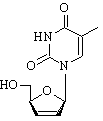
Stavudine is supplied in 15 mg, 20 mg, 30 mg and 40 mg capsules, and as an oral solution. The 30 and 40 mg capsules are most often dispensed. The 40 mg capsule (shown) is dark orange and imprinted with "BMS 1967" and "40" on one side. Dosing may vary.
"It is the only way. If you want to get newly infected people into therapy, you have to do direct-to-consumer advertising because these people would not otherwise have any reason to go to a doctor."
Jack Lamberton, a New York investment analyst, on the Bristol-Myers Squibb advertising campaign for Zerit
"Absolutely infuriating."
John Stansell, MD, on the same campaign
Also known as: d4T
Background and description. Cleared for marketing by the US Food and Drug Administration (FDA) in June 1994, stavudine became the fourth agent available for the treatment of HIV infection and the second antiretroviral released under accelerated approval. Stavudine is a nucleoside reverse transcriptase inhibitor (NRTI). Stavudine is indicated in combination with other antiretrovirals for the treatment of HIV infection. In 1999 the FDA revised stavudine's package insert to reflect its inclusion as a first-line component of combination antiretroviral HIV therapy. Bristol-Myers Squibb, the drug's manufacturer, is now investigating a sustained release formulation aimed at making stavudine a once daily drug.
Guidelines classification. The Panel on Clinical Practices for the Treatment of HIV Infection classifies stavudine as "strongly recommended" when used in combination with lamivudine (Epivir) or didanosine (Videx).
Dose. The recommended dose of stavudine is weight-dependent. For patients weighing 132 lb or more, the recommended dose is one 40 mg capsule every 12 hours. For patients weighing less than 132 lb, the recommended dose is one 30 mg capsule every 12 hours.
Food restrictions. Stavudine may be taken with or without food.
Storage. Stavudine capsules should be stored in a closed container and kept at a temperature of 59 [degrees] to 86 [degrees] F.
Side effects and toxicity. Peripheral neuropathy is the toxicity most commonly produced by stavudine; the neuropathy is dose-related and occurs more often in patients with advanced HIV disease. Drug-related neuropathy usually resolves when stavudine is promptly discontinued, although the symptoms may worsen temporarily when the drug is stopped. Elevated liver function tests are common with stavudine therapy, with clinically significant increases occurring in approximately 12% of those taking the drug. As a class, NRTIs have been implicated in damage to mitochondrial DNA and may therefore play a role in the development of metabolic and morphologic abnormalities. Although rare, life-threatening lactic acidosis has also been associated with the use of NRTIs.
Drug interactions. Stavudine and zidovudine (Retrovir) should not be combined since there is potential antagonism. Because additive neurotoxicity is possible, stavudine should not be combined with zalcitabine (Hivid).
Resistance and cross-resistance. Mutations at positions 69, 75 and 151 are associated with resistance to stavudine; however, phenotypic resistance is slow to emerge and a consistent genotypic correlate has not been identified. The mutation at position 151 is associated with resistance to the entire NRTI class. The insertion at position 69 can also lead to broad NRTI resistance. Mutants resistant to zidovudine often retain susceptibility to stavudine. Zidovudine resistance mutations have been shown to occur in antiretroviral naive patients assigned to stavudine/ didanosine therapy, although this resistance has not been substantiated phenotypically.
Clinical data. START 1 compared a regimen of stavudine/lamivudine/indinavir (Crixivan) to a regimen of zidovudine/lamivudine/indinavir in treatment naive patients. Both combinations produced similar increases in CD4 T cell count and reductions in viral load through 48 weeks. Data from other studies indicate that stavudine alone reduces viral load by approximately 0.8 log and several trials have included it in 3-drug combinations that are effective in suppressing viral load.
Patient assistance. Bristol-Myers Squibb provides a patient assistance program for those who qualify. For more information, call 800.272.4878.
COPYRIGHT 2000 The Center for AIDS: Hope & Remembrance Project
COPYRIGHT 2000 Gale Group