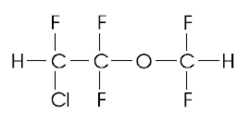
THE BIOLOGICAL CHANGES and the adverse effects produced by the main inhalation anesthetics have been studied mostly in experimental animals; human studies have mainly dealt with patients receiving presurgical anesthesia. Studies are lacking on subjects chronically exposed to these substances for occupational reasons.[1] Enflurane (ethrane; CAS 13838-16-9), alone or in combination with nitrous oxide ([N.sub.2]O), is used widely as general inhalation anesthetic. Occupationally exposed groups include anesthesiologists, other physicians (e.g., surgeons), and operating room nurses.
Enflurane has structural analogies with bis-chloromethyl ether, which is very carcinogen.[2] Available data, however, exclude potential mutagenicity or carcinogenicity of enflurane,[3-5] and no teratogenic effects have been demonstrated in rats or mice.[6,7] There is, however, no doubt that one of the chronic effects of enflurane is marked depression of leukopoiesis (granulocytopenia and lymphocytopenia) and UDP-glucuronyltransferase activity observed in rats exposed to nonanesthetic concentrations (subchronic exposure).[8] In addition, gross alteration in certain neuropsychic performance indices has been shown in volunteers exposed to subanesthetic concentrations.[1,9,10] Therefore, we believe it is important to (a) develop methodologies to correlate biological indicators of occupational exposure within individual environmental measurements and (b) to identify exposure levels that can be used in longitudinal epidemiologic studies on exposure-effect correlation.[11] In this study, we describe a method for the biological monitoring of occupational exposure to enflurane.
The National Institute for Occupational Safety and Health (NIOSH)[12] recommends a time-weighted threshold limit value (TLV) of 1 ppm for enflurane, based on the assumption that it is possible to maintain such low concentrations in operating rooms. A TLV of 75 ppm, as a time-weighted average (TWA), is recommended by the American Conference of Governmental Industrial Hygienists (ACGIH), based on the assumption that enflurane is a safer anesthetic agent than halothane or trichloroethylene, and no adverse effects are known at subanesthetic concentrations.[13,14] The ACGIH also suggests that the adjustment for presence of other anesthetic waste gases in the operating room is of importance. The exposure concentrations of [N.sub.2]O and enflurane in operating rooms are usually 50:1; therefore, the enflurane concentration should be below 10 ppm to maintain the TLVs of both gases.[14]
Biological monitoring of occupational exposure to enflurane can be achieved if concentrations of the biotransformation products of the anesthetic in blood and urine or concentrations of enflurane in alveolar air or in venous blood are measured. Of all the biotransformation products, inorganic fluorides measured in blood are used as a biological indicator,[15-17] although recent studies have shown limited usefulness[18]; the blood concentrations of occupationally exposed subjects did not differ significantly from the control groups.[1,15]
Small quantities of enflurane are eliminated unchanged in urine. In the assessment of individual exposure to enflurane, we hypothesize that the method of choice should be the measurement of enflurane urine concentrations. Enflurane in bladder urine, in fact, can be regarded as a time-weighted sample of enflurane in inhaled air during the time urine is formed, similar to what has been observed for [N.sub.2]O,[19,20] halothane,[11,21] isoflurane,[22] and other inert substances.[23]
We investigated the relationship between enflurane concentrations in operating rooms (TWA, inspiratory concentration [IC], ppm) and the corresponding urinary concentration (UC, [mu]g/I) in operating theater personnel. Samples of urine produced throughout exposure were collected anaerobically at the end of exposure. We also calculated some biological exposure limits that corresponded to the threshold values proposed in the literature.
Materials and methods
Subjects. The study involved 159 subjects (1 19 men and 40 women) aged 40.8 [+ or -] 7.3 y. Anesthesiologists, surgeons, and nurses were studied during routine operating activities in 18 different operating theaters of eight hospitals in Italy. Five members of a university hospital staff who were not exposed to anesthetic served as control subjects. Monitoring was done during anesthesia for general and orthopedic surgery. All types of anesthetic work were included (mask anesthesia, intubation anesthesia, etc.).
Waste gas scavenging was used in six operating theaters. The occupational exposure included, in addition to enflurane, [N.sub.2]O (48.3 [median value] [+ or -] 1.8 ppm [geometric standard deviation]). The environmental concentration of enflurane in the breathing zone was measured by personal passive samplers.
