Lexapro
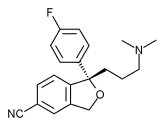
Escitalopram is a medication developed by the Danish pharmaceutical company Lundbeck, that acts as a selective serotonin reuptake inhibitor or SSRI. It is typically used as an antidepressant to treat depression associated with mood disorders although also may be used in the treatment of body dysmorphic disorder and anxiety. In the United States, the drug is marketed under the name Lexapro® by Forest Laboratories, Inc. In Canada and Europe, the drug is marketed under the name Cipralex® by Lundbeck Canada, Inc. more...
Escitalopram oxalate is derived from the drug citalopram which is made up of two mirror-image isomers, only one of which is thought to be an effective medication. Lundbeck has split the isomers apart, taken the active isomer and has licensed it as the new drug escitalopram. Escitalopram is the pure S-enantiomer (single isomer) of the racemic bicyclic phthalane derivative citalopram.
Escitalopram was released shortly before the patent for citalopram was due to expire. The expiration of a patent means other companies can legally produce cheaper generic versions. Escitalopram is subject to a new patent. Critics have argued that escitalopram, and the subsequent marketing campaign to persuade mental health professionals to prescribe it, is a cynical ploy to promote sales of a virtually identical but considerably more expensive drug. In defence of the new drug, Lundbeck has claimed that the drug has additional benefits over the older citalopram and other antidepressant drugs on the market, mainly better tolerability .
Withdrawal symptoms
A possible withdrawal symptom from Escitalopram is a type of spontaneous nerve pulse, described by some patients as a feeling of small electric shocks, which may be accompanied by dizziness. These pulses may be short in duration, only milliseconds long, affect any region of the body, and recur up to several times a minute, throughout all waking hours. They can be increased by physical activity, but are not solely linked to muscular activity.
Read more at Wikipedia.org