CONTINUING EDUCATION
To earn CEUs, see test on page 20.
LEARNING OBJECTIVES
Upon completion of this article, the reader will be able to:
* Differentiate between primary and secondary iron overload disorders.
* Describe the nature and origin of three clinical features noted in hereditary hemochromatosis (HH).
* Discuss the molecular basis of iron metabolism regulation.
* Identify four genes and their products that are implicated in HH.
* Construct an algorithm of laboratory tests currently employed in the diagnosis of HH.
* Discuss the merits and limitations of screening tests in the identification of HH.
As an electron carrier in the cytochromes of the electron transport chain, as part of the functional groups of most enzymes of the Kreb's cycle, and as the oxygen-binding component in myoglobin and hemoglobin, iron is central to aerobic metabolism. When excessive amounts of iron accumulate in the body, cells are damaged and tissues are destroyed. Since iron is a catalyst in the formation of powerful oxidants--such as the highly reactive hydroxyl free radical--risk of damage to cell membranes, lipids, proteins, and nucleic acids is heightened.
[ILLUSTRATION OMITTED]
Of necessity, iron uptake and release by the body are carefully controlled. Conditions of iron overload can arise from a variety of causes. In primary overload disorders, iron absorption from a normal diet is increased due to inherited alterations in factors that control iron uptake and retention. Secondary iron overload may arise in patients with chronic disorders of erythropoiesis or hemolytic anemias (e.g., sideroblastic and sickle-cell anemias, beta thalassemia), as a consequence of intramuscular or intravenous iron therapy, due to excessive dietary or supplement ingestion of iron or from multiple blood transfusions. (1,2)
Iron overload in the United States arises primarily from a genetic disorder known as hereditary hemochromatosis (HH). In HH, not only is there an excess of iron in the body but there is also an abnormal distribution of the iron. (3,4) Abnormal serum iron measurements, confirmed by genetic testing, are the basis for a diagnosis of hereditary hemochromatosis. (5) This article will review clinical features of the disorder, describe the molecular basis for iron homeostasis, summarize the likely roles and interactions of multiple genes in regulating the body's iron balance, present current diagnostic and therapeutic approaches to HH, and consider screening strategies.
Disease overview
Among Caucasians of northern European descent, hereditary hemochromatosis is one of the most common genetic disorders. Prevalence is especially high among individuals of Celtic ancestry--the Irish, Scots, and Welsh. The hemochromatosis (HFE) gene responsible for the majority of cases of HH was first identified in 1996 as localized in band region 21.3 on the short (p) arm of chromosome 6. (6) The Centers for Disease Control and Prevention (CDC) estimate that more than 1 million Americans have hemochromatosis. In this country, the prevalence of the disease is close to 0.5%. Approximately five in every 1,000 individuals of Caucasian descent are homozygous for the major HFE mutation and at risk of developing iron overload. Among those of Caucasian descent, the frequency of heterozygosity (carrier status) in the United States is 1:8-10. (1,7,8)
Classic HH is inherited as an autosomal recessive disorder. Although mutations in the HFE gene are the principal cause of HH, mutations in other genes--including HJV (juvenile hemochromatosis) (human chromosome 1 band region q21); HAMP (hepatic antimicrobial peptide) (19q13.1); TfR2 (transferin receptor 2 gene) (7q22), and SLC40A [solute carrier family 40 (iron-regulated transporter)] (2q32)--also have the potential to affect iron homeostasis by altering levels of various proteins essential for regulation of the body's iron balance.
Hepatic damage--ranging in severity from fibrosis to carcinoma--is common in HH. Deposition of excess iron in hepatic parenchymal cells presumably increases oxidative stress on already damaged cells and increases the risk of cirrhotic tissue undergoing malignant transformation. Primary hepatocellular carcinoma is reported to be responsible for about one-third of deaths in patients homozygous for HFE mutation that have already developed cirrhosis. (9) Because of the slow, progressive nature of iron accumulation and subsequent organ damage, individuals do not usually present with symptoms until in their 40s or 50s. Women are less likely than men to develop iron overload, or they may present with clinical expression of the disorder later in life because of blood loss incurred through menstruation and pregnancy. Pancreatic injury, leading first to insulin resistance and subsequently to beta-cell failure, along with changes in skin pigmentation, are also noted in advanced stages of HH.
Early descriptions of the basic features of the disease referred to "bronze diabetes" (diabetes mellitus and bronzing of the skin), along with cirrhosis of the liver. The term "hemochromatosis" was first applied to this triad of clinical features in 1889 when it was recognized that the disease was linked to a progressive increase in iron stores. (10) The body of an adult male typically contains a total of three to four grams of iron; an adult female, three-and-one-half grams. Two-thirds of the body's iron is present in hemoglobin, myoglobin, and iron-containing enzymes, with the remainder occurring in the storage forms, ferritin and hemosiderin. The liver is the major site of iron storage. Iron levels in hereditary hemochromatosis may exceed normal values by as much as 10 times.
