N Engl J Med 1990; 323:1254-1260
[6] White SH, Ibrahim NBN, Forrester-Wood CP, et al. Leiomyomas of the lower respiratory tract. Thorax 1985; 40:306-311
[7] Sadoul P. Evaluation du deficit fonctionnel respiratoire. Bull Eur Physiopath Resp 1983; 19:3-6
[8] Comstock GW, Tockman MS, Helsing KJ, et al. Standardized respiratory questionnaire: Comparison of the old with the new. Am Rev Respir Dis 1979; 119:45-53
[9] Uchida T, Tokumaru T, Kojima H, et al. A case of multiple leiomyomatous lesions of the lung: an analysis of flow cytometry and hormone receptors. Jap J Surg 1992; 22:265-268
(*)From the Laboratory of Biology (Mr. Denoo and Dr. Foidart), Laboratory of Anatomopathology (Dr. Hermans), and Department of Obstetrics and Gynecology (Dr. Foidart), University of Liege, Liege, Belgium, and the Department of Pneumology, Centre Hospitalier du Bois de l'Abbaye, Seraing, Belgium (Dr. Degives).
This work was supported by a grant from the Fonds de la Recherche Scientifique Medicale in Belgium (No. 3.4573.95).
Manuscript received December 10, 1997; revision accepted May 19, 1998.
Correspondence to: Professor Jean-Michel Foidart, Laboratory of Tumor and Developmental Biology, University of Liege, Pathology Tower, B23, Sart Tilman, B-4000 Liege, Belgium; e-mail: jmfoidart@ulg.ac.be
COPYRIGHT 1999 American College of Chest Physicians
COPYRIGHT 2000 Gale Group