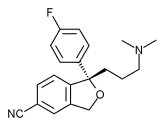
The Food and Drug Administration has approved escitalopram oxalate for the treatment of generalized anxiety disorder.
Escitalopram (Lexapro), a selective serotonin reuptake inhibitor, was originally approved in August 2002 for the treatment of major depressive disorder.
The FDA based its approval on the results of three randomized, double-blind, placebo-controlled studies that were conducted by Forest Laboratories, maker of Lexapro.
The studies involved about 850 patients, aged 18-80 years, who had been diagnosed with generalized anxiety disorder (GAD).
Participants were given 10 mg/day for 4 weeks and then flexibly dosed to a maximum of 20 mg/day. GAD symptoms were significantly improved in 68% of the patients receiving 10-20 mg daily, compared with 41% of the patients in the placebo group, as measured by the change in their scores from baseline on the Hamilton Anxiety Scale.
Patients in the escitalopram group also experienced an improvement in quality of life as measured by the Quality of Life scale when compared with patients on placebo.
By the end of 8 weeks 36% of the escitalopram patients were in remission compared with 16% of patients on placebo.
Escitalopram was well tolerated, and the most frequently reported adverse events were nausea, ejaculation disorder, insomnia, fatigue, decreased libido, and anorgasmia.
Generalized anxiety disorder is one of the most common mental illnesses in the United States, affecting approximately 4 million adults.
BY DEEANNA FRANKLIN
Senior Writer
COPYRIGHT 2004 International Medical News Group
COPYRIGHT 2004 Gale Group