SAN FRANCISCO -- Of patients with self-described or physician-diagnosed tension or stress headache, 88% actually turned out to meet International Headache Society diagnostic criteria for migraine or probable migraine in a large study. Dr. Robert Kaniecki reported at the annual meeting of the American Academy of Neurology.
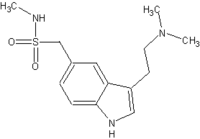
The validity of the revised diagnoses was underscored by the finding that 100 mg of sumatriptan proved significantly more effective than placebo for the relief of these headaches in a double-blind randomized trial. Although the triptans are a mainstay of abortive therapy in migraine attacks, they are not considered effective in relieving tension/stress headache attacks, said Dr. Kaniecki of the University of Pittsburgh.
He reported on 423 patients enrolled in a 54-site study conducted in the United States and Canada. All of the patients presented with a history of self-described or previous physician-diagnosed tension/stress headache. But when the neurologists, primary care physicians, and headache specialists participating in the study evaluated the patients' symptoms using International Headache Society criteria, they determined that 84% of the patients met diagnostic criteria for migraine with or without aura, and another 4% met IHS criteria for probable migraine.
Also, 19% of the patients met diagnostic criteria for dual headaches (migraine plus tension-type headache). Only 12% of the study population met criteria for tension headache only.
Patients with a revised migraine-only diagnosis said they previously believed they suffered from tension/stress headache for several reasons: Their episodes tended to occur when stressed or while relaxing after being stressed, the headaches had a pressing or head-tightening quality, and the headaches entailed neck and/or shoulder pain. It's not widely appreciated by patients that neck pain can be a feature of migraine, Dr. Kaniecki explained.
Overall, 96% of study participants reported using nonsteroidal anti-inflammatory agents and other over-the-counter medications to treat what they had believed were tension headaches; only 15% said they were satisfied with their OTC therapy.
Of the patients who met IHS criteria for migraine, 253 completed the double-blind, randomized treatment phase of the study. Two hours after treatment of an in-progress moderate or severe headache, 49% of patients given 100 mg of sumatriptan reported no or mild pain, a significantly greater response rate than the 38% among the placebo group. At 4 hours, the response rate was 64% with sumatriptan and 45% with placebo.
An intriguing finding requiring additional study was a marked split in sumatriptan responsiveness depending upon whether or not a patient had moderate to severe neck pain at the time of treatment. Among the 92 migraine patients with no or mild neck pain, 74% experienced headache relief within 4 hours after taking sumatriptan, compared with 37% on placebo. In contrast, among the group reporting moderate to severe neck pain, sumatriptan wasn't significantly different than placebo, he said.
Dr. Kaniecki's study was sponsored by GlaxoSmithKline, a company he has served as a paid consultant.
BY BRUCE JANCIN
Denver Bureau
COPYRIGHT 2004 International Medical News Group
COPYRIGHT 2004 Gale Group