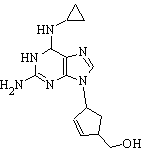
Ziagen
Abacavir (ABC) is the most powerful nucleoside analog reverse transcriptase inhibitor (NARTI) used to treat HIV and AIDS. It has been well tolerated, its main side effect being hypersensitivity reactions, which can be dangerous. It is available under the trade name Ziagen™ by GlaxoSmithKline and the combination drug Trizivir™. more...
Abacavir is one of the most deadly NARTIs, due its ability to trigger to hypersensitivity reactions that lead to sudden death in certain individuals. However, a genetic test is now available that, while not perfect, has reasonable accuracy in predicting whether an individual will be hypersensitive. Over 90% of patients can safely take abacavir.
Strains that are resistant to zidovudine (AZT) or lamivudine (3TC) are generally sensitive to abacavir, whereas strains that are resistant to AZT and 3TC are not as sensitive to abacavir.
History
Abacavir was approved by the Food and Drug Administration (FDA) on December 18, 1998 and is thus the fifteenth approved antiretroviral drug in the United States. Its patent will expire in the United States on 2009-12-26.
Mechanism of action
ABC is an analog of guanosine. It is capable of crossing the blood-brain barrier.
Abacavir is given orally and has a high bio-availability of 83%. It is metabolised primarily through alcohol dehydrogenase or gluconyl transferase.
Read more at Wikipedia.org