Before the introduction of iodized salt in 1922, in certain areas of Switzerland almost every school child had a greatly enlarged thyroid gland, or goiter. Up to 30 percent of the country's young men were rejected for universal military service because of large goiters, although those with small ones were taken. About 1 in every 200 Swiss in these areas had cretinism, a condition characterized by dwarfism and mental retardation.
As early as 1850, cretinism was ascribed to severe iodine deficiency during gestation. Yet, as late as 1930, some Swiss medical experts remained unconvinced that lack of dietary iodine caused goiters.
The iodine content of soil and water was not uniform throughout Switzerland. A 1907 study found that laboratory rats fed water and milk from eight different villages for more than a year developed large goiters when their diet came from areas with insufficient iodine. Rats fed water supplemented by iodide did not develop goiters. Nevertheless, some scientists maintained that unknown agents found in drinking water and food, rather than lack of iodine, were responsible for goiter development.
This argument was refuted by history. After 1922, goiter rapidly disappeared in Switzerland in newborns and children, more slowly in the draftees, and incompletely in elderly adults.
Early in this century, the United States had pockets of goiter in areas where soil was iodine-deficient, such as in the Midwest and the Great Lakes area. Goiters were the chief medical reason for rejecting recruits for Army service during World War I. In 1924, the Michigan Department of Health permitted the sale of salt with the addition of potassium iodide, a soluble form of iodine. Thereafter, iodized salt was distributed nationwide (although iodization is not mandatory in this country).
In response to the 1958 Food Additives Amendment to the Federal Food, Drug, and Cosmetic Act, FDA evaluated the safe use of potassium iodide as a source of dietary iodine. In 1972, FDA affirmed its use at 0.01 percent in table salt as GRAS (generally recognized as safe).
Iodized salt, along with an excellent food distribution system that makes seafood and foods grown in iodine-rich soils available to all, has made goiter due to insufficient dietary iodine a thing of the past in the United States and all developed countries. (See accompanying article.)
Severe iodine shortages still exist in other parts of the world because iodine is distributed unevenly in the environment. The World Health Organization estimates that as many as 200 million people in developing countries have goiters due to iodine deficiency.
Lack of dietary iodine is not the only cause of goiter. The thyroid may enlarge when it is underactive or overactive, inflamed or infected, or when a congenital condition prevents it from manufacturing thyroid hormones. Benign or cancerous lumps that develop in the gland may make it appear enlarged. Appropriate treatment by medication or surgery will reduce the enlarged thyroid gland to normal size in almost all cases.
The Thyroid Gland
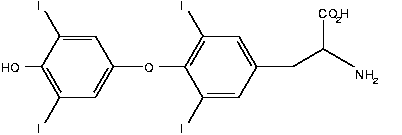
The thyroid gland is a small butterfly-shaped gland that wraps around the wind-pipe just below the Adam's apple. The thyroid takes dietary iodine from the blood and uses it to make iodine-containing hormones called thyroxine ([T.sub.4]) and triiodothyronine ([T.sub.3]). The thyroid stores these hormones until needed. Then the thyroid releases the hormones into the blood-stream and they travel to all parts of the body, affecting almost every cell and most organs, including the heart, brain, liver, kidneys, and skin.
These hormones have many important functions. Because they regulate the rate at which calories are burned, they may cause weight loss or gain. They help slow down or quicken the heartbeat and raise or lower body temperature. They influence the rate at which food moves through the digestive tract, the way muscles contract, and the rate at which dying cells are replaced.
A healthy thyroid gland dispenses just the right amount of hormone to meet the body's metabolic needs. But if the gland becomes diseased, it may produce too little thyroxine (hypothyroidism) or too much (hyperthyroidism). The thyroid is under partial control of the pituitary--a pea-sized gland at the base of the brain. When the pituitary senses a low level of thyroxine in the blood, it secretes thyroid-stimulating hormone (TSH), which stimulates the thyroid gland to make more hormone. Conversely, if the pituitary senses too much hormone, it will secrete less TSH, and thyroxine levels fall.
Hypothyroidism
Of the 6 million to 7 million Americans who have underactive thyroid glands, probably half don't seek medical treatment because they don't know they have a problem. They are likely to accept their symptoms-fatigue, muscle weakness, dry skin, hair loss, depression, cold intolerance, constipation--as signs of normal aging. Hypothyroidism occurs four times more often in women than in men, most frequently in middle-aged women between the ages of 35 and 60, and in the elderly. There is a strong tendency for all kinds of thyroid disorders to run in families.
