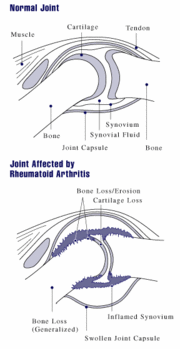
Rheumatoid arthritis (RA) is the most prevalent inflammatory arthritis in adults, affecting 1% to 2% overall, and 2% to 2.3% of the geriatric population. (1) RA is the most common inflammatory synovitis affecting older persons. Older adults with RA can be categorized into one of two groups: those with long-standing disease and those with new-onset agerelated disease.
Patients with long-standing disease generally do not pose a diagnostic challenge because they have been diagnosed for many years. They tend to have classic rheumatoid hand deformities consisting of ulnar deviation, swan neck deformities, or boutonniere deformities. They usually exhibit accumulated erosive changes on x-ray and are frequently rheumatoid factor (RF)-positive.
Often, patients with long-standing disease have already undergone joint replacement surgery. These patients pose a major therapeutic challenge however, because most have already been treated with multiple disease-modifying anti-rheumatic drugs (DMARDs), as well as long-term steroids and non-steroidal anti-inflammatory drugs (NSAIDs). Thus, therapeutic options may be limited, response to new agents less likely, and considerable toxicity to the prior agents may have occurred.
Ten percent to 20% of RA patients may present after age 60 and comprise the group of patients with de novo onset of disease. (2) Elderly-onset RA includes a greater proportion of men, more often has an insidious onset, and is less likely to be complicated by extra-articular disease manifestations, such as rheumatoid nodules on the forearms. It is more frequently large joint-predominant (particularly the shoulder girdle), and is often associated with systemic complaints (eg, fatigue, malaise, weight loss, generalized stiffness, depression) that sometimes precede the onset of articular disease. Laboratory testing may reveal a striking inflammatory response (eg, elevated erythrocyte sedimentation rate [ESR]), and there is a lower incidence of RF-positivity than in those with long-standing disease. (3)
Patients with elderly-onset disease pose a diagnostic challenge to geriatricians because of the seeming lack of specificity about their complaints. Most patients present with constitutional symptoms, such as fatigue and loss of appetite. In addition, they describe diffuse aching pain, especially in the upper extremities, in association with an overwhelming sense of generalized morning stiffness. Physical exam is notable for marked synovitis of the hands, consisting of symmetrical, proximal joint synovitis (wrist, MCP [metacarpal phalangeal], PIP [proximal interphalangeal]), and dorsal soft tissue swelling, which can be severe. Classic rheumatoid hand deformities are absent. Shoulder synovitis with soft tissue swelling and pain on range of motion is also seen. Laboratory parameters usually demonstrate an elevated sedimentation rate and mild anemia associated with chronic disease. RF is usually absent. X-rays generally show a lack of erosive changes and reveal only soft tissue swelling and periarticular osteoporosis. (4)
RA and aging
RA is an autoimmune and inflammatory disease in which B- and T-cell dysfunction can be identified, both in the joint and systemically, some of which may precede clinical disease. These abnormalities may mimic and accelerate changes that are known to accompany normal immunosenescence. (5) Many studies (6) have focused on the impact of chronic inflammation on endocrine, neurologic, and musculoskeletal aging, as well as the increased morbidity and mortality in patients with RA resulting from comorbid conditions such as infection, osteoporosis, and cardiovascular disease, all of which are prevalent in geriatric patients. These considerations heighten appreciation for the need to treat RA early and more aggressively to reduce comorbid disease and improve long-term outcomes.
Differential diagnosis
Differential diagnostic possibilities for RA include polymyalgia rheumatica (PMR), the pseudo-rheumatoid pattern of pseudogout, gout, reflex sympathetic dystrophy, and osteoarthritis (OA).
PMR can be excluded with the presence of objective upper extremity synovitis, which is not characteristic of this disease.
Both gout and pseudogout, unlike RA, present with episodic attacks with some resolution of symptoms and signs without treatment. Synovial fluid analysis can be used to determine the presence of intracellular crystals, which are not seen in the synovial fluid of patients with RA. Rheumatoid synovitis, by contrast, is continuous and persistent. The persistent nature of RA synovitis for at least 6 weeks is a central diagnostic criteria for the diagnosis of RA (table 1). (7)
Reflex sympathetic dystrophy is rarely simultaneously bilateral, and although marked upper extremity swelling may be present, painful dysesthesias in between joint areas occur, which are not seen in RA.
