Difference in [PO.sub.2] Between the Right and Left Pulmonary Veins
Background: The right-to-left shunt at the atrial level is responsible for arterial hypoxemia in patients with atrial septal defect.
Objectives: This study investigated the mechanism of arterial hypoxemia in patients with atrial septal defect by measuring the [PO.sub.2] in both the right and left upper pulmonary veins. Subjects and method: We prospectively measured the [PO.sub.2] in the femoral artery and the right and left upper pulmonary veins during cardiac catheterization in 13 adults (median age, 53 years) and 7 children (median age, 7 years) with secundum atrial septal defect. The adults and children were studied consecutively. Contrast echocardiography was performed to evaluate right-to-left shunt in all adults.
Results: Among the children, there were no patients showing arterial hypoxemia, and there was no difference in the [PO.sub.2] ([+ or -] SD) between the right and left upper pulmonary veins (right, 100 [+ or -] 3.8 mm Hg vs left, 100 [+ or -] 7.8 mm Hg; p = 0.92). However, arterial hypoxemia was present in 11 of the 13 adult patients, although contrast echocardiography showed more than a moderate degree of right-to-left shunt in only four adults. The [PO.sub.2] was lower in the left upper pulmonary vein than it was in the right upper pulmonary vein in all adult patients (right, 91.6 [+ or -] 13.8 mm Hg vs left, 73.0 [+ or -] 11.5 mm Hg; p [is less than] 0.0001).
Conclusion: The [PO.sub.2] was lower in the left upper pulmonary vein than it was in the right upper pulmonary vein in adults with atrial septal defect. Care must be taken in measuring pulmonary blood flow if the [PO.sub.2] in the left upper pulmonary vein is low enough to influence oxygen content. The decreased [PO.sub.2] in the left upper pulmonary vein may contribute to arterial hypoxemia in addition to right-to-left shunt at the atrial level in adults with atrial septal defect.
(CHEST 1999; 115:679-683)
Key words: atrial septal defect; intracardiac shunt; [PO.sub.2]; pulmonary vein
Abbreviations: ASD = atrial septal defect; VC = vital capacity; V/Q = ventilation/perfusion ratio
Right-to-left shunt at the atrial level is responsible for decreased saturation of arterial blood in patients with patent foramen ovale and increased venous pressure[1,2] or in patients with large atrial septal defect (ASD) and severe tricuspid regurgitation.[3] In either situation, there is an assumption that saturation of the right and left pulmonary veins is equally high if arterial hypoxemia is caused only by this reverse shunt. To our knowledge, there has been no report of a comparison between the right and left pulmonary veins regarding [PO.sub.2].
In the present study, we investigated whether [PO.sub.2] is equal in the right and left upper pulmonary veins in both adults and children with ASD in order to look for a cause of arterial hypoxemia that is other than the right-to-left shunt.
MATERIALS AND METHODS
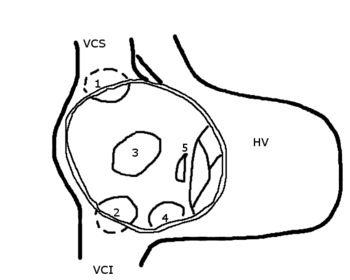
Subjects consisted of 13 adults (median age, 53 years; range, 40 to 78 years) and 7 children (median age, 7 years; range, 5 to 13 years) with secundum ASD. The adults and children were studied consecutively. Using an NIH catheter, cardiac catheterization was performed from the femoral approach in all patients. The catheters were easily advanced into the right and left upper pulmonary veins through the ASD. These upper pulmonary veins were identified as right or left when the catheter was superior to the cardiac silhouette and rightward and leftward, respectively. Blood was drawn from both the right and left upper pulmonary veins as well as from other cardiac chambers before the injection of contrast medium: 5 mL of blood in adults and 2 mL in children. Because an NIH catheter has six side holes about 2 cm proximal to the closed end, we were very careful about keeping the sampling position in the pulmonary veins at a distance from the cardiac silhouette such that it would not be contaminated by blood from the left atrium. If the hemoglobin concentration of any blood sample differed [is greater than] 0.3 g/dL from other blood samples, it was discarded as a sampling error. [PO.sub.2] and [PCO.sub.2] were measured (Chiron 288 blood gas system; Chiron Diagnostics Corp; Medfield, MA), and the oxygen content was then calculated mathematically.