The most common cause of hypothyroidism is a chronic, progressive condition called Hashimoto's disease. Hashimoto's disease is an autoimmune disorder, in which the body's immune system produces antibodies that attack its own tissues. As the antibodies slowly destroy the thyroid gland, it cannot manufacture sufficient amounts of thyroid hormones for proper body functioning. This causes the pituitary to secrete more TSH, thus forcing the thyroid gland to work harder, resulting in an enlarged gland, or goiter.
People who have received x-ray treatment for cancers of the head and neck may eventually become hypothyroid, if the thyroid gland was inadvertently exposed to radiation. Lower-dose radiation commonly used from 1920 to 1960 to treat children for acne, enlarged thymus gland, scalp tingworm, birthmarks, enlarged tonsils, and adenoids also increases the risk of hypothyroidism. (As many as 7 percent of children and adolescents who had such radiation therapy developed thyroid cancer 10 to 40 years later.) Goiter is not usually present in these types of hypothyroidism, because the thyroid gland has shrunk.
The thyroid gland can be infected by viruses and bacteria that cause it to become temporarily underactive, but these infections can be treated with medications and do not usually result in permanent hypothyroidism.
One in every 4,000 to 5,000 babies is born hypothyroid in the United States. This occurs when babies are born without a thyroid gland or with one that functions poorly. Some babies inherit disorders that prevent their thyroid glands from making sufficient thyroid hormone. Because cretinism in babies is irreversible if not treated with thyroid hormone as soon as possible after birth, all newborns in the United States are screened for hypothyroidism.
When hypothyroidism is suspected, the doctor bases the diagnosis on the patient's symptoms and blood tests that measure the amount of thyroxine and TSH circulating in the blood. A high level of TSH, which is a sign that the pituitary detects thyroxine deficiency, and a low level of thyroxine are indications that the thyroid is underactive.
Treatment of Hashimoto's disease has come a long way from the days when thyroid glands from sheep, pigs or cows were sauteed and eaten. Beginning in the 1880s, liquid extracts of these glands were given intravenously or orally, then manufactured into tablets, which were in use until the 1960s. Today, synthetic thyroxine developed in the early 1970s is the mainstay of thyroid hormone replacement. Drugs most commonly used are Synthroid or Levothroid (levothyroxine). People with Hashimoto's disease must take these medications for the rest of their lives, because the thyroid gland has been permanently damaged.
One complication of severe and long-standing hypothyroidism is called myxedema. Persons with this disorder become very cold and drowsy, and may eventually lapse into a coma. Uncommon in tropical climates, this rare condition may be brought on by cold weather, infection, or the use of some drags, especially sedatives. About 50 to 60 percent of people with hypothyrnid coma die. FDA approved Triostat (liothyronine sodium), an intravenous thyroid hormone replacement drug for the treatment of myxedema coma and pre-coma, in 1991.
Hyperthyroidism
Another autoimmune disorder that affects the thyroid gland is Graves' disease, the most common type of hyperthyroidism. Antibodies do not destroy the thyroid gland, as in Hashimoto's disease, but stimulate it to produce too much thyroid hormone.
No one knows what causes Graves' disease. Sex hormones play a part; the disease is seven times more common in women than men, and frequently begins after hormonal changes, such as pregnancy. Stressful situations, such as a death in the family, appear to trigger the autoimmune reaction. The tendency to develop the disease is inherited. Graves' disease afflicts both President and Mrs. Bush.
Hyperthyroidism may also result when one or more nodules or lumps in the thyroid gland become overactive--for no known reason--and secrete too much hormone. Another form of hyperthyroidism, called subacute thyroiditis, may arise following a vital infection, such as mumps, measles, or even a cold. The inflamed thyroid leaks out excessive amounts of thyroid hormones into the bloodstream. This disease is temporary and usually treated by aspirin, propranolol (Inderal) or corticosteroids.
Symptoms of an overactive thyroid include goiter, weight loss despite increased food intake, rapid heartbeat, scant menstrual periods, warm, moist skin, finger tremor, sensitivity to heat, fatigue, muscle weakness, and frequent bowel movements.
Any type of hyperthyroidism may cause an individual to develop a staring look, because increased levels of thyroid hormone cause elevation of the upper eyelids. However, only in Graves' disease do the muscles and fat around and behind the eyes become so inflamed that eyes are pushed forward, bulging out of theft sockets. The technical term for this condition is exophthalmos.
Blood tests will confirm hyperthyroidism when the levels of thyroxine and triiodothyronine circulating in the blood are abnormally high and when TSH concentrations are low. If the diagnosis needs further confirmation, a radioactive image, or scan, of the thyroid is made. The patient swallows a small amount of radioactive iodine in water or in capsule form. After a 24-hour wait, a picture of the thyroid gland is taken. The scan can tell whether the whole gland is overactive, as in Graves' disease, or whether an overactive nodule is causing the problem.