Osteoarthritis involving the hands affects the DIPs (distal interphalangeal) and PIPs. The hypertrophic bone found in osteoarthritis results in enlarged bony nodules of these joint areas known as Heberden's nodes and Bouchard's nodes, respectively. MCP and wrist synovitis are not seen in osteoarthritis, but are commonly seen in patients with rheumatoid arthritis. Therefore, although pinch strength may be impaired in patients with osteoarthritis, grip strength, which reflects proximal hand joint integrity, will be preserved. Generalized diffuse morning stiffness, constitutional symptoms, and elevated sedimentation rates are not seen in osteoarthritis. Fluid obtained from an osteoarthritic joint is typically bland rather than inflammatory, and x-rays predominantly show productive rather than erosive changes.
A variant of the spectrum of findings presented by elderly-onset RA and PMR is the syndrome of remitting seronegative symmetrical synovitis with pitting edema (RS3PE), in which patients present with more sudden onset of synovitis prominently affecting the hands and wrists, and often complicated by flexor tenosynovitis. This variant form of inflammatory arthritis is particularly sensitive to low-dose corticosteroid therapy. (3)
Treatment
It is important to distinguish older patients with RA from those with osteoarthritis and other differential diagnostic possibilities, because the symptoms, although debilitating, can either improve remarkably or remit with pharmacologic treatment. It is also important to consider activity of disease when weighing treatment options, because in some instances, long-standing RA may "burn out," yielding anatomic and functional limitations mainly due to superimposed osteoarthritis. Functional status and independence can be maintained in older adults with RA if the disease is recognized and treatment strategies are carefully considered.
The initial strategy for treating older patients with RA is to palliate the pain caused by the inflammatory synovitis. Although salicylates and NSAIDs are first-line treatments in younger RA patients, the high daily doses that are required to provide pain relief cause considerable toxicity in older patients, including renal insufficiency, hypertension, edema, exacerbation of congestive heart failure, and mental status changes. (8) Although the risk of bleeding is markedly reduced with the cyclooxygenase-2 (COX-2) inhibitors, all of the above mentioned side effects are still seen with these agents. Many older RA patients, especially those with de novo onset, are responsive to treatment with low-dose steroids, such as prednisone, 5 mg [pounds sterling]/d. (9) The rapid dramatic improvement of symptoms experienced with such a low dose shifts the risk-to-benefit ratio in favor of using steroids as first-line agents in all older RA patients.
For older patients who have the pattern of disease seen in younger patients, such as those with long-standing erosive disease or persistent severe synovitis despite low-dose steroids, or those with a higher steroid requirement in whom steroid tapering produces a flair, treatment with DMARDs should be initiated (table 2). (10,11) Patients who require treatment with DMARDs should be referred to a rheumatologist due to the potential toxicity of these agents. Nevertheless, two DMARDs have low toxicity profiles and should be emphasized: hydroxychloroquine sulfate (Plaquenil) and sulfasalazine (Azulfidine). Meta-analyses have shown these agents to cause disease remittance, although their effects occur slowly. (12) Neither agent requires extensive monitoring, and both can be safely used in frail older patients with multiple medical problems or compliance issues related to dementia. For these reasons, these agents should have a prominent role in the management of RA in older adults. In addition, supportive devices, physical and occupational therapy, as well as joint replacement surgery are of great benefit to older RA patients.
The role of biologic agents, such as tumor necrosis factor alpha (TNFa) antagonists, has yet to be precisely defined, but is problematic in the older RA patient because adverse effects include severe infections, especially tuberculosis, as well as sepsis, congestive heart failure, and malignancies, particularly lymphomas. (11,13,14) Because many older RA patients are PPD positive, have known malignancies in remission (such as breast, prostate, or melanoma), may have underlying cardiac dysfunction as well as chronic sources of infections (eg, venous stasis ulcers, recurrent urinary tract infections), these agents are potentially more hazardous in this population than in younger RA patients who have fewer comorbidities, and are therefore less commonly used.
Summary
RA commonly affects older adults and causes debilitating symptoms. If properly diagnosed and treated, RA can be dramatically controlled, with restoration of quality of life and prevention of functional decline.
Take Home
* The spectrum of RA in older adults may overlap with crystal arthropathy (gout and pseudogout) and PMR.
* Approximately 50% of patients with elderly-onset RA have a distinct form of disease that may be highly inflammatory, lack standard criteria for diagnosis, and be progressive in nature.
* Because osteoarthritis is common in older adults, it may coexist with RA, or be a consequence of longstanding inflammatory disease.
* NSAID toxicity and disease responsiveness provide a definite role for low-dose corticosteroids in management.
* Disease-modifying agents, such as hydroxychloroquine and sulfasalazine, are generally well-tolerated.
* Comorbid conditions, such as infection, osteoporosis, and cardiovascular disease, can be both causes and effects. They significantly affect morbidity and mortality, and should influence decisions about using newer therapies, such as the TNFa antagonists.
