Zocor
Simvastatin is a hypolipidemic drug belonging to the class of pharmaceuticals called "statins". It is used to control hypercholesterolemia (elevated cholesterol levels) and to prevent cardiovascular disease. more...
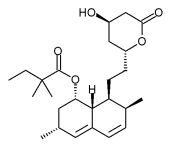
Simvastatin is a synthetic derivate of a fermentation product of Aspergillus terreus.
Uses
Simvastatin is a powerful lipid-lowering drug that can decrease low density lipoprotein (LDL) levels by up to 50%. It is used in doses of 10 mg up to 80 mg. Higher doses (160 mg) have been found to be too toxic, while giving only minimal benefit in terms of lipid lowering. There is no real effect on HDL and triglyceride levels.
From recent research it has become apparent that simvastatin and other statins inhibit the progression of atherosclerosis beyond their effects on LDL. A large number of explanations has been proposed, for example its inhibitory effect on macrophages in the atherosclerotic plaque lesions.
Rationing
Since its introduction, there has been a large debate surrounding the price for lipid-lowering treatment and its benefits on atherosclerosis. Although this has affected the other statins as well, simvastatin was the first statin drug to be used extensively in clinical practice.
A number of large epidemiological studies were conducted to discover which patients would benefit most from statin drugs; most studies involve simvastatin as the study drug. The most influential studies were the Scandinavian Simvastatin Survival Study (4S) and the Heart protection study (HPS).
It has now become apparent that patients with one or more risk factors for cardiovascular disease (such as diabetes mellitus, hypertension or a positive family history) can benefit from statins—even if they do not have substantially elevated cholesterol levels.
Simvastatin was introduced in the late 1980s, and in many countries it is now available as a generic preparation. This has led to a decrease of the price of most statin drugs, and a reappraisal of the health economics of preventive statin treatment.
In the UK, simvastatin (in a dose of 10mg) has recently become available to purchase from pharmacies without prescription.
Pharmacology
All statins act by inhibiting HMG-CoA reductase, the rate-limiting enzyme of the HMG-CoA reductase pathway, the metabolic pathway responsible for the endogenous production of cholesterol.
The drug is the form of an inactive lactone that is hydrolized after ingestion to produce the active agent. It is a white, nonhygroscopic, crystalline powder that is practically insoluble in water, and freely soluble in chloroform, methanol and ethanol.
Marketing
Reference: Drug Discovery Today editorial, 2005
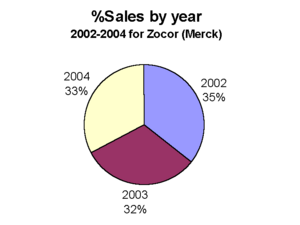
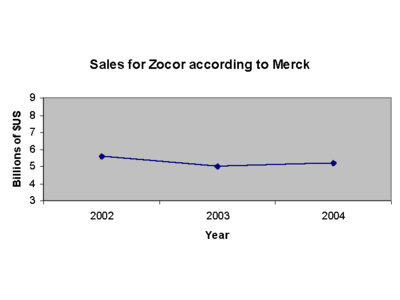
Brand names: Zocor®, Zocor Heart Pro®, marketed by the pharmaceutical company Merck & Co. and Denan (Germany), Liponorm, Sinvacor, Sivastin (Italy), Lipovas (Japan), Lodales (France), Zocord (Austria and Sweden) and other.
Read more at Wikipedia.org