Urine collection and enflurane urinary concentrations. Urine samples (10 ml) were collected from all subjects at the beginning and at the end of each exposure period in 120-ml glass Supelco vials with airtight plugs (without silicone). The five control subjects provided urine samples collected on a Monday morning.
The vials were equipped with a Minimert valve to enable a headspace gas sample of enflurane over the urine to be withdrawn after partial pressure equilibrium. The subjects voided the bladder rapidly in areas known to be free of enflurane. Prior to measurement, the vials were stored at 37 [degrees]C for 2 h and shaken periodically to speed the separation and partial pressure equilibrium of enflurane vapor between the urine and the headspace air over it. A headspace air sample was analyzed by gas chromatography (HP 5880 A) plus mass selective detection (HP 5970 A). The analytical conditions for the gas chromatograph were as follows: cross-linked column, 5% phenyl methyl silicone (internal diameter = 0.2 mm; length = 25 m); column temperature, 25 [degrees]C; carrier gas, helium; retention time of enflurane, 1.80 min; sampler volume, 1 [mu]l. The analytical conditions for the mass selective detector were: monitored ion, 51; dwell time, 100 ms; selected ion monitoring window time, 0.1 amount mass unit; electromultiplier voltage, 2 000V.
Urinary concentration (UC) of enflurane was determined by the following formula:
UC = [X.sub.2]/UV X ([lambda] X UV/VA + 1), where UV and VA are the urine volume and the air headspace volume, respectively; [X.sub.2] is the amount of enflurane in the headspace after equilibration; and [lambda] is the enflurane Ostwald partition coefficient (urine/air) at 37 [degrees]C, which corresponds to 1.93.[24]
Recoveries of 98 to 102% were obtained by adding enflurane to urine. The limit of detection for our technique was 0.1 [mu]g/I. The coefficient of variation (CV) for enflurane on 20 determinations was 4.9% (mean value = 20 [mu]g/I).
Time-weighted average environmental concentration measurement in the breathing zone. The measurement of the TWA of enflurane concentration in the breathing zone was made by means of a diffusive personal sampler (Orsa 5; Draeger); each sampler was exposed for 2 h (two samplers were used throughout the monitored period of 4 h[25]). The precision and accuracy of passive monitors were within acceptable limits over the tested range. Thirty passive monitors were tested at five concentration levels (0.5, 1, 2, 5, and 10 ppm). An overall recovery of 96.5% and a relative standard deviation (RSD) of 4.9% were observed. The limit of detection was 0.01 ppm.
Results
The median value for exposure to enflurane for 4 h was 1.31 ppm (GSD = 2.54; range = 0.3-8 ppm) (Fig. 1). Only 40% of the environmental concentrations measurements were lower than the 1-ppm limit value proposed by NIOSH (Fig. 2).[12]. The median value of enflurane in urine samples was 4.52 [mu]g/I (GSD = 2.52; range = 0.1-24.9 [mu]g/I) (Fig. 3). In a control group that included 5 subjects who were staff members at a university hospital and who had not been exposed to anesthetic gases, the urinary enflurane measurements were performed in urine collected on Monday (8:00 a.m.); no peaks corresponding with the retention time of enflurane were identified.
The correlation between UC ([mu]g/I) and mean weighted IC (mg/[m.sup.3]) was significant (r = 0.77, p = .0001) as determined by the following regression equation:
UC = 2.196 X IC + 2.048. The regression line did not start from the origin of the Cartesian coordinates but intercepted the y axis at 2.048 [mu]g/I (Fig. 4). This finding indicates that at the beginning of the exposure period there was some enflurane in the urine of the exposed subjects, i. e., some of the anesthetic was retained in the body over night.
Discussion
Numerous epidemiologic studies suggest that chronic exposure to volatile anesthetics in the operating room represents a health risk for occupationally exposed subjects.[11] Thus, we found it useful to develop a method for the biological monitoring of occupational exposure to enflurane.
Biological compartments proposed for biological monitoring of anesthetic agents include (a) alveolar air (mean concentration in expired air),(26) (b) venous blood,(27) and (c) urine.(11,19,20,22) Anesthetics are found in various body compartments; the concentrations depend on the level of exposure (variable parameter) and on their solubility coefficients (constant parameter).(1) They are eliminated through the kidney unchanged or as catabolic transformation products. At the end of occupational exposure, when the environmental concentrations are zero, a large percentage of the anesthetic is also eliminated via the respiratory route. in urine, enflurane is eliminated both unchanged and as a metabolite.
