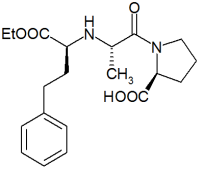
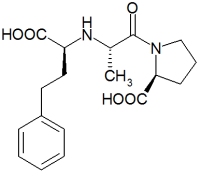
International pharmaceutical and health care company, F H Faulding & Co Limited (Faulding), Adelaide, South Australia, has announced that its Faulding Pharmaceuticals division has received final approval from the Food and Drug Administration (FDA) in the United States for its abbreviated new drug application (ANDA) for enalaprilat injection, following the expiry of the paediatric exclusivity period on 22 August 2000. Enalaprilat injection is a generic version of Vasotec(R), which is indicated for treatment of hypertension and is marketed by Merck & Co. Total brand sales in 1999 for Vasotec(R) I.V. were approximately US$41 million.
Faulding Pharmaceuticals will also launch enalapril maleate tablets 2.5mg, 5mg, 10mg and 20mg under the Purepac brand in the United States, having obtained distribution and marketing rights from Ranbaxy, an Indian pharmaceutical company. Ranbaxy received final FDA approval for their ANDAs for enalapril tablets on 22 August 2000. Enalapril maleate tablets are a generic version of Vasotec(R) in its oral form, which is also indicated for treatment of hypertension and marketed by Merck & Co. Total brand sales in 1999 for the oral version of Vasotec(R) were approximately US$690 million.
Commercial distribution of both enalaprilat injection and enalapril maleate tablets will commence immediately.
Frank Condella, president and CEO of Faulding Pharmaceuticals, said "Faulding Pharmaceuticals has a strong pipeline of products to ensure good growth. We are also pursuing innovative means of augmenting that pipeline through marketing arrangements with other pharmaceutical companies who can benefit from our distribution network and established customer relationships."
"The launch of enalaprilat injection is a good example of the strength of our internal development and regulatory ability with Faulding receiving tentative approval from the FDA in less than 12 months when the average approval time is 20 months. The launch of enalapril maleate tablets demonstrates our ability to expand our range of products in other ways. We believe that Faulding is unique in its offering of both the I.V. and oral forms of this product."
Faulding Pharmaceuticals currently has 14 products awaiting approval with the FDA.
Faulding is a worldwide health and personal care company, listed on the Australian Stock Exchange. Faulding's principal businesses are generic oral and injectable pharmaceuticals, consumer health products, the provision of distribution and retail management services to pharmacies and logistics management services to hospitals. Faulding's products are sold in over 70 countries.
For more information, call +61 8 8205 6500.
COPYRIGHT 2000 Worldwide Videotex
COPYRIGHT 2000 Gale Group