Treatment of Graves' disease has improved a great deal since the 1800s, when the only therapy was rest and sedation and the death rate was 50 percent. The first breakthrough in the treatment of this disorder came in the 1940s, when physician Edwin Astwood and colleagues at the New England Medical Center in Boston discovered drugs that blocked the ability of the thyroid gland to use iodine in making thyroid hormone. These anti-thyroid drugs, propylthiouracil (PTU) and methimazole (Tapazole), are used when prompt control of hyperthyroidism is necessary, especially in children. Elderly patients with heart disease or arrhythmias can be safely treated temporarily with these drugs.
Anti-thyroid drugs offer several advantages over the most commonly used treatment, radioactive iodine. They do not generally cause permanent thyroid damage, are less expensive, and also may not need to be given indefinitely. However, though they control a hyperactive thyroid gland, they are not necessarily a cure. After long-term drug treatment, only 35 percent of patients experience remission, according to the American Thyroid Association. They may also cause adverse reactions-- rashes, hives, fever, joint pains-in 1 out of 20 who take them. One serious complication is that the drugs may decrease the number of neutrophils (white blood cells) in the blood, causing a lowered resistance to infection. Rarely, all these white cells disappear from the blood, producing a potentially fatal condition known as agranulocytosis.
Many physicians prefer to treat patients who have Graves' disease or overactive nodules in their thyroid glands with radioactive iodine. The patient swallows the radioactive iodine in capsule or liquid form. The radioactive iodine passes from the stomach into the bloodstream and collects in the thyroid gland, where its radioactivity damages the thyroid cells. Within 48 hours or so, radioactive iodine disappears from the body, either eliminated in the urine or else transformed by decay into a nonradioactive state. Most hyperthyroid patients recover normal thyroid gland function, or even become hypothyroid, three to six months after radioactive iodine treatment. Those who remain hyperthyroid may need a second or even a third dose.
Removing most of the thyroid gland surgically--a delicate procedure that needs to be done by a thyroid surgery specialist-results in a permanent cure for overactivity. Many physicians recommend surgery for patients under 21, for those allergic to anti-thyroid drugs, and for patients with very large goiters.
Nevertheless, surgery is done much less frequently than it used to be. Leonard Wartofsky, M.D., chief of the Endocrine-Metabolic Service of the Department of Medicine at Walter Reed Army Medical Center in Washington, D.C., tells of a survey of physician members of the American Thyroid Association, asking what therapy they would advise for a typical case of hyperthyroidism. Seventy percent of the doctors recommended radioactive iodine, 29 percent anti-thyroid drugs, and 1 percent surgery. "Twenty years ago it would have been 20 percent for surgery ," Wartofsky said. "Radioactive iodine is easier and much more readily acceptable by patients."
Commonly, radioactive iodine and surgery eventually result in an underactive thyroid, treatable by a thyroid hormone tablet once a day. This is a less common occurrence with anti-thyroid drug therapy.
After any one of the three treatments, physicians may also prescribe beta-blocking drugs, such as Inderal or Tenorrain (atenolol), that block the action of circulating thyroid hormone on body tissues. They are used to slow down the heart rate, lessen nervousness, and generally reduce symptoms until the chosen treatment has had a chance to take effect.
"It's necessary for people with thyroid conditions to see a doctor for the rest of their lives because thyroid conditions evolve and change with time," says Wartofsky. "Whether we' re talking about hyper- or hypothyroidism, there's always the possibility of progression or change. Patients should see their doctors on a once-a-year basis. I'd even settle for once every two or three years in some circumstances. Medications have to get renewed anyway, and just touching base with their physicians is a good idea."
Recognizing symptoms of thyroid disease and seeking medical help to ease them are the first steps toward a healthy life for people with thyroid disorders. Whether correcting a thyroid problem requires specialized surgery for overactivity or a pill for underactivity, thyroid hormone balance can be achieved.
Evelyn Zamula is a freelance writer in Potomac, Md.
Some Iodine-Rich Foods
Iodine deficiency is no longer a problem in the United States, with virtually all Americans getting enough of the substance in their diets. The Recommended Daily Allowance (RDA) for iodine for adolescents and adults is 150 micrograms a day (1,000 micrograms = 1 milligram) and 200 micrograms a day for pregnant women. Most Americans consume more than the RDA, but are usually unaffected by excess iodine unless they have underlying thyroid disease.
The following chart gives examples of foods relatively high in iodine.
COPYRIGHT 1992 U.S. Government Printing Office
COPYRIGHT 2004 Gale Group