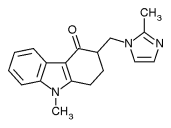
Ondansetron is increasingly prescribed off label for nausea and vomiting in pregnancy, and a new study suggests it is safe for this indication.
In the prospective observational study, the drug, which is typically used for treating nausea and vomiting in cancer chemotherapy patients, was not associated with an increased risk of fetal malformations, reported Adrienne Einarson, R.N., of the University of Toronto and her colleagues (BJOG 2004;111:940-3).
The investigators studied women exposed to ondansetron (Zofran), other antiemetics (including Diclectin, metoclopramide, phenothiazines, and ginger), and/or no drugs or only nonteratogenic drugs. Outcomes from 176 pregnancies in each group have been evaluated.
In the ondansetron group there were 169 live births, five miscarriages, and two therapeutic abortions. There were six major malformations, for a rate of 3.5%. The mean birthweight was 3,362 g. There were no statistical differences between the three groups in any of the study end points.
Ondansetron appears safe for the fetus, but many more cases would need to be studied before a definitive conclusion about the safety of the drug could be made, the investigators said.
COPYRIGHT 2004 International Medical News Group
COPYRIGHT 2004 Gale Group