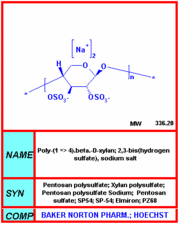
Elmiron
Pentosan polysulfate (sold under the name Elmiron) was the first --and only-- oral medication approved by the US FDA for the treatment of interstitial cystitis, also known as painful bladder syndrome. Interstitial cystitis patients struggle with symptoms of urinary frequency, urgency, pressure and/or pain, as well as nocturia (frequent urination at night), dyspareunia (painful intercourse), pain and/or discomfort while sitting in a car, while driving and/or travelling. more...
The origin/cause of IC is unknown though a number of theories are currently under consideration. Urine cultures are typically negative for infection, yet it is not unusual for patients to believe that they have had infections for years rather than IC. Why? Because the symptoms of an infection are nearly identical to the symptoms of IC.
Diagnosis has been greatly simplied in recent years with the development of two new methodologies. The Pelvic Pain Urgency/Frequency (PUF) Patient Survey, created by C. Lowell Parsons, is a short questionnaire that will help doctors identify if pelvic pain could be coming from the bladder. The KCL Test, aka the Potassium Sensitivity Test, uses a mild potassium solution to test the integrity of the bladder wall. Though the latter is not specific for IC, it has been determined to be helpful in the evaluation of the use of compounds, such as pentosan, which are designed to help repair the GAG layer. The previous "gold standard" test for IC was the use of hydrodistention with cystoscopy. Researchers, however, determined that this visual examination of the bladder wall was also not specific for IC and that the test, itself, can contribute to the development of small glomerulations (aka petechial hemorrhages) often found in IC. Thus, a diagnosis of IC is one of exclusion, as well as a review of clinical symptoms.
Pentosan is believed to work by providing a protective coating to the damaged bladder wall. The critical flaw of the medication, however, is its exceptionally poor bioavailability when taken orally. Research presented late in 2005 by Alza Pharmaceuticals (Metabolism of pentosan polysulfate sodium (PPS) in healthy human volunteers. Xenobiotica. 2005 Aug;35(8):775-84.) demonstrates that more than 84% of the medication is excreted, intact, in feces without providing any beneficial effect. Their research found that only 6% was excreted through urine. Thus, it must be taken for several months for most patients to achieve some benefit. More recently, however, Elmiron has been studied as part of a "rescue instillation" which is placed directly in the bladder and can, perhaps, provide better effectiveness. C. Lowell Parsons has presented a research study which shows a 90% effectiveness in reducing the symptoms of IC patients by using this instillation.
Patients who have taken pentosan orally report a variety of side effects, primarily GI complaints such as diarrhea, heartburn, stomach pain. Hair loss, headache, rash, insomnia have also been reported. One concern is the potential for blood thinning with this medication. Some patients have reported that they bruise more easily. In some cases, patients are asked to stop medication before any major surgical procedures to reduce the likelihood of bleeding.
Read more at Wikipedia.org