It has been assumed that in asthmatic patients with Wolff-Parkinson-White (WPW) syndrome, ablative therapy for the condition is necessary for the safe treatment of the asthma with [[beta].sub.2]-adrenergic drugs. The following case report illustrates that inhaled albuterol was safely administered to an asthmatic patient with electrocardiographic evidence of preexcitation, without the need of an ablative procedure. This case report is, to our knowledge, the first in the literature in which the cardiac rhythm of a patient with WPW syndrome was monitored during repeated inhalations of a [[beta].sub.2]-agonist.
Patients with Wolff-Parkinson-White (WPW) syndrome, characterized by constant preexcitation and a short antegrade accessory pathway refactory period, are thought to be at risk for potentially fatal ventricular tachyarrhythmias during atrial fibrillation. On the other hand, patients with intermittent preexcitation are belived to be at lesser risk.[1] Isoproterenol infusion has been shown to enhance antegrade and retrograde conduction of the accessory pathway in patients with WPW syndrome, including those with intermittent preexcitation.[1]
Since it has been assumed that in asthmatic patients with WPW syndrome, ablative therapy for the condition is necessary for the safe treatment of the asthma with [beta]-adrenergic agonist,[2] we challenged our patient with an inhaled [[beta].sub.2]-agonist while continously monitoring for cardiac arrhthmias. The patient tolerated the challenge well and subsequent used an albuterol metered dose inhaler (MDI) without complications.
CASE REPORT
A 29-year-old woman with a diagnosis of WPM syndrome since 1984 presented with a 3-year history of nonproductive cough that was refractory to therapy with over-the-counter cough suppressants. She stated that the cough was present at night as well as during the day and was getting progressively worse. There were no seasonal changes. The cough was neither related to meals nor was it precipitated by a change in position. It was, however, exacerbated by upper respiratory tract infections. She denied shortness of breath, chest tightness, wheezing, and exacerbation of the cough with exercise or exposure to cold air. There was no history of hay fever, weight loss, fever, or anorexia. The patient denied palpitations but complained of increasing episodes of lightheadaches. She was not taking any medications at the time of presentation.
Her medial history was remarkable for two pregnancies resulting in a miscarriage and preeclampsia, and a complicated pregnancy requiring intensive care but not intubation. She denied ever smoking cigarettes. Her family history was negative for allergic rhinitis and asthma.
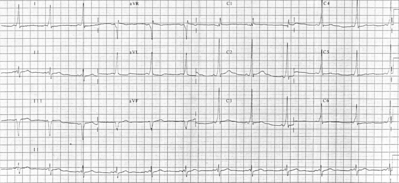
Results of the physical examination were within normal limits (WNL). Chemistry profile, complete blood cell count, and thyroid studies were all WNL. Sinus and chest roetgenograms were normal. Skin tests were positive for tree, grass, ragwood, cockroach, Dermatophagoide farine, and D pteronyssisnus. An ECG confirmed the presence of venticular preexcitation with no sinus arrhythmia (Fig 1). A two-dimensional echocardiogram was normal and did not show any evidence of Ebstein's anomaly.
We elected to asses the safety of a inhaled [[beta].sub.2]-adrenergic agent in our patient by administering three 2.5-mg doeses of nebulized albuterol over 1 h while continously monitoring the heart rhythm (Fig 2). Two premature ventricular contractions (PVCs) and occasional atrial premature beats with enhanced conduction through the accessory pathway were noted without subsequent induction of atrioventricular reciprocating tachyarrhythmia. Compared with her baseline drug-free monitoring, no increase in ectopy was noted after administration of the [[beta].sub.2]-agonist.
One week after the inhaled abuterol challenge, spirometry revealed the following data: FVC, 2.67 L (83 percent of predicted); [FEV.sub.1], 2.36 L (85 percent); [FEV.sub.1]/FVCV88.5; and FEF25-75, 2.97 L/s (99 percent). After two inhalations from a albuterol MDI, the results were as follows: FVC, 2.86 L (90 percent); [FEV.sub.1], 2.62 L (95 percent); [FEV.sub.1]/FVC = 91.6; and FEF25-75, 4.21 L/s (141 percent). The percent changes were 7, 11, 35, and 42, respectively. From the spirometry results, a diagnosis of cough-variant asthma was made.
Thereafter, albuterol MDI was administered every 4 h as needed while her cardiac rhythm was evaluated by continuous ambulatory monitoring. A 24-h Holter monitor showed a heart rate of 55 to 100 beats per minute, 2 PVCs, and 184 atrial premature beats. No reciprocating tacharrythmias were detected. Long-term therapy for her asthma with inhaled steriods and alubterol has been both well tolerated and successful in controlling her symptoms.
DISCUSSION
This report describes the use of an inhaled selective [[beta].sub.2]-agonist for the treatment of bronchial asthma in a patient with WPW syndrome, a condition in which infusion of nonselective [beta]-adrenergic agents has been shown to enhance antegrade and retrograde conduction through the necessary pathway.
In general, it is well known that selective [beta]-adrenergic agonists may cause PVCs and ventricular and nodal tachycardias,[3] although cardiac stimulation is considerably reduced by administration via the inhalation route.[4] Due to the need to administer an inhaled [beta]-adrengeric agonist for the evaluation and possible treatment, we studied the safety of inhaled albuterol in our patient.
The mechanism by which inhaled [beta]-adrenergic agonists induce arrhythmias is not known, but it has been postulated that they may be caused by a combination of hypoxemia, hypokalemia, and direct chronotropic effects of the [beta]-adrenergic agonist or fluorocarbons used as propellants.[5] In a patient with WPW syndrome, enhanced conduction or change in the refractory periods of an accessory pathway is an additional concern.
We selected to administer albuterol since it has been shown to be less arrhythmogenic than other [[beta].sub.2]-adrenergic agonists00.[6] Under constant ambulatory monitoring, the inhalation of albuterol failed to cause tachyarrhythmias. In addition, the atrial premature beats with enhanced conduction through the accessory pathway noted did not precipitate atrioventricular reciprocating tachycardia. Subsequent use of this agent in combination with an inhalation steriod has proved to be effective and safe while controlling her symptoms. This suggest that additional clinical safety trials in other such patients are warranted.
REFERENCES
[1] Yamamoto T, Yeh SJ, Lin FC, Wu DT. Effects of isoproterenol on accessory pathway conduction in intermittent or concealed Wolff-Parkinson-White syndrome. Am J Cardiol 1990: 65:1438-42
[2] Otake H, Watanabe G, Mukai A, Tubota M, Misaki T, Iwa T, et al. Treatment of Wolff-Parkinson-White syndrome associated with asthma. Kokyu To Jundan 1990: 38:1015-19
[3] Banner AS, Sunderrajan EV, Agarwal MK, Addington WW. Arrhytmogenic effects of orally administered bronchodilators. Arch Intern Med 1979; 139-434-37
[4] Wilson AF. Cardiovascular effects: [boto sub.2] agonists. J Asthma 1990: 27:111-16
[5] Tattersfield A. Autonomic drugs: In: Clark TJH, Godfrey S, eds. Asthma. 2nd ed. London: Chapman and Hal, 1983: 301-35
[6] Riding WD, Dinda P, Chatterjee SS. The bronchodilator and cardiac effects of five pressure-packed aerosols in asthma. Br J Dis Chest 1970;64:37-45
COPYRIGHT 1993 American College of Chest Physicians
COPYRIGHT 2004 Gale Group