Abstract
With the continuing development of clinical drug resistance among bacteria, the need for new, effective agents to treat multi-drug-resistant Gram-positive infections remains important. With treatment options limited, it has become critical to identify antibiotics with novel mechanisms of activity. Several new drugs have emerged as possible therapeutic alternatives. This review focuses on agents newly introduced and FDA-approved for the treatment of skin and skin structure infections: linezolid and quinupristin/dalfopristin.
Discussion
There has been an alarming increase in the incidence of Gram-positive infections, including resistant bacteria such as methicillin-resistant Staphylococcus aureus (MRSA) and drug-resistant pneumococci. While vancomycin has been considered the drug of last defense against Gram-positive multi-drug-resistant bacteria, the late 1980s saw a rise in vancomycin-resistant bacteria, including vancomycin-resistant enterococci (VRE). More recently, strains of vancomycin-intermediate resistant Staphylococcus aureus (VISA) have been isolated (1). Gram-positive bacteria such as Staphylococcus aureus and Streptococcus pyogenes are often the cause of skin and skin structure infections, ranging from mild pyodermas to complicated infections, including post-surgical wound infections, severe carbunculosis, and erysipelas. With treatment options limited, it has become critical to identify antibiotics with novel mechanisms of activity. Several new drugs have emerged as possible therapeutic alternatives, including linezolid and quinupristin/dalfopristin.
LINEZOLID
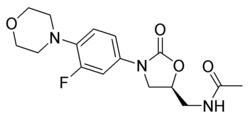
Linezolid (Fig. 1) is an oxazolidinone antibiotic, shown to be effective for nosocomial and community-acquired pneumonias, vancomycin-resistant Enterococcus faecium (VREF) infections, and skin infections due to certain staphylococcus or streptococcus species (2). The oxazolidinones are a novel class of antibiotics first discovered in 19873, the first new antibiotics to have been discovered in the past 35 years.
[FIGURE 1 OMITTED]
Mode of Action
Cellular Mechanism
Oxazolidinones, and specifically linezolid, are theorized to act by inhibiting the initiation phase of translation and thus interfering with bacterial protein synthesis (4). It is thought that-linezolid binds to the 23S portion of the 50S ribosomal subunit, preventing initiation complex formation. This early inhibition of protein synthesis is a unique mechanism and limits cross-resistance with other antimicrobial agents, since there is no pre-existing resistance mechanism in nature (4).
Pharmacokinetics
Oral bioavailability of the antibiotic in a normal host is 100%. The drug can be administered without regard to meals. Food may slightly decrease the rate of absorption, but it has no effect on the amount of the drug absorbed. The drug shows a protein binding of only 31% and a half-life of 5 to 7 hours. Linezolid administered orally at 600 mg every 12-hour dosing provides for average steady-state plasma concentrations exceeding MIC90 concentrations for staphylococci, streptococci, and enterococci. It is primarily metabolized by oxidation of the morpholine ring, which produces two inactive metabolites. Its metabolism is unaffected by the cytochrome P450 enzyme system. These kinetics do not differ in patients with mild to moderate renal or hepatic compromise. In patients over the age of 5, the 10 mg/kg/dose every 12 hours displays similar pharmacokinetic properties.
In Vitro Activity
In vitro studies have shown linezolid to be effective against many antibiotic-resistant Gram-positive organisms, including MRSA, penicillin-resistant Streptococcus pneumoniae, and VRE (5-9). Linezolid is bacteriostatic against most susceptible organisms, but has shown bactericidal activity against Clostridium perfringens, Bacteroides fragilis, and some strains of Streptococcus pneumoniae (10). In addition to its coverage of antibiotic-resistant Gram-positive organisms, it has some broad-spectrum activity against Gram-positive cocci, Gram-negative anaerobes, and some mycobacteria. It has also shown moderate in vitro inhibitory activity against Haemophilus influenzae and Moraxella catarrhalis, although it was not effective against Enterobacteriaceae and Pseudomonas aeruginosa.
Clinical Indications
Linezolid is currently FDA-approved for the treatment of various Gram-positive infections, including nosocomial and community-acquired pneumonias, complicated and uncomplicated skin and skin structure infections, and vancomycin-resistant Enterococcus infections. Head-to-head studies have been conducted comparing linezolid and standard antibiotic therapies in the treatment of skin and soft tissue infections. One study evaluated 332 adult patients with uncomplicated skin infections (cellulitis, skin abscesses, and furuncles) secondary to staphylococcus and streptococcus (11). This double blind, randomized study compared linezolid 400 mg twice daily with clarithromycin 250 mg twice daily for a course of 7-14 days. Following treatment, 91% of the linezolid-treated patients had a clinical cure, compared to 93% in the clarithromycin group. This study demonstrated linezolid to be equally as effective as clarithromycin.