Using hand-agitated 5% dextrose in water from an antecubital vein, contrast echocardiography was done during normal breathing in all adults (Table 1). We used the transesophageal technique in 11 adult patients to confirm whether the right-to-left shunt was directed toward the left upper pulmonary vein. The degree of right-to-left shunt on contrast echocardiography was classified as follows: severe if the bubble was seen entirely in the left atrium; mild if the bubble was slightly in the left atrium; and moderate if in between. Ventilatory function tests were done in all adult patients except for one, and pulmonary ventilation by [sup.13]Xe gas and perfusion scan by [sup.99m]Tc macroaggregated albumin were done in 10 adult patients. Statistical analysis was done using Student's paired t test. None of the patients had a history of thoracotomy.
(*) M = male; F = female; Af = atrial fibrillation; SR = sinus rhythm; CTR = cardiothoracic ratio; R-Shunt = right-to-left shunt; - = none; 1+ = mild; 2+ = moderate; 3+ = severe; TR = tricuspid regurgitation; Qp = pulmonary flow using the right upper pulmonary vein as a reference chamber; Qs = systemic flow; PAR = pulmonary arterior resistance; Scinti = V/Q scan of the lung; m = V/Q mismatch; n = normal; (-) = not done; % VC = percent vital capacity; Rt PV = right pulmonary vein; Lt PV = left pulmonary vein; FA = femoral artery.
RESULTS
In the children, there was no arterial hypoxemia and no difference in [PO.sub.2] ([+ or -] SD) between the right and left upper pulmonary veins (right, 100 [+ or -] 3.8 mm Hg vs left, 100 [+ or -] 7.8 mm Hg; p = 0.92; Fig 1). In the adults, arterial hypoxemia was present in 11 patients, if arterial [PO.sub.2] [is greater than] 80 mm Hg is considered normal, and [PO.sub.2] ([+ or -] SD) was lower in the left upper pulmonary vein than in the right upper pulmonary vein (right, 91.6 [+ or -] 13.8 mm Hg vs left, 73.0 [+ or -] 11.5 mm Hg; p [is less than] 0.0001; Fig 2). The [PCO.sub.2] in the left upper pulmonary vein was statistically greater than that of the right upper pulmonary vein (right, 37.4 [+ or -] 5.4 mm Hg vs left, 39.4 [+ or -] 6.7 mm Hg; p = 0.03; Fig 3). The difference in oxygen content between the right and left upper pulmonary veins ranged from 2 to 34 mL/L, with the difference being [is greater than] 5 mL/L in 11 adult patients (Fig 4). The pulmonary blood flow in patient 1 was measured as 4.1 L/min when only the oxygen content of the right pulmonary upper vein was used, whereas it was measured as 8.3 L/min when the averages of both the fight and left upper pulmonary veins were used. Contrast echocardiography showed that the right-to-left shunt was mild in eight adult patients and that there was no shunt in one patient. During contrast transesophageal echocardiography, no bubble was seen in the left upper pulmonary veins of all 11 adult patients. In two adult patients who had severe reverse right-to-left shunt, acquired cyanosis was seen, and severe tricuspid regurgitation and atrial fibrillation were observed to be present (patients 1 and 4). The mean cardiothoracic ratio was 62%. Both the [FEV.sub.1]/FVC ratio and the percent vital capacity (VC) were normal in five adult patients, and either the [FEV.sub.1]/VC ratio or the percent VC was abnormal in seven adult patients (Fig 5). Five adult patients showed decreased perfusion in the left lung, and one adult patient showed decreased perfusion in the fight lung. A ventilation/perfusion (V/Q) mismatch was present in the left lung in four adult patients (patients 1, 2, 4, and 12) and in the fight lung in one adult patient (patient 3). There was one adult patient whose perfusion and ventilation were equally disturbed with no V/Q mismatch (patient 9).
