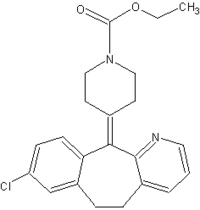
Hypospadias is a birth defect that affects approximately seven in 1,000 male infants in the United States. In affected infants, the urethral opening is located along the underside of the penis, scrotum, or perineum; the condition usually is corrected by surgery. Hypospadias is classified in order of increasing severity as first, second, or third degree. In 2002, a study in Sweden noted that among male infants born to women who while pregnant had taken loratadine (Claritin[R]), a nonsedating antihistamine commonly used for seasonal allergies, hypospadias prevalence was twice that of the general population (1). However, insufficient data were available to determine the severity of the hypospadias cases, and the study did not control for confounding variables (e.g., family history of hypospadias or maternal age). In 2003, a prospective study using data from four countries indicated that five of 142 pregnancies in women exposed to loratadine resulted in infants with major malformations, a prevalence consistent with that of the general population; none had hypospadias (2). To further assess any potential association between loratadine and hypospadias, CDC analyzed data from the National Birth Defects Prevention Study (NBDPS). This report summarizes the results of that analysis, which determined that no increased risk for second- or third-degree hypospadias existed among women who used loratadine in early pregnancy (Table). These results might be useful for women and health-care providers to address concerns about loratadine use and hypospadias.
NBDPS is an ongoing, multistate, case-control study of environmental and genetic risk Factors for major birth defects that can be used in response to public health concerns regarding rare drug exposures and birth defects (3,4). Infants are identified through birth defect surveillance systems in eight states; mothers undergo a detailed interview by telephone in English or Spanish. For this analysis, the case population was defined as male infants with second- or third-degree hypospadias. Infants with first-degree hypospadias are not included in NBDPS because the mildest form of hypospadias is much less completely ascertained by routine surveillance. Infants were excluded if they had 1) known or suspected chromosome abnormalities, 2) single gene conditions, or 3) other recognized multiple congenital anomaly phenotypes. The control population consisted of live-born male infants with no major birth defects, selected at random from the same populations as the case group. Excluded from the analysis were 86 infants whose mothers had incomplete interviews and 30 infants (28 in the case population and two in the control population) who had fathers or brothers with hypospadias. The study populations consisted of 563 male infants with hypospadias and 1,444 male infant controls; all were born during October 1, 1997-June 30, 2001.
Exposure was defined as any maternal use of loratadine from 1 month before pregnancy through the first trimester. To control for confounding by indication, exposure to other nonsedating or sedating antihistamines during the same period also was assessed. Potential confounding factors tested by multivariate logistic regression analysis included maternal age, maternal race/ethnicity (i.e., non-Hispanic white, non-Hispanic black, Hispanic, and other), birth month, and state of residence at delivery.
Of 563 male infants with hypospadias, 46 (8.2%) had multiple major birth defects that were not recognized phenotypes, and 517 (91.8%) had hypospadias with no other major birth defects. Among the 1,957 mothers of infants in the case and control populations, 33 (1.7%) reported using loratadine during the exposure period. Univariate analyses showed no association between this use of loratadine and hypospadias (Table). Use of nonsedating antihistamines (including loratadine) and sedating antihistamines also were not associated with hypospadias. Multivariate adjusted odds ratio estimates did not vary significantly from the univariate estimates. In addition, no association between loratadine use and hypospadias was determined when cases with multiple major defects were excluded or when different exposure periods were examined.
Reported by: M Werler, ScD, Slone Epidemiology Center, Boston Univ School of Public Health, Massachusetts. C McCloskey, MD, Center for Drug Evaluation and Research, Food and Drug Administration. LD Edmonds, MSPH, R Olney. MD, MA Honein, PhD, Div of Birth Defects and Developmental Disabilities, National Center on Birth Defects and Developmental Disabilities; J Reefhuis, PhD, EIS Officer, CDC.
Editorial Note: The findings in this report indicated that hypospadias was not associated with use of loratadine during the period from 1 month before pregnancy through the first 3 months of pregnancy. During 1998-1999, loratadine was the drug most advertised directly to consumers (5) and was used by 3% of women of childbearing age (6). In November 2002, loratadine was approved by the Food and Drag Administration for over-the-counter use (7). Antihistamines are used widely by the general population, including women of child-bearing age, 20%-30% of whom have allergic conditions, primarily rhinitis and sinusitis (8). Because an estimated 50% of all pregnancies in the United States are unintended (9), women frequently are exposed inadvertently to medications before learning they are pregnant.
This report is subject to at least two limitations. First, NBDPS does not track all birth defects. Because first-degree hypospadias is excluded, the potential association between this mildest form of hypospadias and loratadine could not be assessed. Second, women are interviewed about their pregnancy exposures after delivery, and recall of drug use might be different among mothers of infants with major birth defects compared with mothers of infants without major birth defects.
The results of this analysis might be useful for women and health-care providers to address concerns about loratadine use and hypospadias. These results do not provide definitive information on the overall safety of loratadine. Women should continue to consult their health-care providers before using any medications during pregnancy. Future studies of medications and birth defects, possibly using NBDPS, are needed to address some of the current knowledge gaps on the effects of medication use during pregnancy.
Acknowledgments
This report is based in part on contributions by CA Hobbs, MD, Univ of Arkansas for Medical Sciences, Little Rock, Arkansas. GM Shaw, DrPH, S Carmichael, PhD, California Birth Defects Monitoring Program, Emeryville, California. PA Romitti, PhD, Univ of Iowa, Iowa City, Iowa. K Kelley, Slone Epidemiology Center, Boston Univ School of Public Health; M Anderka, MPH, Massachusetts Dept of Public Health. M Royle, PhD, New Jersey Dept of Health and Senior Svcs. C Druschel, PhD, New York State Health Dept. M Canfield, PhD, P Langlois, PhD, Texas Dept of Health.
References
(1.) Kallen B. Use of antihistamine drugs in early pregnancy and delivery outcome. J Matern Fetal Neonatal Med 2002;11:146-52.
(2.) Moretti M.E., Caprara D., Coutinho C.J., et al. Fetal safety of loratadine use in the first trimester of pregnancy: a multicenter study. J Allergy Clin Immunol 2003;111:479-83.
(3.) Yoon P.W., Rasmussen S.A., Lynberg M.C., et al. The national birth defects prevention study. Public Health Rep 2001;116(suppl 1):32-40.
(4.) Rasmussen S.A., Olney R.S., Holmes L.B., Lin A.E., Keppler-Noreuil K.M., Moore C.A. Guidelines for case classification for the National Birth Defects Prevention Study. Birth Defects Res Part A Clin Mol Teratol 2003;67:193-201.
(5.) Findlay S.D. Direct-to-consumer promotion of prescription drugs: economic implications for patients, payers and providers. Pharmacoeconomics 2001;19:109-19.
(6.) Kaufman D.W., Kelly J.P., Rosenberg L., Anderson T.E., Mitchell A.A. Recent patterns of medication use in the ambulatory adult population of the United States: the Slone survey. JAMA 2002;287:337-44.
(7.) Food and Drug Administration. FDA approves OTC Claritin. FDA Consum 2003;37:3.
(8.) Schatz M., Zeiger R.S. Diagnosis and management of rhinitis during pregnancy. Allergy Proc 1988;9:545-54.
(9.) Henshaw S.K. Unintended pregnancy in the United States. Fam Plann Perspect 1998;30:24-9, 46.
COPYRIGHT 2004 U.S. Government Printing Office
COPYRIGHT 2004 Gale Group