Exposure to enflurane at anesthetic doses produces an increase in blood concentrations of inorganic fluorides,(1) and there is evidence of wide individual variability in metabolism.(18) In occupationally exposed subjects, inorganic fluorides cannot be considered as reliable biological indicators for the following reasons: (a) the metabolized portion is fairly small and decidedly smaller than for other halogenated anesthetics (2.5%), making it subject to small variations[16,28]; (b) blood concentrations of fluorides may be influenced by dietary and personal hygiene habits (e.g., use of particular toothpastes)[29]; (c) drinking water contains fairly high concentrations of fluorides; and (d) not all the newly formed fluorides are eliminated through the kidney because they are partly incorporated in bone matrix.[30] The results of a recent study confirm the doubts about the usefulness of inorganic fluorides as indicators of occupational exposure to enflurane; the blood concentrations of anesthetists and operating theater nurses did not differ significantly from the control groups.[15]
In a study by Carlsson et al.,[15] the concentrations of fluorides in plasma in subjects occupationally exposed to environmental levels less than 2 ppm never exceeded 2 [mu]mol/l, and the difference between the initial and final mean concentrations in a group exposed to enflurane only for 2 wk (no accumulation) was not significant (difference = 0.1 3 [+ or -] 0.78 [mu]mol/l).
Significant increases in plasma fluorides were observed in persons exposed for 4 h to 200 ppm of environmental enflurane (from 0.78 to 2.14 [mu]mol/l). The maximum value was reached after 1 h of exposure and remained constant for the duration of exposure. Blood values of fluorides fell rapidly after cessation of exposure; after 12 h, the levels did not differ significantly from those recorded prior to exposure.[15]
Enflurane disappears rapidly from body tissues and disappears completely from blood during the period after the workshift,[31] thus precluding the use of blood fluorides 16 h after exposure as an indicator of the mean level of exposure during previous days.
Cattaneo et al.[31] found that blood levels of enflurane increase significantly during the course of a surgical operation. At environmental exposure levels between 3 and 8 ppm, blood concentrations measured daily for 10 consecutive d varied between 1.2 and 280 [mu]g/I. There were no significant differences that could be attributed to the different duties performed in the operating theater (anesthetist, surgeon, nurse).
On the basis of our experience of biological monitoring of occupational exposure to enflurane, we propose the measurement of the unchanged anesthetic in urine samples collected at the end of exposure as a biological index. The urinary concentrations should be proportional to those in arterial blood, with a proportional coefficient corresponding to the [lambda] blood/air value. Repeated arterial blood samples could serve as a means for calculation of time-averaged uptake in the body, but this method is impractical. The urine leaving the kidney can be regarded as a sample from arterial blood (the bladder serves as a collecting and mixing vessel for urine that contains a time-weighted average of enflurane uptake). The hypothesis that the enflurane content in bladder urine reflects the time-weighted exposure has been confirmed for other inert substances.[23]
A close relationship was found between the time-weighted average environmental concentration in the breathing zone and the urinary concentration in samples collected after 4-h exposure for the groups of occupationally exposed subjects (r = 0.77).
The enflurane environmental threshold values proposed in the literature are (a) 75 ppm by ACGIH, 1 (b) 10 ppm as a technical limit by ACGIH,[14] and (c) 1 ppm by NIOSH.[12] Our data show that the corresponding urinary concentrations (based on 4-h exposure) are, respectively, (a) 166 [mu]g/I (extrapolated value), (b) 24 [mu]g/I, and (c) 4 [mu]g/I.
If a biological exposure limit is to be proposed, it is necessary to consider the corresponding 95% lower confidence limit of the regression line (UC = 2.02 X IC + 1.52). The biological thresholds are 153 [mu]g/I for an environmental exposure of 75 ppm, 22 [mu]g/I for an environmental exposure of 10 ppm, and 3.5 [mu]g/I for an environmental exposure of 1 ppm.
The following descriptions of the different measurement options show the advantage of using urinary concentrations as a biological indicator of enflurane exposure.
The respiratory compartment communicates directly with the environment and, therefore, follows its variations very rapidly. For this reason, single alveolar samples performed during exposure reflect the instantaneous environmental values immediately preceding sampling. Thus, in order to obtain an alveolar value that will represent a mean exposure value, numerous alveolar samples must be made during exposure (multiple instantaneous samples).