In addition to damage to the liver, pancreas, and skin, chronic iron overload jeopardizes functioning of the heart. Pituitary damage leading to impaired gonadotropin release often occurs and can cause loss of sex drive, along with erectile dysfunction and testicular atrophy in men and amenorrhea or early menopause in women. Arthritis arises from joint damage caused by iron deposition. (12) Initial signs and symptoms of hemochromatosis are not exclusively associated with iron overload. Nationwide studies indicate that--on average--three doctors and nine years are required before a patient is correctly diagnosed. (13) Fatigue, weakness, depression, weight loss, abdominal pain, and arthralgia, especially in the fingers and hands, are sufficiently nonspecific to make early diagnosis difficult, unless there is a family history of hemochromatosis or laboratory tests yield biochemical evidence of iron overload. Genetic testing can aid in clinical decision making but cannot be used alone in disease diagnosis. The presence of abnormal transferrin saturation (TS) and serum ferritin (SF) levels provide evidence that increased risk has become the reality of iron overload.
[FIGURE 1 OMITTED]
Basics of iron homeostasis
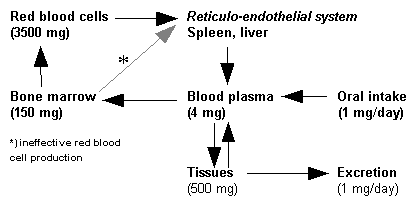
The principal means by which the body normally loses iron include sloughing of ferritin-containing intestinal cells and menstruation. But it is not by modulating iron loss that the body closely regulates iron levels; it is iron uptake that is precisely controlled to maintain iron balance. (14,15) In the duodenum, dietary free iron is enzymatically reduced from the ferric (+3) form to ferrous (+2) iron and taken up from the intestinal lumen into the enterocytes by the iron transport protein, divalent metal transporter 1 (DMT1). In addition to being necessary for iron uptake in the intestine, DMT1 also is instrumental in the uptake of iron by erythropoietic cells. Once absorbed, iron may be stored as ferritin in the enterocytes, or it may cross the enterocyte and be exported into the circulation by another iron transport protein, ferroportin 1 (fpn1). In the export process, iron is reoxidized to Fe(+3). (16)
Ferroportin also seems to be important in recycling iron by moving it out of macrophages of the liver, spleen, and bone marrow that have recovered hemoglobin iron by phagocytizing old or damaged red blood cells and returning it to the circulation. In the plasma, ferric iron binds to transferrin. From its complex with transferrin, iron is delivered into cells by binding to transmembrane glycoprotein, transferrin-receptors (TfR). Receptor-mediated endocytosis of ferric transferrin follows. It has been suggested that HFE protein interacts with TfRs to regulate cellular iron uptake.
An alternative model focuses on the formation of a complex of ferroportin and hepcidin, a liver-synthesized peptide, as the key event in iron hemostasis. Hepcidin production would increase in the liver when iron bound to transferrin is taken into cells via TfRs. When released into the circulation, hepcidin would function as the hormonal regulator of iron uptake. (17) By binding with the cellular membrane transport protein ferroportin, hepcidin would reduce intestinal iron absorption and decrease the release of cellular iron, chiefly from macrophages and hepatic stores. Formation of a ferroportin-hepcidin complex, its subsequent internalization and degradation, would effectively lock iron in these cells by preventing iron efflux.