[Figures 1-5 ILLUSTRATION OMITTED]
DISCUSSION
Patients with ASD may become cyanotic with increasing age. This is caused by a reverse shunt through the defect, which is caused by progressive tricuspid regurgitation, which, in turn, is due to pulmonary hypertension. A jet originating from tricuspid regurgitation can cross the ASD, resulting in a right-to-left shunt.[3]
Performing contrast echocardiography by using hand-agitated 5% dextrose in water from peripheral veins, is the most sensitive method for looking for the reverse shunt. We performed contrast echocardiography under normal breathing so as not to enlarge the reverse shunt by straining. In the present study, although the degree of right-to-left shunt was more than moderate in only 4 of the 13 adult patients, arterial hypoxemia was present in 11 adult patients, if arterial [PO.sub.2] of more than 80 mm Hg is considered normal. Therefore, the degree of arterial hypoxemia seen in the adult group cannot be completely explained by this reverse shunt.
[PO.sub.2] was lower in the left upper pulmonary vein than in the right pulmonary vein in all of the adult patients, but there was no difference between the fight and left pulmonary veins in the children. Therefore, this difference is not congenital but acquired. It is not clear whether this is specific to adult ASD patients, because drawing blood from both pulmonary veins is possible only in the presence of ASD or patent foramen ovale. Hypoxemia in the left upper pulmonary vein significantly contributed to arterial hypoxemia in the adult group, because the difference in oxygen saturation between the fight and left upper pulmonary veins may derive from the right-to-left shunt being directed to the left upper pulmonary vein. However, because the transesophageal contrast echocardiography, which can visualize the left upper pulmonary vein easily and was performed in 11 of the 13 adult patients, did not show any bubble toward the left upper pulmonary vein during right-to-left shunting in any patient, this possibility is unlikely.
As patients with ASD age, the main pulmonary artery may enlarge and compress the left bronchus, which is progressing pulmonary vascular disease. Pulmonary vascular resistance was [is greater than] 4 Wood units in 5 of the 13 adult patients. In addition, the increase in heart volume of the left thoracic cavity may collapse that part of the lung, resulting in V/Q mismatch. V/Q mismatch was present in 5 of the 10 adult patients undergoing the pulmonary V/Q scan: in the left lung in four patients, and in the fight lung in one patient.
The result of ventilatory function tests in the present study showed that either [FEV.sub.1]/FVC or percentage of VC was abnormal in seven patients, reflecting the above speculation. Hypoxemia in the left upper pulmonary vein was present even in the remaining five patients with normal pulmonary function tests whose cardiothoracic ratio was rather small in comparison with that of patients with abnormal pulmonary function tests. Accordingly, the difference in [PO.sub.2] between the fight and left pulmonary veins cannot be completely explained by this mismatch.
During cardiopulmonary bypass, some blood returns to the left atrium while the aorta is cross-clamped.[4] An abundant network of collaterals has been observed between the bronchial vein and the pulmonary vein in normal lungs obtained at autopsy.[5] In patients with inflammatory pleuritis, neovascularization can develop from the internal thoracic artery and intercostal arteries and drain into pulmonary veins.[6] Pulmonary veins are also reported as a draining chamber for collaterals derived from portal hypertension.[7,8] Most of the collaterals reported so far were located in the left pulmonary vein.[9] In the present study, the pulmonary scintigram showed hypoperfusion of the left lung in 5 of the 10 adult patients undergoing pulmonary ventilation and the perfusion scan. This decreased pulmonary blood flow in the left lung may change bronchial circulation after an increase in pulmonary blood flow for a long period of time. The deoxygenated blood, after passing through the capillary phase from the bronchial artery or intercostal artery, might drain into the left pulmonary vein more often than into the fight vein.