The venous blood compartment is used widely as a medium in biological monitoring. It is similar in some ways to the alveolar compartment in that sampling must be instantaneous. Venous blood, however, is not in direct communication with the environment; therefore, it can be assumed that blood concentration values reflect a greater degree of weighting with respect to exposure levels than do alveolar values.
The urinary compartment is the site of excretion of anesthetics absorbed via the inhalation route. The urinary concentration values of metabolites and anesthetics eliminated unchanged are always integrated over time because the bladder acts as a collection and mixing vessel for the urine formed in the kidney (outcome of rapid exchanges between blood and fluids fiftered and absorbed in the nephron).
Given that the anesthetics are poorly soluble, equilibration between environmental air, alveolar air, and arterial and renal venous blood occurs within a few minutes. This means that the fluctuations in environmental concentrations are reflected rapidly at the urinary level because the urine newly formed in the kidney can be considered as a liquid sample in equilibrium with renal blood.[19-21]
Some volatile chemicals, such as toluene, styrene, methanol, and anesthetics, appear to be eliminated in the kidney by a diffusion process determined by the equilibration of partial pressure in urine and plasma. As a result, the urine/blood ratio equals the urine/blood distribution coefficient, and the concentration of the determinant in urine is independent from urine output. The adjustment for creatinine may not be justified if the excretion mechanism of the determinant is different from the excretion mechanism of creatinine.[32]
The proposed method can be performed easily and quickly. Obviously, urine collection must be performed with the necessary precautions; any loss of enflurane must be avoided and samplers used must be gas-tight.
The proposed biological values are 153 [mu]g/I for an environmental exposure of 75 ppm, 22 [mu]g/I for an environmental exposure of 10 ppm, and 3.5 [mu]g/I for an environmental exposure of 1 ppm. The proposed values can be regarded as TWA samplers, reflecting exposure for a 4-h period.
Submitted for publication December 23, 1992; revised; accepted for publication March 30, 1993. Requests for reprints should be sent to: Dr. Marcello Imbriani, Clinica del Lavoro, Via S. Boezio, 24, 271 00 Pavia, Italy.
References
[1.] Pezzagno G, Imbriani M, Ghittori S, Capodaglio E. Inhalation anesthetics. In: Alessio L, Berlin A, Boni M, Roi R, Eds. Luxembourg: Commission of the European Communities--Industrial Health and Safety, 1989; pp 48-76.
[2.] Figuerova WG, Raszkowski R, Wells W. Lung cancer in chloromethyl ether workers. N Engl J Med 1973; 288:1096.
[3.] Baden JM, Mazze RI, Wharton RS, Hitt BA, Simmon VF, Mazze R. Mutagenicity of halogenated ether anesthetics. Anesthesiol 1977; 46:346.
[4.] Eger EI, White AE, Brown CL, Biava CG, Corbett TM, Stevens WC. A test of the carcinogenicity of enflurane, isoflurane, halothane, methoxyflurane and nitrous oxide in mice. Anesth Analg 1978; 57:678.
[5.] White AE, Takehisa S, Eger EI, Wolff S, Stevens WC. Sister chromatid exchanges induced by inhaled anesthetics. Anesthesiol 1979; 50:426.
[6.] Horats K, Kessler G, Van Kreybig T. Wirkund von Ethrane auf die fertilitat and vorgeburtliche entwieklung der ratte. Berlin: Springer-Verlag, 1974, pp 82.
[7.] Kessler G, Kreybig T. Effect of enflurane of prenatal development of rats. Anaesth Intensivm Prax 1975; 13:301.
[8.] Peduto VA, Bernabei PA, Pieracciuoli E, Novelli GP. Effect of prolonged exposure to non-anesthetic amount of enflurane on leukopoiesis and glucuronoconjunction. Acta Anaesth Ital 1977; 28:15.
[9.] Berlin S, Collins GI, Adam N. Effect of low concentrations of enflurane on probability learning. Br J Anaesth 1978; 50:1179.
[10.] Cook TL, Smith M, Winter PM, Starkweather JA, Eger EI. Effect of subanesthetic concentrations of enflurane and halothane on human behavior. Anesth Analg 1978; 57:434.
[11.] Imbriani M, Ghittori S, Zadra P, Imberti R. Biological monitoring of the occupational exposure to halothane (fluothane) in operating room personnel. Am J Ind Med 1991; 20:103-12.
[12.] National Institute for Occupational Safety and Health. Criteria for a recommended standard. Occupational exposure to waste anesthetic gases and vapor. Publication no. DHEW 77; 1977, p. 74.