Hepcidin was originally discovered in the urine and possesses bactericidal properties. The gene that encodes for hepcidin is designated as HAMP, and its expression may be regulated by HFE protein. (18,19) Hepcidin expression is increased in iron loading and decreased by iron deficiency. Any mutation that results in a deficiency of hepcidin production or in altered function of hepcidin's target, the iron export protein ferroportin, could lead to excessive accumulation of iron in the cells. (17)
Genetic basis of HH
The phenotype or outward, physical manifestations of iron overload may arise from different genotypes. Even more challenging is the fact that the same genotype may be expressed to a different degree phenotypically in different individuals depending upon environmental factors such as diet and alcohol consumption, as well as other non-HFE-related genetic factors. The National Center for Biotechnology Information (NCBI) Online Mendelian Inheritance in Man (OMIM) database lists four types of HH as well as several subtypes (see Table 1). (9)
In Type 1 HH, the HFE gene is mutated. More than 40 alleles of the HFE gene have been reported. (20) Two HFE mutations, C282Y and H63D, account for the majority of cases of hereditary hemochromatosis. (21) In the most common mutation associated with an actual clinical diagnosis of HH, a guanine-to-adenine replacement in DNA results in the substitution of the amino acid cysteine by tyrosine at position 282 (282C[right arrow]Y) in the protein coded for by HFE. (22) The HFE protein plays a key, though not yet clearly defined, role in regulating the overall efficiency of intestinal iron absorption. Suggested roles for the HFE protein in regulating iron absorption include 1) binding with a complex containing TfR1 on the enterocyte; 2) facilitation of transferrin-bound iron uptake; or 3) modulation of the expression of hepcidin. Depending upon the population examined, from 85% to 100% of HH patients have been reported to be homozygous for C282Y. (9,23,24) The frequency of the C282Y mutation is especially high among northern European populations. More than 80% of northern European patients with HH are homozygous for the C282Y mutation in the HFE gene. (19) In Ireland, the gene frequency is such that >20% of the population is heterozygous (carriers of the gene). (3) Molecular studies suggest that the mutation is fairly recent--perhaps having occurred as a chance event in a single Celtic or Viking (Scandinavian) ancestor in northwestern Europe 2,000 years (or 100 to 140 generations) ago. (1,3,6)
Although the prevalence of C282Y is high, its penetrance is low. This means that even if an individual is homozygous for C282Y, that person is on the whole still not especially likely to manifest clinical evidence of iron overload. (25) Most individuals who are heterozygous for HFE-mutations will not develop clinical manifestations of HH although counseling and ongoing monitoring are desirable.
The most common HFE mutation is H63D. By itself, however, H63D is not associated with an iron-overloading syndrome; that is, the individual's iron parameters remain normal. Replacement of histidine by aspartate at position 63 (63 H[right arrow]D) in the HFE protein arises from this mutation. Among Caucasians, the frequency of this allele is reported to be 15% to 20%, but its frequency is as high as 30% among the Basque population in Spain. (22) Basques currently occupy the border area of France and Spain and seem to be genetically quite different from their European neighbors. The possibility has been raised that they have a greater proportion of ancestral (or Paleolithic) autosomal alleles than other European populations. (26) It has been suggested that the H63D mutation of HFE gene evolutionarily predates that of the C282Y allele. (27) The H63D allele appears to act synergistically with the C282Y mutation, and 2% of compound heterozygotes have clinical symptoms of HH. (28,29)
Even today, the World Health Organization (WHO) considers iron deficiency to be the number one nutrition disorder worldwide. As much as 80% of the world's population, especially woman and children, may be iron deficient. (30) There is speculation that individuals possessing mutations of the HFE gene may have been favored by natural selection since they would have enhanced uptake of iron from iron-poor diets, thus minimizing their risk for iron deficiency. A further benefit of genetic mutation may have been conferred on heterozygotes who were unlikely to develop iron overload but in whom alterations in intracellular iron levels might protect against infectious agents such as Yersinia and Francisella--causative agents of plague and tularemia, respectively. Selection pressure that maintains the heterozygous state for hemoglobin S in populations at risk of malaria provides a similar model. (3,31)
Hemochromatosis Type 2 (also known as juvenile hemochromatosis) is an autosomal recessive disorder existing in two forms. The first (subtype A) is caused by a mutated hemojuvelin (HJV) gene (chromosome 1 band region q21). The HJV gene codes for a membrane protein, hemojuvelin. The second (subtype B) originates from mutation in the hepcidin gene on chromosome 19, band q13, which encodes hepcidin antimicrobial peptide or HAMP. In both of these HH subtypes, hepcidin is either absent or undetectable in the urine. Onset of juvenile HH may be as early as the teens but usually presents in the second and third decades of life. Tissue iron accumulation is more rapid than in adult-onset forms. Congestive heart failure or arrhythmias due to myocardial accumulations of iron may cause death from heart failure before the age of 30. (6)
[FIGURE 2 OMITTED]
Another autosomal recessive disorder, hemochromatosis Type 3, is attributed to mutations in the TFR2 located in band region q22 on chromosome 7. Among the various forms of HH, only Type 4 has been identified as having an autosomal dominant pattern of inheritance. It originates from a mutation of gene SLC40A1 on chromosome 2, band region q32, which encodes for the intestinal and macrophage iron exporter protein ferroportin. Patients with Type 4 HH mostly develop iron loading of the liver macrophages or Kupffer cells, but saturation of plasma transferrin is relatively low. Other Type 4 patients present with high transferrin saturation and iron-loaded hepatocytes. (17,32)
Diagnostic testing
Laboratory abnormalities encountered in HH include increased levels of alanine and aspartate aminotransaminases, or AST/ALT, elevated serum iron transferrin saturation (>60%), increased serum ferritin, and higher amounts of hepatic parenchymal cell stainable iron. (9) Serum TS and SF levels are commonly the first laboratory measurements performed for symptomatic individuals. Typically, serum iron-binding proteins are one-third saturated, and the reference range for percent saturation of transferrin is 20% to 50%. Nonfasting TS between 45% and 60% is considered borderline high, with a value above 60% considered an indication of iron overload. Since elevations of TS have been associated with consumption of high doses of vitamin C, iron supplementation, and estrogen preparations, their use should be discontinued if possible at least 24 hours prior to testing.