Another randomized, double-blind, multicenter trial compared the efficacy and safety of linezolid, an oxazolidinone, with those of oxacillin-dicloxacillin in patients with complicated skin and soft tissue infections (12). A total of 826 hospitalized adult patients were randomized to receive linezolid (600 mg intravenously) every 12 hours, or oxacillin (2 grams intravenously) every 6 hours. Following sufficient clinical improvement, patients were switched to the respective oral agents, linezolid 600 mg orally every 12 hours, or dicloxacillin 500 mg orally every 6 hours. Primary efficacy variables were clinical cure rates in both the intent-to-treat (ITT) population and clinically evaluable (CE) patients, and microbiological success rate in microbiologically evaluable (ME) patients. Safety and tolerability were evaluated in the ITT population. Demographics and baseline characteristics were similar across treatment groups in the 819 ITT patients. In the ITT population, the clinical cure rates were 69.8% and 64.9% in the linezolid and oxacillin-dicloxacillin groups, respectively (P = 0.141 [95% confidence interval, -1.58 to 11.25]). In 298 CE linezolid-treated patients, the clinical cure rate was 88.6%, compared with a cure rate of 85.8% in 302 CE patients who received oxacillin-dicloxacillin. In 143 ME linezolid-treated patients, the microbiological success rate was 88.1%, compared with a success rate of 86.1% in 151 ME patients who received oxacillin-dicloxacillin. Both agents were well tolerated; most adverse events were of mild-to-moderate intensity. No serious drug-related adverse events were reported in the linezolid group (12).
Linezolid was also found to be as effective as vancomycin (73.2% versus 73.1%) in a study for the treatment of MRSA infections, in which the most common diagnosis was skin and soft tissue infections (13). Other studies have also supported these findings (14). In addition, a clinical success rate of 81% was noted in infections caused by vancomycin-resistant Enterococcus. There is support that treatment with linezolid may be superior to comparator antibiotics in patients with complicated skin
infections that also have comorbid conditions (15). There have been reports of development of resistance to linezolid developed in some patients with Enterococcus faecium (16).
Dosage Regimens
The recommended dosage of linezolid depends on the severity of the skin or soft tissue infection. The recommended dosage for uncomplicated infections is 400 mg every 12 hours for 10 to 14 days. For complicated infections, 600 mg twice daily either via intravenous infusion or orally is recommended. Because the absolute bioavailability after oral dosing is nearly 100%, no dosage changes are needed when switching from intravenous to oral therapy (4).
Contraindications/Cautions
Linezolid is generally well tolerated, with the most common adverse effects being diarrhea (8.3%), headache (6.5%), and nausea (6.2%) (10). Since linezolid is a non-selective, reversible inhibitor of monoamine oxidase, it may interact with serotonergic or adrenergic agents (17). Like many other antibiotics, it may cause pseudomembranous colitis, as a result of overgrowth of Clostridium difficile. Approximately 2% of patients develop thrombocytopenia, which appears to be dependent on duration of therapy. The effect is reversible; however, the manufacturer recommends monitoring patients with preexisting thrombocytopenia or patients whose treatment will exceed two weeks. No deaths related to thrombocytopenia have been reported. Linezolid has been reported to cause acute generalized exanthematous pustulosis (AGEP), which is difficult to distinguish from an underlying staphylococcus infection (31).
Conclusions
Linezolid is the first of a novel class of antibiotics called oxazolidinones. Although relatively new, linezolid shows great promise in treating a variety of Gram-positive organisms, including MRSA and VRE.
QUINUPRISTIN/DALFOPRISTIN
Quinupristin/dalfopristin is the combination of two semisynthetic pristinamycin derivatives (Figs. 2, 3) and is the first parenteral streptogramin antibacterial agent. Both quinupristin and dalfopristin have antibacterial capability individually, but demonstrate synergistic activity when used in combination. Much of the clinical experience with this antibiotic is derived from five comparative trials and an FDA-sanctioned emergency-use program for patients without alternative therapies.
