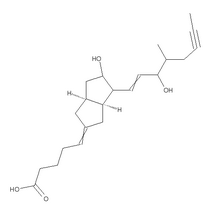
GENERAL
Introduction: Although well-recognized in adults, RSD is rarely diagnosed in children. Management is still controversial and includes, mobilization and physical therapy, spinal cord stimulation, transcutaneous electrical nerve stimulation, steroids, tricyclic antidepressants, anticonvulsants, non-steroidal anti-inflammatory drugs, injections of calcitonin, vasodilators and calcium channel blocker or alpha-sympathetic blocker. In this study, we describe the treatment of RSD in children using Iloprost, a prostacyclin analog that mimics sympathicolysis. We report our treatment regime, the clinical course, complications and the outcome in our first seven patients.
Patients and Methods: Seven female patients with a mean age of 9 years (6 to 11 years) suffering from reflex sympathetic dystrophy (RSD) stage II were included in this prospective study. Inclusion criteria were RSD stage II - III, an age between 4 to 12 years, no previous operative procedures and duration of symptoms for a minimum of 6 months. Diagnosis of RSD was based on the presence of neuropathic pain, such as burning, dysaesthesia, paresthesia, and hypalgesia to cold, and physical signs of autonomic dysfunction such as skin cyanosis, mottling, hyperhidrosis, edema and coldness of the extremity. Treatment regime consisted of two infusions of Iloprost (IlomedinO, Schering AG, Germany) administered over 6 hours on two consecutive days. Additionally, all patients underwent physiotherapy as part of their inpatient treatment and were offered psychological counselling.
Results: One day after the last infusion, all seven patients were free of pain and full weight-bearing was possible. The side-effects of Iloprost were a headache in all patients and vomiting in two patients. Two patients relapsed, one 3 months and one 5 months after primary treatment. These two patients received a second series of infusions and were again free of pain within two days. During a mean follow-up period of 30 months all patients remained asymptomatic.
Conclusion: These preliminary results indicate that the treatment of RSD with Iloprost in combination with psychological counselling is a safe and effective treatment regime. Infusion therapy is a non-frightening procedure which may be an important factor considering the possible psychogenic etiology of RSD in children. Additional psychological counselling helps patients and their parents to develop coping strategies which may help to avoid relapses.
Radler, C., Petje G., Aigner N., Walik N., Ganger R., Grill F.
Orthopaedic Hospital Vienna-Speising, Vienna, Austria
Copyright British Editorial Society of Bone & Joint Surgery 2003
Provided by ProQuest Information and Learning Company. All rights Reserved