[13.] ACGIH. Exposure indices. Proposed by American Conference of Governmental Hygienists for 1991-92. Cincinnati, OH: ACGIH, 1991.
[14.] ACGIH. Documentation of the threshold limit values and biological exposure indices. Cincinnati, OH: ACGIH, 1986.
[15.] Carlsson P, Ekstrand J, Hallen B. Plasma fluoride and bromide concentrations during occupational exposure to enflurane or halothane. Acta Anaesthesiol Scand 1985; 29:669-73.
[16.] Chase RE, Holaday V, Fiserova-Bergerova V, Saidman MD, Mack MD. The biotransformation of exthrane in man. Anesthesiol 1971; 35:262.
[17.] Cousin MJ, Mazze RI. Biotransformation of enflurane (ethrane) and isoflurane (forane). Int Anesthesiol Clin 1974; 12:111.
[18.] Strube PJ, Hulands GH, Halsey MJ. Serum fluoride in morbidly obese patients: enflurane compared with isoflurane anaesthesia. Anaesthesia 1987; 42:685.
[19.] Imbriani M, Ghittori S, Pezzagno G, Capodaglio E. Nitrous oxide ([N.sub.2]O) in urine as biological index of exposure in operating room personnel. Appl Ind Hyg 1988; 8:223-27.
[20.] Sonander HG, Stenqvist O, Nilsson K. Urinary [N.sub.2]O as a measure of biologic exposure to nitrous oxide anaesthetic contamination. Ann Occup Hyg 1983; 27:73-79.
[21.] Stevens MP, Walrand J, Buchet JP, Lauwerys R. Evaluation dell'exposition a l'enflurane et au protoxide d'azote en sall d'operation par des measures l'ambiance et des measures biologiques. Cah de Med de Trav 1987; 34:41-44.
[22.] Imbriani M, Ghittori S, Pezzagno G, Capodaglio E. Evaluation of exposure to isoflurane (forane). Environmental and biological measurements in operating room personnel. J Toxicol Environ Health 1988; 25:393-402.
[23.] Ghittori S, Imbriani M, Pezzagno G, Capodaglio E. The urinary concentration of solvents as biological indicator of exposure: proposal for the biological equivalent exposure for nine solvents. Am Ind Hyg Assoc J 1987; 48:786-90.
[24.] Imbriani M, Ghittori S, Pezzagno G, Capodaglio E. Urine-air partition coefficients for some industrially important substances. G Ital Med Lav 1985; 7:133-40.
[25.] Lauterberger WJ, Kring CB, Morello JA. Theory of passive monitors. In: Kelley WD, Ed., Dosimetry for chemical and physical agents. Ann Am Conf Ind Hyg 1981; 1:91-99.
[26.] Wilson HK. Breath analysis. Physiological basis and sampling techniques. Scand J Work Environ Health 1986; 12:174.
[27.] Krapez JR, Saloojee V, Hind CJ, Hackett GM, Cole PV. Blood concentrations of nitrous oxide in theatre personnel. Br J Anaesth 1980; 52:1143.
[28.] Halsey MJ, Sawyer DC, Eger EI, Bahlman SH, Impelman DMK. Hepatic metabolism of halothane, methoxyflurane, cyclopropane, ethrane and forane in miniature swine. Anesthesiol 1971; 35:43.
[29.] Ekstrand J. Use of pharmacokinetic models and techniques in fluoride research. In: Shupe J, Petterson H, Leone N, Eds., Fluorides. Effects on vegetations, animals and humans. Salt Lake City, UT: Paragon Press, 1983.
[30.] Fiserova-Bergerova V. Biokinetics of inhalation anesthetics. In: Capodaglio E, Mapelli G, Eds., Atti convegno:rischio professionale da anestetici per inalazione. Pavia, Italy: La Goliardica Pavese: 1988; 1:1-40.
[31.] Cattaneo AD, Rovatti M, Peloso A, Zattoni J, Tomellini M. Gas and vapour pollution in operating rooms. Blood and environmental enflurane concentrations. Acta Anaesth Italica 1983; 34:1087. [32.] Rosenberg J, Fiserova-Bergerova V, Lowry L. Biological monitoring. IV. Measurements in urine. Appl Ind Hyg 1989; 4:16-20.
COPYRIGHT 1994 Heldref Publications
COPYRIGHT 2004 Gale Group