Depending on the immunoassay used, ferritin concentration is typically 20 [micro]g/L to 300 [micro]g/L in men and 10 [micro]g/L to 120 [micro]g/L in women. (33) Serum ferritin levels are considered high for men and postmenopausal women if over 300 [micro]g/L and high for premenopausal women if over 200 [micro]g/L. Of note, elevated SF levels are commonly seen in people with chronic alcohol consumption, hepatitis B or C, obesity-related steatohepatitis, infection, cancer, heart disease, AIDS, metabolic disorders, and inflammatory conditions. (21) Although less frequently used, measurement of the unbound iron-binding capacity, which is the reserve capacity of transferrin to bind iron, has been suggested to offer advantages greater than those of TS. (34) After confirming elevated TS and SF levels, genetic testing may be performed. If the patient is found to be heterozygous for the HFE gene, liver function tests (LFT) may be conducted to differentiate HFE-related iron overload from other hepatic disorders. If homozygous for the HFE genotype, >40 years of age, with SF <1000 [micro]g/L, and LFTs within reference range, the patient is referred for phlebotomy. If the patient is homozygous for the HFE gene, age is <40, with SF >1000 [micro]g/L, and elevated LFTs, the patient is referred for liver biopsy and phlebotomy. (35)
Treatment and management of hereditary iron overload by phlebotomy requires removal of 500 mL of blood once or twice a week, perhaps for up to several years, until there is a marked decrease in serum transferrin saturation and serum ferritin. Following this initial, aggressive treatment phase to achieve iron depletion (SF <50 [micro]g/L; TS <50%), phlebotomy frequency is reduced in the management phase of HH. Periodic monitoring of ferritin levels is required for life, with phlebotomy performed often every two to six months. (28) Use of a chelating agent, such as deferoxamine, is also helpful in reducing iron stores when phlebotomy is not appropriate, as in cases of anemia, cardiac dysfunction, and advanced liver cirrhosis. It must be administered, however, parenterally and is expensive, while phlebotomy is safe, effective, and inexpensive. The hormone erythropoietin, which increases the production of red blood cells, may be given to improve phlebotomy tolerance in Type 4 HH patients. (10)
At this time, the American Red Cross (ARC) does not accept individuals with hemochromatosis as blood donors. (36) An ARC regional pilot program--expected to be complete by the end of this year--for hemochromatosis donors, however, will be evaluated for possible system-wide implementation. Variance from existing federal regulations regarding labeling and frequency of blood collection can be requested by hospitals and blood banks for application to patients with HH. The guidance document and current list of sites with variances are available from the U.S. Food and Drug Administration (FDA). (37)
Liver biopsy is often performed in patients whose SF levels exceed 1000 [micro]g/L to differentiate hereditary hemochromatosis from other hepatic diseases. If HH is diagnosed, a liver biopsy may be performed to determine the extent of liver damage when 1) SF levels are >1000 [micro]g/L, or 2) if SF is <1000 [micro]g/L but LFT results are abnormal. (8) To actually determine hepatic iron concentration and prognosis based on the extent of tissue damage, liver biopsy is the gold standard.
Public health screenings
Universal screening (i.e., testing asymptomatic individuals) for HH remains a controversial issue. The American Hemochromatosis Society is highly supportive of genetic testing for anyone whose ancestry might put them at increased risk of the disease or for those who have nonspecific signs or symptoms that might arise from HH. (38) While the prognostic value of genetic testing may be limited given the low penetrance of HH and the uncertain predictive value of specific genotypes for future disease, the Society sees no reason not to advocate screening. Identification of persons with at-risk genotypes could motivate these individuals to communicate with family members about the possibility of disease development, to seek genetic counseling, and to inform the family practitioner of an increased need for ongoing monitoring of iron status and hepatic parameters. A proactive approach seems to offer more positives than negatives given that if a clinical problem is detected early, the inexpensive implementation of therapeutic phlebotomy can prevent development of life-threatening tissue damage. The organization disseminates information on independent laboratories it has identified that offer relatively inexpensive biochemical and genetic testing services. The laboratories hold appropriate certification and, along with test results, provide interpretative information and access to genetic counseling. Particularly attractive is the option offered of testing a cheek-brush sample rather than requiring a blood draw. Genetic test kits may be ordered online for approximately $200. (39)
The CDC does not advocate population screening for HFE mutations, but it does recommend family-based screening for detection of HH. Beginning this year, it contracted with a research group to enroll Chicago- and Boston-area HH patients (both cities have substantial numbers of residents of Celtic ancestry) in order to examine the role of family-based detection in the early diagnosis of HH. (40) The potential value of large-population-based studies as a public-health response to addressing various aspects of HH has been considered for some time.
