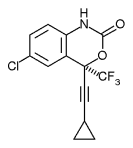
DuPont Pharmaceuticals has received Food and Drug Administration approval for it new AIDS drug, Sustiva(r) (efavrienz), Sustiva(r) is the third non-nucleoside reverse transcriptase inhibitor. Experts says it is more potent than the other two non-nucleoside drugs, delavirdine and nevirapine, If t is as powerful as some claim Sustiva may simplify a drug regimen by lowering the number pills patients take on a daily basis to reduce their viral load. Results from clinical trials indicate Sustiva reduceplasma viral RNA to less than 400 copies/mL in antiretroviral-naive and treatment-experienced patients in two-, three-and four-drug combinations.
Meeting the FDA's fast-track criteria, Sustiva(r) was administered with AZT and 3TC, and was shown to be more effective in a larger number of patients than the standard regimen, consisting of indinavir, AZT and 3TC. In addition, more subjects from indinavir, AZT and 3TC control arm discontinued theraphy due to adverse events. Criteria for efficacy included plasma viral levels and CD4 cell counts in controlled studies in a period of 24 weeks.
Adverse events related to Sustiva(r) are generally well tolerated and transient. The most notable ones are symptoms associated with the nervous system, such as dizziness, insomnia, somnolence, impaired concentration and abnormal dreaming. These symptoms tend to resolve within a few weeks into treatment. Sustiva(r) is contraindicated in pregnant women because of teratogenic effects seen in treated animals. Patients should also be warned of avoiding fatty meals with Sustiva(r) due to increased absorption of the durg in the presence of lipids.
COPYRIGHT 1998 Lebhar-Friedman, Inc.
COPYRIGHT 2000 Gale Group