CME Geriatrics
Exam
Detach or photocopy this page, place an X in the boxes that correspond to your answers, fill in your name and address, and mail (see address below). Answers must be received by April 1, 2005. A score of at least 80% must be earned to receive CME credit.
Make check for $15 payable to The Page and William Black Post-Graduate School and mail it with this exam to Rae Ann Houghton, Geriatrics, 7500 Old Oak Blvd., Cleveland, Ohio 44130. When submitting more than one exam, attach a separate check for $15 to each exam. Documentation of earned credit and the correct answers will be mailed to you. Allow up to 12 weeks for notification.
Accreditation. This activity has been planned and implemented in accordance with the Essentials and Standards of the Accreditation Council for Continuing Medical Education (ACCME) through the sponsorship of Mount Sinai School of Medicine. Mount Sinai School of Medicine is accredited by ACCME to provide continuing medical education for physicians. Mount Sinai School of Medicine designates this continuing medical education activity for a maximum of 1 credit in category 1 toward the AMA Physician's Recognition Award. Each physician should claim only those hours that he/she spent in the educational activity.
Faculty Disclosure. It is the policy of Mount Sinai School of Medicine to ensure fair balance, independence, objectivity, and scientific rigor in all its sponsored programs. All faculty participating in sponsored programs are expected to disclose to the audience any real or apparent conflict-of-interest related to the content of their presentation, and any discussions of unlabeled or investigational use of any commercial product or device not yet approved in the United States.
References
(1.) Rasch, EK, Hirsch R, Paulose-Ram R, Hochberg MC. Prevalence of rheumatoid arthritis in persons 60 years of age and older in the United States: Effect of different methods of case classification. Arthritis Rheum 2003; 48(4):917-26.
(2.) Ehrlich GE, Katz WA, Cohen SH. Rheumatoid arthritis in the aged. Geriatrics 1970; 25(2):103-13.
(3.) Yazici Y, Paget SA. Elderly-onset rheumatoid arthritis. Rheum Dis Clin North Am 2000; 26(3): 517-26.
(4.) Kerr LD. Inflammatory arthritis in the elderly. Mt Sinai J Med 2003; 70(1):23-6.
(5.) Goronzy JJ, Weyand CM. Aging, autoimmunity and arthritis: T-cell senescence and contraction of T-cell repertoire diversity-catalysts of autoimmunity and chronic inflammation. Arthritis Res Ther 2003; 5(5):225-34.
(6.) Straub RH, Scholmerich J, Cutolo M. The multiple facets of premature aging in rheumatoid arthritis. Arthritis Rheum 2003; 48(10):2713-21.
(7.) Arnett FC. Revised criteria for the classification of rheumatoid arthritis. Bull Rheum Dis 1989; 38(5):1-6.
(8.) Griffin MR, Ray WA, Schaffner W. Nonsteroidal anti-inflammatory drug use and death from peptic ulcer in elderly persons. Ann Intern Med 1998; 109(5):359-63.
(9.) Kirwan JR. The effect of glucocorticoids on joint destruction in rheumatoid arthritis. The Arthritis and Rheumatism Council Low-Dose Glucocorticoid Study Group. N Engl J Med 1995; 333(3):142-6.
(10.) van der Heide A, Jacobs JW, Bijlsma JW, et al. The effectiveness of early treatment with "second-line" antirheumatic drugs. A randomized, controlled trial. Ann Intern Med 1996; 124(8):699-707.
(11.) O'Dell JR. Therapeutic strategies for rheumatoid arthritis. N Engl J Med 2004; 350(25):2591-602.
(12.) O'Dell JR, Haire CE, Erikson N, et al. Treatment of rheumatoid arthritis with methotrexate alone, sulfasalazine and hydroxychloroquine, or a combination of all three medications. N Engl J Med 1996; 334(20):1287-91.
(13.) Moreland LW, Baumgartner SW, Schiff MH, et al. Treatment of rheumatoid arthritis with a recombinant human tumor necrosis factor receptor (p75)-Fc fusion protein. N Engl J Med 1997; 337(3):141-7.
(14.) Maini RN, Breedveld FC, Kalden JR, et al. Therapeutic efficacy of multiple intravenous infusions of anti-tumor necrosis factor alpha monoclonal antibody combined with low-dose weekly methotrexate in rheumatoid arthritis. Arthritis Rheum 1998; 41(9):1552-63.
Dr. Kerr is associate professor of medicine and geriatrics, Mt. Sinai Medical Center, New York, New York. Disclosure: The author has no real or apparent conflict of interest due to affiliation with any company or manufacturer whose products or devices are mentioned in this article.
COPYRIGHT 2004 Advanstar Communications, Inc.
COPYRIGHT 2005 Gale Group