Definition
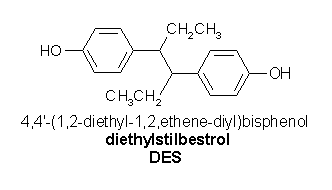
Diethylstilbestrol diphosphate is a synthetic (manufactured) form of the female hormone estrogen. Brand names for diethylstilbestrol include Stilphostrol, and is also referred to as Stilbestrol and DES.
Purpose
Diethylstilbestrol is used to relieve symptoms of advanced breast cancer that has metastasized, or spread, from the breast to other parts of the body. It is used to treat breast cancer in men and in postmenopausal women. Diethylstilbestrol also has been used to relieve symptoms of advanced cancer of the prostate in men.
Description
Diethylstilbestrol was the first form of estrogen made in the laboratory. It was prescribed to millions of women in the 1950s and 1960s to prevent miscarriage and premature birth. This use was discontinued in the 1970s, when abnormalities of the reproductive systems were found in some children of women who took the drug during pregnancy. Furthermore, daughters of women who took this drug during pregnancy are at an increased risk for developing certain types of cervical and vaginal cancers.
Diethylstilbestrol is used to relieve some symptoms of advanced breast cancer in certain men and women. The drug can interfere with the spread of cancer cells that require estrogen to grow and divide.
Diethylstilbestrol sometimes is used to relieve symptoms of advanced prostate cancer in men. This drug can lower the levels of the male hormone testosterone, which is required for the growth and division of these cancer cells. However, diethylstilbestrol can cause severe side effects in men, including breast enlargement and increased risk of heart disease and blood clots. Thus, it is no longer widely used for the treatment of prostate cancer.
Recommended dosage
Diethylstilbestrol usually is given as a pill, which should be taken at the same time each day. The dosage varies depending on body weight and the type of cancer that is being treated. For breast cancer, the dose is 15 mg per day.
For inoperable prostate cancer, the dose is 50 mg three times a day and can be increased up to 200 mg or more three times a day. Maximum dose is 1 gram a day. For the treatment of prostate cancer, diethylstilbestrol can also be injected slowly into a vein. The dosage may be as high as 1 gram per day for five or more days. The dosage then may be lowered to 250-500 mg once or twice per week.
Precautions
Diethylstilbestrol can cause serious birth defects in humans. Children of women who take diethylstilbestrol (DES) during pregnancy may develop reproductive system abnormalities at puberty, and daughters are at an increased risk for developing vaginal cancer. Therefore, this drug should not be taken by pregnant women, or by either the man or the woman at the time of conception. Women should not breast-feed infants while taking this drug, since estrogens pass into the breast milk.
Diethylstilbestrol may not be indicated, or should be used with caution, for individuals whose medical histories include any of the following:
- heart, kidney, or liver damage
- disease of the gallbladder or gallstones
- inflammation of the pancreas
- bone or uterine cancer
- fibroid tumors of the uterus
- unusual vaginal bleeding
- endometriosis (uterine cells in the ovaries or other pelvic organs)
- high cholesterol
- blood clots or circulatory problems in males
Side effects
Diethylstilbestrol affects normal cells as well as cancer cells, so side effects can occur with this medicine. The side effects associated with diethylstilbestrol usually are mild and temporary. Common side effects include:
- enlargement of the breasts
- breast tenderness
- decreased sexual desire
- voice changes
- swelling of the feet and lower legs
- fluid retention
- weight gain
Less common side effects of diethylstilbestrol include:
- nausea and vomiting during the first few weeks of treatment
- changes in vaginal bleeding
- loss of bladder control
- lumps or discharge from the breasts
- stomach, side, or abdominal pain
- yellow skin or eyes
Taking the medicine with food may reduce or prevent nausea.
Rarely, diethylstilbestrol results in the formation of blood clots in the legs or in the lungs. This primarily affects men who are receiving high-dosage treatment for breast or prostate cancers. Symptoms of blood clots include:
- pain, redness, or swelling in the calf
- weakness or tingling in an arm or leg
- faintness
- sudden severe headache
- vision changes
- shortness of breath
- chest pain
- coughing up blood (hemoptysis)
Interactions
Medicines that may adversely affect the liver when taken along with diethylstilbestrol include:
- acetaminophen (as in Tylenol; long-term or high-dose usage)
- amiodarone (Cordarone)
- anabolic steroids (such as nandrolone, oxandrolone, oxymetholone, stanozolol)
- androgens (male hormones)
- antithyroid drugs that are used to treat an overactive thyroid
- birth control pills containing estrogen
- carbamazepine (Tegretol)
- carmustine (BiCNU)
- chloroquine (Aralen)
- dantrolene (Dantrium)
- daunorubicin (Cerubidine)
- disulfiram (Antabuse)
- divalproex (Depakote)
- etretinate (Tegison)
- gold salts to treat arthritis
- hydroxychloroquine (Plaquenil)
- isoniazid
- medicines to treat infections
- mercaptopurine (Purinethol)
- methotrexate (Mexate)
- methyldopa (Aldomet)
- naltrexone (Trexan; long-term or high-dose usage))
- phenothiazines
- phenytoin (Dilantin)
- plicamycin (Mithracin)
- valproic acid (Depakene)
In addition, diethylstilbestrol and other estrogens can prevent cyclosporine (Sandimmune) from being removed from the body, leading to possible kidney or liver problems. Protease inhibitors such as ritonavir (Norvir) may reduce the activity of diethylstilbestrol.
KEY TERMS
- Estrogen
- Female sex hormone.
- Hormone
- Substance produced by the body to regulate the activity of a tissue or organ.
- Metastasis
- Spread of cancer from its point of origin to other parts of the body.
- Prostate
- Gland in males that surrounds the urine tube (urethra) at the base of the bladder.
- Testosterone
- Principal male sex hormone.