Recently, findings were reported from the five-year Hemochromatosis and Iron Overload Screening (HEIRS) study, in which approximately 100,000 individuals were screened and assessed at five U.S. and Canadian medical centers for mutations of the HFE gene and for blood-iron levels. Major findings of the study included 1) the highest prevalence of homozygosity for the HFE mutation C282Y was among non-Hispanic whites (4.4 individuals per 1,000); 2) the prevalence of the C282Y/C282Y genotype among minorities was Native Americans, 1.1/1000; Hispanics, 2.7/10,000; African-Americans, 1.4/10,000; Pacific Islanders, 1.2/10,000; and Asians, 3.9/10 million. Most C282Y homozygotes have elevations in SF and TS. While confirming findings on iron overload and HH in Caucasians from other more limited studies, HEIRS data revealed some surprising information about the prevalence of these disorders in minorities. Asians and Pacific Islanders had the highest average blood-iron test levels although they had the lowest prevalence of the gene mutation. Some African-Americans may have iron overload attributable to genetic mutations other than those common among Caucasians. (27)
Whether insurance coverage will be provided to cover screening tests for HH is problematic. Concern has been expressed as to how insurance companies and, perhaps, even employers will respond to individuals whose screening test is positive but who are asymptomatic. (41) Most insurance companies will reimburse for genetic testing if prior biochemical testing suggests a risk for HH or if there is a family history of the HFE gene. Reimbursement for testing judged to be motivated only by curiosity is unlikely. Some insurance companies may deny coverage or charge exorbitant premiums, knowing a customer has tested positive for a gene linked to hereditary hemochromatosis. Each company's coverage and reimbursement policies differ, and state laws vary regarding genetic discrimination. Understandably, an individual may opt to pay out-of-pocket for confidential testing by a commercial laboratory. (8)
Looking back, looking ahead
Inherited defects in iron homeostasis can lead to progressive damage of the liver, heart, and pancreas. If diagnosis and treatment are delayed, morbidity and mortality are increased. Biochemical and genetic testing offer the means of early identification of an actual or potential iron-overload disorder. Researchers have made tremendous progress in identifying the major players in the genetic control of iron homeostasis. (42) More aggressive education of practitioners and the public could lead to the removal of HH from the list of the top 10 misdiagnosed diseases by physicians. (43) Although most frequently associated with Caucasians of Northern European ancestry, genetic disorders of iron metabolism are now being identified in minority populations.
While we have examined some of the problems of identifying and managing HH, perhaps we should also consider some "opportunities" arising from this so-called "Celtic curse." The treatment of HH includes dietary modification to reduce iron intake and the regular performance of therapeutic phlebotomy. Cooperation between laboratorians and dietitians in educating patients on their condition is one such opportunity; a second is the wider use of hemochromatotic blood. Perhaps the need for special FDA variances should be eliminated, and blood from HH patients with no organ damage should be recruited to augment declining blood supplies. (44,45) In a time of global conflicts and shortages of blood, a clearer understanding of the molecular basis for HH may go a long way toward addressing concerns raised over the use of blood from individuals with hereditary hemochromatosis. (46)
Julie A. Meeker, RD, LDN, is a clinical dietitian at Centegra Health System in Woodstock, IL, and a faculty member in Life Science and Human Services at Harper College in Palatine, IL. Sharon M. Miller, PhC, MT(ASCP), CLS(NCA), is professor emeritus of Clinical Laboratory Sciences, College of Health and Human Sciences, Northern Illinois University, DeKalb, IL.
References
1. Powell L. Recent concepts in iron metabolism and HFE mutations. J Gastroenterol Hepatol. 2004;19(Suppl 7):S258-S263.
2. Ghai S, Sharma R, Kulshreshtha B. Multiorgan involvement in thalassaemia major. Postgrad Med J. (serial online). 2003;79:358. Available at: http://www.postgradmedj.com. Accessed July 13, 2005.
3. Beutler E. Iron absorption in carriers of the C282Y hemochromatosis mutation. Am J Clin Nutr. 2004;80:799-800.
4. Iron-overload disease due to hereditary hemochromatosis. Centers for Disease Control and Prevention. Available at: http://www.cdc.gov/genomics/info/factshts/hemoactiv.htm. Accessed July 19, 2005.