[FIGURES 2-3 OMITTED]
Mode of Action
Cellular Mechanism
Quinupristin and dalfopristin enter bacterial cells by diffusion and bind to different sites on the 50S ribosomal subunit, resulting in an irreversible inhibition of bacterial protein synthesis (18). Dalfopristin blocks the reaction catalyzed by the peptidyl transferase catalytic center of the 50S ribosome via inhibition of substrate attachment to the P-site and the A-site of the ribosome; quinupristin inhibits peptide chain elongation. The synergistic effect of the combination appears to result from the fact that these compounds target early and late steps in protein synthesis (19).
Pharmacokinetics
Quinupristin/dalfopristin is rapidly cleared from the blood and is widely distributed. Its elimination is through the bile into the feces; however, its clearance may be slightly reduced in patients with severe chronic renal failure. Its pharmacokinetics are unaffected by age or gender. Quinupristin has a half-life of approximately 1 hour, and dalfopristin has a half-life of approximately 30 minutes. The post-antibiotic effect of the drug is prolonged to greater than 7.4 hours against streptococci, regardless of penicillin susceptibility (20). Quinupristin/dalfopristin inhibits the biotransformation rate of cytochrome P450 substrates in vitro.
In Vitro Activity
Quinupristin/dalfopristin has inhibitory activity against a broad spectrum of Gram-positive bacteria including MRSA, vancomycin-resistant E. faecuium (VREF), and drug-resistant Streptococcus pneumoniae. It is bactericidal against methicillin-resistant staphylococci and Streptococcus pneumoniae and bacteriostatic against most E. faecuium in vitro. Quinupristin/dalfopristin also has demonstrated synergy with other antibiotics. Rifampin is synergistic with quinupristin/dalfopristin against MRSA, and doxycycline is synergistic against VREF in vitro.
Clinical Indications
FDA indications for quinupristin/dalfopristin are serious infections associated with VREF bacteremia, and complicated skin and skin structure infections caused by methicillin-sensitive Staphylococcus aureus or Streptococcus pyogenes. VREF infections are difficult to treat, and few therapeutic options are currently available. These pathogens are resistant to most beta-lactam and aminoglycoside antibiotics. Judicious use of vancomycin is currently being advocated to reduce the incidence of resistant organisms, but their presence continues. In 1995, quinupristin was approved for emergency use. During this emergency-use basis in the treatment of VREF infections in which no other treatments were available, quinupristin/dalfopristin had a 71% success rate (21) and a significantly lower mortality rate than patients using other agents (22). In another study, patients with complicated skin and skin structure infections who were administered quinupristin/dalfopristin had almost identical clinical success rate (68%) when compared to those using vancomycin, oxacillin, and/or cefazolin (71%) (23). In addition, in the treatment of patients with Gram-positive nosocomial pneumonia, it was found to be equally as efficacious as vancomycin (24).
Dosage Regimens
For complicated skin or skin structure infections, the recommended dose is 7.5 mg/kg intravenously twice daily for at least seven days. The drug can be administered up to three times daily for bacteremic patients. Dose adjustment to 5 mg/kg is recommended for patients with hepatic insufficiency. No dose adjustment is needed for elderly or renally-impaired patients.
Contraindications/Cautions
Approximately 63% of patients receiving quinupristin/dalfopristin reported at least one adverse effect. Evaluation of these adverse effects is difficult, since it is often assessed in the context of severe underlying illnesses. Adverse venous events at the intravenous site of administration of the drug were the most common. Reports of pain and/or inflammation during its administration were reported in 34.9% to 74.0% of patients (23,25). Atrophy, edema, hemorrhage, hypersensitivity, burning, and thrombophlebitis were also reported. A statistically significant number of venous events occurred with quinupristin/dalfopristin compared with oxacillin, cefazolin, or vancomycin (66.2 vs. 28.4%) (21) Suggested but unproven management options to limit these events include administration in a larger volume of fluid or via a central line. Mild to moderate myalgias and/or arthralgias have been reported (26). Gastrointestinal events also occurred, with 4.6% experiencing nausea, 2.7% experiencing vomiting and diarrhea, and 2.5% developing a rash (27). The most common laboratory abnormalities reported were an increase in hepatic transaminases and bilirubin (27). It is contraindicated in patients with known hypersensitivity to streptogramins, or in cases of coadministration with any drugs metabolized by the CYP3A4 enzyme system (including some anti-HIV agents, vinca alkaloids, benzodiazepines, immunosuppressives, corticosteroids, and calcium channel blockers). In addition, particular care should be taken when using medications that prolong the QT interval (e.g. astemizole, cisapride, disopyramide, lidocaine, quinidine, and terfenadine) (27). Caution is also recommended if using cyclosporin concomitantly (28). Resistance to quinupristin/dalfopristin has been encountered infrequently among vancomycin-resistant E. faecium, and resistance among staphylococci is rare in the United States (29,30).
