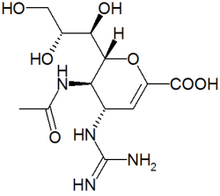
Zanamivir
Zanamivir is a neuraminidase inhibitor used in the treatment of and prophylaxis of both influenza A and influenza B. Zanamivir was the first neuraminidase inhibitor commercially developed. It is currently marketed by GlaxoSmithKline under the trade name Relenza®. more...
Development
Zanamivir was discovered in 1989 by scientists at the Australian biotechnology company Biota Holdings, working in conjunction with the CSIRO and the Victorian College of Pharmacy. The development was part of Biota's ongoing program to develop antiviral agents through rational drug design. An outline of the successful rational design strategy used was published in a paper in Nature (von Itzstein et al., 1993).
The strategy relied on the availability of the crystal structure of influenza neuraminidase which was achieved by x-ray crystallography. It was known as far back as 1974 that 2-deoxy-2,3-didehydro-N-acetylneuraminic acid (DANA), a sialic acid analogue, was an inhibitor of neuraminidase (Meindl et al., 1974). Using the crystal structure of neuraminidase and DANA as a starting point, the researchers employed a computer-aided process to attempt to design a molecule which better fitted (and therefore inhibited) the active site of neuraminidase. Zanamivir, a transition-state analogue inhibitor of neuraminidase, was the result.
In 1990, zanamivir was licensed to Glaxo (now GlaxoSmithKline) for exclusive worldwide development and marketing. In 1999, the product was approved for marketing in the US and subsequently has been registered by GSK in a total of 70 countries.
Limitations
Whilst zanamivir proved to be a potent and effective inhibitor of influenza neuraminidase and inhibitor of influenza virus replication in vitro and in vivo, this didn't necessarily translate into a successful clinical treatment for influenza. In clinical trials it was found that zanamivir was able to reduce the time to symptom resolution by 1.5 days provided therapy was started within 48 hours of the onset of symptoms.
A further limitation concerns the poor oral bioavailability of zanamivir. This meant that oral dosing was impossible limiting dosing to the parenteral routes. Zanamivir, therefore, is administered by inhalation - a route that was chosen for patient compliance with therapy. But even this route of administration is not acceptable to many in the community.
A troubled commercial venture
Biota, being only a small company, was not able to bring the drug to market by itself. Consequently, it was licensed to Glaxo (now GlaxoSmithKline) to complete development and to market internationally as Relenza, delivered via Glaxo's proprietary, and some would say cumbersome, Diskhaler inhalation device. The license agreement entitled Biota to receive a 7% royalty on Glaxo's sales of Relenza.
A combination of factors has resulted in the limited commercial success of zanamivir (Relenza). The relatively small effect on the timecourse of influenza symptoms, the inhalation dosage form, a less-than-ideal device, and high expense make it a difficult product to market well. And although zanamivir was the first neuraminidase inhibitor to the market, it had only a few months lead over the second entrant, oseltamivir (Tamiflu), with an oral formulation much preferred by patients. Faced with this competition, GSK effectively abandoned the product.
Read more at Wikipedia.org