The difference in [PO.sub.2] between the right and left upper pulmonary veins caused the difference in the calculation of oxygen content to range from 3 to 34 mL. This difference in oxygen content was sometimes severe enough to influence the measurement of pulmonary blood flow but was often insignificant because the relationship between [PO.sub.2] and saturation is not linear but sigmoid. When the shunt ratio is measured using oximetry in patients with ASD, blood drawn from either pulmonary vein is used as a reference.[10] This is based on the assumption that the oxygen contents in the right and left pulmonary veins are equal. In the first patient who had severely disturbed pulmonary function, presumably due to increased heart volume and combined pulmonary vascular disease, the calculated pulmonary blood flow was two times greater when the average of the fight and left upper pulmonary veins was used as a reference than when only the fight upper pulmonary vein was used. The use of average oxygen contents is also based on the assumption that both the fight and left pulmonary flows are equal, which was not proved to be correct.
Care must be taken in measuring an intracardiac shunt in patients with ASD if only the fight upper pulmonary vein is used as a reference. In addition to right-to-left shunt at the atrial level, decreased [PO.sub.2] in the left upper pulmonary vein may contribute to decreased arterial [PO.sub.2].
Study Limitations
We did not offer a very clear explanation as to the cause of this difference; we simply described the phenomenon. Even during thoracic surgery, it is impossible to take blood samples from both pulmonary veins simultaneously when the fraction of inspired oxygen is 20% of the blood. Further study is necessary but can be done only in the presence of an ASD or patent foramen ovale.
ACKNOWLEDGMENT: We acknowledge the helpful advice and comment of Professor Peter Harris, formerly of the Royal Brompton and National Heart Hospital, London, England.
REFERENCES
[1] Remy-Jardin M, Remy J, Wallaert B. Right-to-left shunting through a patent foramen ovale without pulmonary hypertension. Chest 1990; 97:1250-1252
[2] Dear WE, Chen P, Barasch E. Sixty-eight-year-old woman with intermittent hypoxemia. Circulation 1995; 91:2284-2289
[3] Kai H, Koyanagi S, Hirooka Y, et al. Right-to-left shunt across atrial septal defect related to tricuspid regurgitation: assessment by transesophageal Doppler echocardiography. Am Heart J 1994; 127:578-584
[4] Baile EM, Ling H, Heyworth JR, et al. Bronchopulmonary anastomotic and noncoronary collateral blood flow in humans during cardiopulmonary bypass. Chest 1985; 87:749-754
[5] Murata K, Itoh H, Todo G, et al. Bronchial venous plexus and its communication with pulmonary circulation. Invest Radiol 1986; 21:24-30
[6] Chino M, Kawaguchi T, Sakai T, et al. Intercostal-to-pulmonary arterial anastomosis, complicated by high-output heart failure: case report. Angiology 1991; 42:256-260
[7] Sano A, Kuroda Y, Moriyasu F, et al. Porto-pulmonary venous anastomosis in portal hypertension demonstrated by percutaneous transhepatic cine-portography. Radiology 1982; 144: 479-484
[8] Sano A, Nishizawa S, Sasai K, et al. Contrast echocardiography in detection of portopulmonary venous anastomosis. Am J Roentgenol 1984; 142:137-140
[9] Sano A, Nishizawa S, Sasai K, et al. Demonstration of porto-pulmonary venous anastomosis by cine-portography and contrast echocardiography [in Japanese]. Nihonrinshougazouigakuzasshi 1983; 2:402-412
[10] Grossman W, Baim DS. Cardiac catheterization, angiography, and intervention. 4th ed. Philadelphia, PA: Lea & Febiger, 1991; 166
(*) From the Department of Cardiology (Drs. Iga, Izumi, Kitaguchi, Himura, Gen, and Konishi), and the Department of Pediatrics (Dr. Matsumura), Tenri Hospital, Tenri City, Japan.
Manuscript received June 6, 1998; revision accepted October 13, 1998.
Correspondence to: Takashi Konishi MD, Department of Cardiology, Tenri Hospital, 200 Mishimacho, Tenri City, 632-8552 Japan; e-mail: igakan@kcn.ne.jp
COPYRIGHT 1999 American College of Chest Physicians
COPYRIGHT 2000 Gale Group