Conclusions
Quinupristin/dalfopristin is the first parenteral streptogramin and offers a unique alternative treatment against multi-drug-resistant Gram-positive bacteria. Because of its potency, bactericidal activity, long post-antibiotic effect, and rare resistance, it has excellent potential for treatment of serious Gram-positive infections. However, its efficacy should be weighed against possible adverse effects, tolerability, and interactions prior to utilizing this potent antibiotic. In seriously ill patients with unresponsive infections and minimal other potential treatment options, it should be considered the treatment of choice.
Summary
None of the aforementioned drugs are a solution to antibiotic resistance. However, many of the drugs have demonstrated significant efficacy against previously resistant Gram-positive organisms. Linezolid and quinupristin/dalfopristin appear to utilize novel mechanisms of action and may be particularly useful for selected clinical situations for treatment of community-acquired or nosocomial skin and soft tissue infections. Prudent use of these medications for presumed resistant organisms should be practiced because the development of resistance from overuse is a real concern.
References
(1.) Center for Disease Control. Reduced susceptibility of Staphylococcus aureus to vancomycin-Japan, 1996. MMWR Morb Mortal Wkly Rep 1997; 46:624-6.
(2.) Zyvox [package insert]. Kalamazoo, MI; Pharmacia & Upjohn Company, 2000.
(3.) Slee AM, Wuonola MA, McRipley RJ, Zajac I, Zawada MJ, Bartholomew PT, et al. Oxazolidinones, a new class of synthetic antibacterial agents: in vitro and in vivo activities of DuP 105 and DuP 721. Antimicrob Agents Chemother 1987; 31:1791-7.
(4.) Swaney SM, Aoki H, Ganoza MC, Shinabarger DL. The oxazolidinone linezolid inhibits initiation of protein synthesis in bacteria. Antimicrob Agents Chemother 1998; 42:3251-5.
(5.) Eliopoulos GM, Wennersten CB, Gold HS, Moellering RC Jr. In vitro activities of new oxazolidinone antimicrobial agents against enterococci. Antimicrob Agents Chemother 1996; 40:1745-7.
(6.) Ford CW, Hamel JO, Wilson DM, Moerman JK, Stapert D, Yancey RJ Jr., et al. In vivo activities of U-100592 and U100766, novel oxazolidinone antimicrobial agents, against experimental bacterial infections. Antimicrob Agents Chemother 1996; 40:1508-13.
(7.) Jorgensen JH, McElmeel ML, Trippy CW. In vitro activities of the oxazolidinone antibiotics U-100592 and U-100766, two novel fluorinated oxazolidinones. Antimicrob Agents Chemother 1997; 41:465-7.
(8.) Mercier RC, Penzak SR, Rybak MJ. In vitro activities of investigational quinolone, glycylcycline, glycopeptide, streptogramin, and oxazolidinone tested alone and in combinations against vancomycin-resistant Enterococcus faecium. Antimicrob Agents Chemother 1997; 41:2573-5.
(9.) Noskin GA, Siddiqui F, Stosor V, Hacek D, Peterson LR. In vitro activities of linezolid against important Gram-positive bacterial pathogens including vancomycin-resistant enterococci. Antimicrob Agents Chemother 1999; 43:2059-62.
(10.) Clemett D, Markham A. Linezolid. Drugs 2000; 59:815-27.
(11.) Perry CM, Jarvis B. Linezolid: a review of its use in the management of serious Gram-positive infections. Drugs 2001; 61:525-51.
(12.)Stevens DL, Smith LG, Bruss JB, McConnell-Martin MA, Duvall SE, Todd WM, et al. Randomized comparison of linezolid (PNU-100766) versus oxacillin-dicloxacillin for treatment of complicated skin and soft tissue infections. Antimicrob Agents Chemother 2000; 44:3408-13.
(13.)Stevens DL, Herr D, Lampiris H, Hunt JL, Batts DH, Hafkin B. Linezolid versus vancomycin for the treatment of methicillin-resistant Staphylococcus aureus infections. Clin Infect Dis 2002; 34:1481-90.
