KEYWORDS: Strongyloidiasis; Seroepidemiology; Indirect Immunofluorescence Antibody Test; Filter paper; Strongyloides ratti.
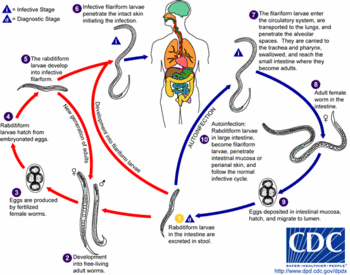
Strongyloides stercoralis (Bavay, 1876) infection is ranked in 5th place among the helminthiasis in the world with an estimated 35 million infected individuals, mostly living in tropical and semitropical countries3. The frequency of nonsymptomatic or disseminated forms has become a serious public health concern due to the lack of reliable tools for the diagnosis of this disease5,7.
The development of immunological tests for the diagnosis of strongyloidiasis can be helpful in epidemiological and clinical evaluations. Studies have been done trying to standardize a serological test for the diagnosis of this helminthiasis15. The main limitations found in developing such tests are the low antigen yielding which does not permit further fractionation and analysis13, added to the difficulties in collecting blood samples for conducting seroepidemiological inquiries.
Immunological tests using the third-stage filariform larvae of S. stercoralis and Strongyloides ratti (Sandground, 1925), have shown comparable results6,9. Thus, the use of S. ratti has been preferred to overcome the problem in obtaining antigens in sufficient amount.
The use of blood samples collected on filter paper was proved to be useful for the serodiagnosis of parasitic diseases, as well as for seroepidemiological surveys in human and veterinary investigation. Thus this procedure has been applied with success in research on schistosomiasis2, Chagas' disease4,11,14,16, leishmaniasis1,12 and toxoplasmosis17.
Since the use of blood samples collected on filter paper for the diagnosis of human strongyloidiasis is very scarce, the objective of the present work is to utilize this procedure in a serological inquiry applying the indirect immunofluorescence antibody test (IFAT). In this test, the third stage filariform larvae of S. ratti was chosen as antigen because this parasite stage presented a sensitivity of 92.5% and specificity of 97.1%6.
Abadia dos Dourados is a city located in the Alto Paranaiba region in State of Minas Gerais, Brazil, its population is about 6,424 inhabitants. In July 1996, 207 individuals who live in the rural area were selected at random. These individuals were later identified according to sex and age, after their agreement for the participation in this project. Blood samples were obtained by digital puncturing with disposable lancets, collected on filter paper (Klabin 80 g/m^sup 2^), as described16 by SOUZA & CAMARGO, and stored in the Parasitology Laboratory of the Universidade Federal de Uberlandia at 4 deg C until November of 1996, when the blood elution was performed.
Filter papers were cut into 1.2 cm diameter discs using a metallic mold, and they were put individually in plastic bungs. On each bung 0.3 ml of phosphate buffered saline 0.01 M, pH 7.2 (PBS) were added and kept during 18 hours at 4 deg C. The IFAT with the eluates was done according to COSTA-CRUZ et al.6 using FITC-conjugated antihuman IgG at an ideal titer of 150. The analysis of the reactions was done using an immunofluorescence microscope (OLYMPUS BH-2 RFCA) at 400 X.
Of the 207 blood samples, 91 (44.0%) were from males and 116 (56.0%) from females. The age varied from two to 78 years. In the IFAT, 16 cases (7.7%) were seroreagents for strongyloidiasis. Table 1 shows the infection frequency of anti-Strongyloides antibodies in the inhabitants of the rural area in Abadia dos Dourados according to sex and age. Positive individuals of all age-groups were identified, with the youngest being seven years and the oldest being 74 years old. Applying the chi^sup 2^ test, there was no statistical significant difference between sex and age (p>0.05).
In view of the sensitivity (Sens) and specificity (Spec) of IFAT, we have calculated the true prevalence (PT) by a probabilistic model8. So, the obtained prevalence was corrected based on the following equation: True prevalence = (Obtained prevalence + Spec - 1)/(Sens + Spec - 1) or PT = (0.077 + 0.971 - 1)/(0.925 + 0.971 - 1) = 0.054. This correction seems plausible considering that the immunological test probably happen false negative results, as well as crossed reactions with others Nematoda infections. This model was also applied in the seroepidemiology of schistosomiasis mansoni10 by HOSHINO-SHIMIZU et al.
Among several epidemiological factors which could be related to the incidence and prevalence of strongyloidiasis, the habit of walking barefoot seemed a preponderant factor, since 197 (95.2%) of the 207 examined individuals used to be barefooted. But applying the chi^sup 2^ test there was no significant association between walking barefoot and the presence of seropositive cases of Strongyloides. Moreover, 14 of 16 seroreagent cases (87.5%) walked barefoot, 201 (97.1%) handled soil with unprotected hands corresponding to 13 positives cases, 187 (90.3%) usually defected directly on the soil, and 102 (49.3%) commonly drank unfiltered water. There is an association of this factors and its eventually explain the occurrence of strongyloidiasis in this region.
The collection of blood samples carried out on filter paper in seroepidemiological inquiries of strongyloidiasis is thought to be highly recommendable, since it is of low cost, easy storage and transportation and provides reliable data.
ACKNOWLEDGEMENTS
We wish to thank Conceicao R. Machado for collecting the blood samples, Dra. Maria Cristina Rink for the statistical analysis and Dr. David G. Francis for a critical reading of the manuscript.