5. Chung RT, Greenberger NJ. Hereditary hemochromatosis--early diagnosis can lead to cure. Patient Care. 2003;37:54-59.
6. Pietrangelo A. Hereditary hemochromatosis--A new look at an old disease. N Engl J Med. 2004;350:2383-2397.
7. Hemochromatosis. National Institute of Diabetes, and Digestive and Kidney Disease. Available at: http://digestive.niddh.nih.gov/ddiseases/pubs/hemochromatosis/. Accessed June 25, 2005.
8. Hemochromatosis: How genes cause hemochromatosis. DNA direct. Available at: http://www.dnadirect.com/resource/conditions/hfe/GH_Hemo_Genes_Cause.jsp. Accessed May 21, 2005.
9. Hemochromatosis. Online Mendelian Inheritance in Man. Johns Hopkins University. Available at: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?db=OMIM. Accessed June 24, 2005.
10. Franchini M, Veneri D. Recent advances in hereditary hemochromatosis. Ann Hematol. 2005;84:347-352.
11. Training: Iron overload disease due to heredity hemochromatosis. CDC Program Activities & Priorities 2001. Centers for Disease Control and Prevention. Available at: http://www.cdc.gov/genomics/info/factshts/hemoactiv.htm. Accessed July 20, 2005.
12. Mallory MA, Lee SW, Kowdley KV. Abnormal liver test results on routine screening. How to evaluate, when to refer for a biopsy. Postgrad Med. 2004;115(3):53-62.
13. Dennison A. Easily treated iron-overload disorders underdiagnosed. Daily News Central. Available at: http://health.dailynewscentral.com/content/view/000682/47. Accessed April 29, 2005.
14. Roy CN, Enns CA. Iron homeostasis. New tales from the crypt. Blood. 2000;96(13):4020-4027.
15. Andrews NC. Molecular control of iron metabolism. Best Pract Res Clin Haematol. 2005;18(2):159-169.
16. Fraenkel PG, Traver D, Donovan A, Zahrieh D, Zon LI. Ferroportin 1 is required for normal iron cycling in zebrafish. J Clin Invest. 2005;115:1532-1541.
17. Ganz T. Hepcidin--a regulator of intestinal iron absorption and iron recycling by macrophages. Best Pract Res Clin Haematol. 2005;18(2):171-182.
18. Fleming RE, Sly WS. Hepcidin: A putative iron-regulatory hormone relevant to hereditary hemochromatosis and the anemia of chronic disease. PNAS. 2001;98(15):8160-8162.
19. Merryweather-Clarke AT, Cadet E, Bomford A, et al. Digenic inheritance of mutations in HAMP and HFE results on different types of hemochromatosis. Hum Mol Genet. 2003;12(17):2241-2247.
20. Objections to genetic testing. Iron Overload Diseases Association. Available at: http://www.ironoverload.org. Accessed March 29, 2005.
21. Hemochromatosis for health care professionals. Centers for Disease Control and Prevention. Available at: http://www.cdc.gov/hemochromatosis/training/course_summary/index.htm. Accessed April 20, 2005.
22. Tomatsu S, Orii KO, Fleming RE, et al. Contribution of the H63D mutation in HFE to murine hereditary hemochromatosis. PNAS. 2003;100(26):15788-15793.
23. Lee J-Y, Yoo K-H, Hahn S-H. HFE gene mutation, C282Y causing hereditary hemochromatosis in Caucasians is extremely rare in Korean population. J Korean Med Sci. 2000;15(2):179-182.
24. Cazzola M. Novel genes, proteins, and inherited disorders of iron overload: iron metabolism is less boring than thought. Haematologica. 2002;87(2):115-116.
25. Waalen J, Nordestgaard BG, Beutler E. The penetrance of hereditary hemochromatosis. Best Pract Res Clin Haematol. 2005;18(2):203-220.
26. Flores CF. Reduced genetic structure of the Iberian peninsula revealed by Y-chromosome analysis: implications for population demography. Eur J Hum Genet. 2004;12:855-863.
27. Adams PC, Reboussin DM, Barton JC, et al. Hemochromatosis and iron-overload screening in a radically diverse population. N Engl J Med. 2005;352(17):1769-1778.
28. Hemochromatosis mutation detection, C282Y, H63D, and S65C. ARUP Laboratories. Available at: http://www.aruplab.com/media/pdf/testing/tech_bulletins/hemochrom)may02.pdf. Accessed July 26, 2005.
29. Tavill A. Clinical implications of the hemochromatosis gene. N Engl J Med. 1999;341(10):755-757.
30. Dietary supplement fact sheet: Iron. Office of Dietary Supplements. Available at: http://ods.od.nih.gov.factsheets/iron.asp. Accessed June 28, 2005.
31. Moalen S, Weinberg ED, Percy ME. Hemochromatosis and the enigma of misplaced iron: implications for infectious disease and survival. Biometals. 2004;17:135-139.