(14.) Batts DH. Linezolid--a new option for treating Gram-positive infections. Oncology (Huntingt) 2000; 14(Suppl 6):23-9.
(15.) Wilson SE, Solomkin JS, Le V, Cammarata SK, Bruss JB. A severity score for complicated skin and soft tissue infections derived from phase III studies of linezolid. Am J Surg 2003; 185:369-75.
(16.) Auckland C, Teare L, Cooke F, Kaufmann ME, Warner M, Jones G, et al. Linezolid-resistant enterococci: report of the first isolates in the United Kingdom. J Antimicrob Chemother 2002; 50:743-6.
(17.) Barrett JF. Linezolid Pharmacia Corp. Curr Opin Investig Drugs 2000; 1:181-7.
(18.) Bryson HM, Spencer CM. Quinupristin-dalfopristin: antibacterial activity, pharmokinetic profile, therapeutic trials, tolerability, and current status. Drugs 1996; 52:406-15.
(19.) Cocito C, Di Giambattista M, Nyssen E, Vannuffel P. Inhibition of protein synthesis by streptogramins and related antibiotics. J Antimicrob Chemother 1997; 39 Suppl A:7-13.
(20.) Lamb HM, Figgitt DP, Faulds D. Quinupristin/dalfopristin. A review of its use in the management of serious Gram-positive infections. Drugs 1999; 58:1061-97.
(21.) Landman D, Quale JM. Management of infections due to resistant enterococci: a review of therapeutic options. J Antimicrob Chemother 1997; 40:161-70.
(22.) Linden, PK, Pasculle AW, McDevitt D. Effect of quinupristin/dalfopristin on the outcome of vancomycin-resistant Enterococcus faecium bacteremia: comparison with a control cohort. J Antimicrob Chemother 1997; 39 Suppl A:145-51.
(23.) Nichols RL, Graham DR, Barriere SL, Rodgers A, Wilson SE, Zervos M, et al. Treatment of hospitalized patients with complicated Gram-positive skin and skin structure infections: two randomized, multicentre studies of quinupristin/dalfopristin versus cefazolin, oxacillin, or vancomycin. Synercid Skin and Skin Structure Infection Group. J Antimicrob Chemother 1999; 44:263-73.
(24.) Fagon J, Patrick H, Haas DW, Torres A, Gibert C, Cheadle WG, et al. Treatment of Gram-positive nosocomial pneumonia. Prospective randomized comparison of quinupristin/dalfopristin versus vancomycin. Nosocomial Pneumonia Group. Am J Respir Crit Care Med 2000; 161:753-62.
(25.) Moellering RC, Linden PK, Reinhardt J, Blumberg EA, Bompart F, Talbot GH. The efficacy and safety of quinupristin/dalfopristin for the treatment of infections caused by vancomycin-resistant Enterococcus faecium. Synercid Emergency-Use Study Group. J Antimicrob Chemother 1999; 44:251-61.
(26.) Olsen KM, Rebuck JA, Rupp ME. Arthralgias and myalgias related to quinupristin-dalfopristin administration. Clin Infect Dis 2001; 32:e83-6.
(27.) Rubinstein E, Prokocimer P, Talbot GH. Safety and tolerability of quinupristin/dalfopristin: administration guidelines. J Antimicrob Chemother 1999; 44 Suppl A:37-46.
(28.) Stamatakis MK, Richards JG. Interaction between quinupristin/dalfopristin and cyclosporine. Ann Pharmacother 1997; 31:576-8.
(29.) Eliopoulos GM. Quinupristin-dalfopristin and linezolid: evidence and opinion. Clin Infect Dis 2003; 36:473-81.
(30.) Abbanat D, Macielag M, Bush K. Novel antibacterial agents for the treatment of serious Gram-positive infections. Expert Opin Investig Drugs 2003; 12:379-99.
(31.) Sidoroff A, Halevy S, Bavinck JN, Vaillant L, Roujeau JC. Acute exanthematous pustulosis (AGEP): a clinical reaction pattern. J Cutan Pathol 2001 Mar; 28(3):113-9.
ADDRESS FOR CORRESPONDENCE:
Jeffrey M Weinberg MD
Department of Dermatology
St. Luke's-Roosevelt Hospital Center
1090 Amsterdam Avenue, Suite 11D
New York, NY 10025
Phone: 212-523-5898
Fax: 212-523-5027
Email: jwein@bway.net
COPYRIGHT 2003 Journal of Drugs in Dermatology
COPYRIGHT 2003 Gale Group