REFERENCES
1. AFONSO-CARDOSO, S.R.; MACHADO, M.I.; COSTA-CRUZ, J.M. et al. Leishmaniose tegumentar canina no municipio de Uberlindia, Minas Gerais. Diagndstico clinico e sorologico de ces naturalmente infectados. Rev. Cent. Cienc. biomEd. Univ. Fed. Uberlandia, 5:14-21,1989. 2. ANDERSON, R.I.; SADUN, E.H. & WILLIAMS, J.S. - A technique for the use of minute amounts of dried blood in fluorescent antibody tests for schistosomiasis. Exp. Parasit., 11:111-116, 1961.
3. BOUREE, P.; TAGOURDEAU, A.; BARTHELEMY, M. et al. - Anguillulose: analyse clinique, biologique et epidemiologique de 350 observations. Nouv. Presse mid., 10: 679-681, 1981.
4. CAMARGO, M.E.; SILVA, G.R.da; CASTILHO, E.A. & SILVEIRA, A.C. - Inqugrito sorologico da prevalencia de infecgao chagasica, no Brasil, 19751 1980. Rev. Inst. Med. trop. S. Paulo, 26: 192-204, 1984. 5. CELEDON, J.C.; MATHUR-WAGH, U.; FOX, 1. et al. - Systemic strongyloidiasis in patients infected with the human immunodeficiency virus. A report of 3 cases and review of the literature. Medicine (Baltimore), 73: 256-263, 1994.
6. COSTA-CRUZ, J.M.; BULLAMAH, C.B.; GONCALVES-PIRES, M.R.ft et al. - Cryo-microtome cuts of coproculture larvae of Strongyloides stercoralis and Strongyloides ratti as antigen sources for the immunodiagnosis of human strongyloidiasis. Rev. Inst. Med. trop. S. Paulo, 39: 313-317, 1997. 7. DREYER, G.; FERNANDES-SILVA, E.; ALVES, S. et al. - Patterns of detection of Strongyloides stercoralis in stool specimens: implications for diagnosis and clinical trials. J. clin. Microbiol., 34: 2569-2571, 1996. 8. GART, J.J. & BUCK, A.A. - Comparison of a screening test and a reference test in epidemiologic studies. II. A probabilistic model for the comparison of diagnostic tests. Amer. J. Epidem., 83: 593-602, 1966.
9. GROVE, D.I. & BLAIR, J. - Diagnosis of human strongyloidiasis by immunofluorescence, using Strongyloides ratti and S. stercoralis larvae. Amer. J. trop. Med. Hyg., 30: 344-349, 1981. 10. HOSHINO-SHIMIZU, S.; DIAS, L.C.S.; KANAMURA, H.Y et al. Seroepidemiology of schistosomiasis mansoni. Mem. Inst. Oswaldo Cruz, 87 (suppl. IV): 303-306, 1992.
I1I. MINEO, J.R.; COSTA, J.M.; PIMENTA, C.V. et al. - Prevalencia da infeccao chagdsica em criancas de cinco municipios da regi8o Sudoeste de Minas Gerais, Brasil. Rev. Cent. Cienc. biomed. Univ. Fed. Uberlandia, 1: 3-6, 1985.
12. NUNES, V.LB.; YAMAMOTO, Y.I.; REGO-JUNIOR, FA. et al. - Aspectos epidemiologicos da leishmaniose visceral em cies de Corumba, Mato Grosso do Sul. Pesq. vet. bras., 8: 17-21, 1988. 13. ROSSI, C.L.; TAKAHASHI, E.E.H.; TEIXEIRA, A.L.S. et al. - Avaliacio de preparacoes antigenicas de Strongyloides stercoralis para o imunodiagnostico da estrongiloidiase. Rev. Soc. bras. Med. trop., 26: 8387, 1993.
14. SADUN, E.H.; DUXBURY, R.E.; WILLIAMS, J.S. & ANDERSON, R.I. Fluorescent antibody test for serodiagnosis of African and American trypanosomiasis in man. J. Parasit., 49: 385-388,1963.
15. SATO, Y; KOBAYASHI, J. & SHIROMA, Y. - Serodiagnosis of strongyloidiasis. The application and significance. Rev. Inst. Med. trop. S. Paulo, 37: 3541, 1995.
16. SOUZA, S.L. & CAMARGO, M.E. - The use of filter paper blood smears in a practical fluorescent test for American trypanosomiasis serodiagnosis. Rev. Inst. Med. trop. S. Paulo, 8: 255-258,1966.
17. WALTON, B.C. & ARJONA, I. - Utilization of whole blood specimens on filter paper for the indirect fluorescent antibody test for toxoplasmosis. J. Parasit., 57: 678-679, 1981.
Received: 08 June 1998
Accepted: 08 October 1998
(1) Laboratorio de Parasitologia, Departamento de Patologia, Universidade Federal de Uberlandia, Uberlandia, MG, Brasil.
(2) Instituto de Patologia Tropical e Sadde Publica, Universidade Federal de Goias, Goiania, GO, Brasil.
Correspondence to: Julia Maria Costa-Cruz. Laboratdrio de Parasitologia, Departamento de Patologia, Universidade Federal de Uberlandia. Av. Pardi 1720, 38400-902 Uberlandia, MG, Brasil. Fax: +55(34) 2182333.
Copyright Instituto de Medicina Tropical de Sao Paulo Sep/Oct 1998
Provided by ProQuest Information and Learning Company. All rights Reserved