32. De Domenico I, Ward DM, Nemeth E, et al. The molecular basis of ferroportin-linked hemochromatosis. PNAS. 2005;102(25):8955-8960.
33. Mroz RC Jr. Electrolytes. In: Anderson SC, Cockayne S, eds. Clinical Chemistry: Concepts and Applications. New York: McGraw-Hill; 2003: 401-440.
34. Adams PC, Reboussin DM, Leinendecker-Foster C, Moses GC, McLaren GD, et al. Comparison of the unsaturated iron-binding capacity with transferrin saturation as a screening test to detect C282Y homozygotes for hemochromatosis in 101168 participants in the Hemochromatosis and Iron Overload Screening (HEIRS) Study. Clin Chem. 2005;51(6):1048-1052.
35. Powell L, Jazwinska EC. Hemochromatosis in heterozygotes. N Engl J Med. 1996;335(24):1837-1839.
36. Hemochromatosis. American Red Cross. Available at: http://www.redcross.org. Accessed July 26, 2005.
37. Variances for collection of blood and blood products from patients with hemochromatosis. Food and Drug Administration. Available at: http://www.fda.gov/cber/gdlns/hemochrom.htm. Accessed June 10, 2005.
38. Prevention through genetic testing. American Hemochromatosis Society. Available at: http://www.americanhs.org/presidentsletter.htm. Accessed July 20, 2005.
39. Discovering your Celtic connection. American Hemochromatosis Society. Available at: http://www.americanhs.org. Accessed March 10, 2005.
40. Iron overload and hemochromatosis: Study on hemochromatosis to begin early 2005. Centers for Disease Control and Prevention. Available at: http://cdc.gov/hemochromatosis/study_2005.htm. Accessed July 20, 2005.
41. Heredity hemochromatosis. American Family Physician. Available at: http://familydoctor.org/758.xml. Accessed March 11, 2005.
42. Gasparini P, Camaschella C. Hereditary hemochromatosis: Is the gene race over? Eur J Hum Genet. 2004;12:341-342.
43. Misdiagnosis of hemochromatosis. Wrong Diagnosis. Available at: http://www.wrongdiagnosis.com/h/hemochromatosis/misdiag.htm. Accessed August 5, 2005.
44. Casella GA, Biella AB, Signorini SC, Tramacere PL, Baldini VA. Hereditary hemochromatosis without organ damage: a rescue resource for blood supply. Eur J Gastroenterol. 2004;16(12):1419-1420.
45. Davey RJ. Recruiting blood donors: challenges and opportunities. Transfusion. 2004;44(4):597-600.
46. Voelker R. Hemochromatosis patients are untapped resource of blood as war, shortages loom. JAMA. 2003;289:1364-1366.
CE test on HEREDITARY HEMOCHROMATOSIS
MLO and Northern Illinois University (NIU), DeKalb, IL, are co-sponsors in offering continuing education units (CEUs) for this issue's article on HEREDITARY HEMOCHROMATOSIS. CEUs or contact hours are granted by the College of Health and Human Sciences at NIU, which has been approved as a provider of continuing education programs in the clinical laboratory sciences by the ASCLS P.A.C.E.[R] program (Provider No. 0001) and by the American Medical Technologists Institute for Education (Provider No. 121019; Registry No. 0061). Approval as a provider of continuing education programs has been granted by the state of Florida (Provider No. JP0000496), and for licensed clinical laboratory scientists and personnel in the state of California (Provider No. 351). Continuing education credits awarded for successful completion of this test are acceptable for the ASCP Board of Registry Continuing Competence Recognition Program. After reading the article on page 10, answer the following test questions and send your completed test form to NIU along with the nominal fee of $20. Readers who pass the test successfully (scoring 70% or higher) will receive a certificate for 1 contact hour of P.A.C.E.[R] credit. Participants should allow four to six weeks for receipt of certificates.
The fee for this continuing education test is $20.
All feature articles published in MLO are peer-reviewed.
Learning objectives and CE questions were prepared by Julie A. Meeker, RD, LDN, a clinical dietitian at Centegra Health System in Woodstock, IL, and a faculty member in Life Science and Human Services at Harper College in Palatine, IL; and Sharon M. Miller, PhC, MT(ASCP), CLS(NCA), professor emeritus of Clinical Laboratory Sciences, College of Health and Human Sciences, Northern Illinois University, DeKalb, IL.
1. Individuals diagnosed with all of the following are at increased risk for developing secondary iron overload except
a. hereditary hemochromatosis.
b. beta thalassemia.
c. sideroblastic anemia.
d. parenteral iron therapy.
2. Iron overload disorders in the United States are chiefly the result of
a. excessive mineral supplement ingestion.
b. multiple transfusions.
c. hereditary hemochromatosis.
d. sickle-cell anemia.
3. Hereditary hemochromatosis is most common among individuals whose ancestors were
a. African.
b. Irish.
c. Italian.
d. Native American.
4. The gene whose mutation gives rise to most cases of HH is
a. HFE.
b. HJV.
c. HAMP.
d. TfR.
5. The classic triad of clinical manifestations associated with HH includes all of the following except
a. cirrhosis.
b. diabetes mellitus.
c. increased skin pigmentation.
d. cataracts.
6. Nonspecific symptoms that may be due to HH include
a. arthritis, depression, weight loss, abdominal pain.
b. weight gain, increased libido, fluid retention, headaches.
c. dizziness, shortness of breath, heavy menses, loss of hearing.
d. loss of visual acuity, poor bladder control, increased thirst, heartburn.
7. Signs of organ damage in HH are most likely to appear
a. before the early 20s.
b. in the mid-40s.
c. after the late 60s.
d. in the 80s.
8. Current research suggests that the hormonal regulator of the body's iron stores is
a. hirudin.
b. hemosiderin.
c. ferroportin.
d. hepcidin.
9. The most common genetic mutation associated with clinical symptoms of iron-overload is
a. H63D.
b. HAMP.
c. SLC40A1.
d. C282Y.
10. The most frequently occurring HFE mutation is
a. C282Y.
b. H63D.
c. HAMP.
d. HJV.
11. An individual whose genotype is C282Y/H63D would be classified as having what type of HH?
a. Type 1.
b. Type 2A.
c. Type 2B.
d. Type 3.
e. Type 4.
12. Because the penetrance of the gene mutation is so high, individuals who are homozygous for C282Y will develop symptoms of HH in their teens.
a. TRUE
b. FALSE
13. Mutation of the HJV gene is the basis for
a. HH Type 1.
b. HH Type 2A.
c. HH Type 2B.
d. HH Type 3.
e. HH Type 4.
14. Onset of symptoms of juvenile HH typically occurs
a. earlier in girls than in boys.
b. shortly after birth.
c. within the first year of life.
d. in the teens.
15. The death of a young man of Celtic descent from cardiac arrhythmia could be a consequence of HH Type 2, especially if his parents were known to have hemochromatosis.
a. TRUE
b. FALSE
16. An autosomal dominant pattern of inheritance distinguishes
a. HH Type 1.
b. HH Type 2A.
c. HH Type 2B.
d. HH Type 3.
e. HH Type 4.
17. Primary laboratory tests performed in the diagnosis of HH are a measurement of
a. AST/ALT and UIBC.
b. hepatic iron stores and fasting blood glucose.
c. hemoglobin and hematocrit.
d. serum ferritin and transferrin saturation
18. Which of the following is not associated with elevated levels of serum ferritin?
a. Chronic alcohol consumption.
b. Hepatitis B or C.
c. Anemia.
d. Inflammatory conditions.
19. To determine hepatic iron concentration and prognosis of HH based on extent of tissue damage, the "gold standard" procedure is
a. liver biopsy.
b. UIBC measurement.
c. genetic testing.
d. ALT/AST assessment.
20. An indication of iron overload is a nonfasting transferrin saturation value greater than
a. 45%
b. 60%
c. 300 [micro]g/L.
d. 1000 [micro]g/L.
21. In the early stages of HH management, iron depletion of the patient is achieved when
a. serum ferritin <50 [micro]g/L.
b. hemoglobin <14 mg/dL.
c. hematocrit <5%.
d. AST <35 IU/L.
22. The HEIRS study found the highest prevalence of the C282Y/C282Y genotype was among
a. African-Americans.
b. Hispanics.
c. Asians.
d. non-Hispanic whites.
23. The CDC does not advocate population screening for HFE mutations because of the high cost of genetic testing.
a. TRUE
b. FALSE
24. The American Red Cross accepts individuals with HH for autologous donations only.
a. TRUE
b. FALSE
25. An individual homozygous for C282Y has an 80% to 90% chance of developing iron overload hepatic damage.
a. TRUE
b. FALSE
[GRAPHIC OMITTED]
By Julie A. Meeker, RD, LDN, and Sharon M. Miller, PhC, MT(ASCP), CLS(NCA)
Internet resources
American Hemochromatosis Society
www.americanhs.org
Iron Overload Diseases Association
www.ironoverload.org
Centers for Disease Control and Prevention
www.cdc.gov
American Red Cross
www.redcross.org
National Center for Biotechnology Information
www.ncbi.nlm.nih.gov
American Academy of Family Physicians
www.aafp.org
Mayo Clinic
www.mayoclinic.com
Iron Disorders Institute
www.irondisorders.org
COPYRIGHT 2005 Nelson Publishing
COPYRIGHT 2005 Gale Group