METHOD OF PREPARATION
1. Calculate the required quantity of each ingredient for the total amount to be prepared.
2. Accurately weigh and/or measure each ingredient.
3. Mix the ergotamine tartrate, lactose, and sucrose until the mixture is uniform.
4. Moisten the powders with a 1:1 mixture of the alcohol: water mixture until a dough consistency is obtained.
5. Separate the tablet-triturate mold and place the cavity plate on a hard surface, such as a pill tile.
6. Press the drug mixture into the cavities until the mixture is uniform and firm.
7. Place the cavity plate over the peg plate of the mold, and gently press the cavity plate down over the pegs. Leave the formed tablets on top of the pegs to air dry.
8. Remove the tablets carefully, package and label.
PACKAGING
Package in a well-closed, light0resistant container.
LABELING
Use only as directed. Keep out of reach of children.
STABILITY
A beyond-use date of 6 months can be used for this preparation.1
USE
Ergotamine tartrate sublingual tablets are used in the treatment of migraine headaches.2
QUALITY CONTROL
Quality-control assessment can include overall average weight and individual weight variation, disintegration and dissolution observations, active-drug assay, physical appearance (color, uniformity), physical stability (discoloration, crumbling).
DISCUSSION
Ergotamine tartrate was formerly available as Ergostat Sublingual Tablets, which contained hydroxypropyl cellulose, FD&C yellow No. 6, aluminum lake, lactose, magnesium stearate, mannitol, artificial peppermint flavor, pregelatinized starch, saccharin, saccharin sodium, and starch. Ergostat tablets, formerly manufactured by Parke-Davis and later by Eli Lilly, were primarily used in the treatment of migraine headaches; ergostat tablets became unavailable in 1951.
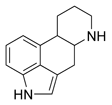
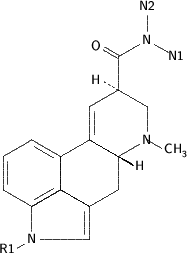
Ergotaminetartrate [(C^sub 33^H^sub 35^N^sub 5^O^sub 5^)^sub 2^ X C^sub 4^H^sub 6^O^sub 6^, MW 1313.4] occurs as colorless crystals or as a white to yellowish-white, crystalline odorless powder. It melts at about 180 deg C with decomposition. It is soluble 1 g in about 3200 mL of water; in the presence of a slight excess of tartaric acid, 1 g dissolves in about 500 mL of water; it is slightly soluble in alcohol. Ergotamine tartrate should be stored in well-closed, light-resistant containers.1-2
Lactose (C^sub 12^H^sub 22^O^sub 11^, MW 342.30, milk sugar, saccharum lactis) is available either anhydrous or as the monohydrate. It is soluble in water to the extent of about 1 g in 4.63 mL but is practically insoluble in ethanol. If stored under conditions of greater than 80% humidity, mold growth may occur. Lactose will exhibit a Maillard-type reaction (a brownish discoloration) when heated with compounds that contain a primary amine group. This reaction occurs more rapidly under alkaline conditions.3
Sucrose (beet sugar, cane sugar, refined sugar, saccharose) is obtained from sugar cane, sugar beet, or other sources. It occurs as colorless crystals, crystalline masses or blocks, or as a white crystalline powder that is odorless and has a sweet taste. When finely divided, it is hygroscopic and can absorb up to 1% water. Sucrose is soluble in water 1:0.5, and 95% ethanol 1:170, and is practically insoluble in chloroform. Sucrose is stable at room temperature.4
Alcohol (C^sub 2^H^sub 5^OH, MW 46.07, ethyl alcohol, ethanol, grain alcohol) is a clear, colorless mobile and volatile liquid with a slight, characteristic odor and a burning taste. Alcohol USP refers to 95% ethanol, and dehydrated alcohol refers to 99.5% alcohol. It has a specific gravity between 0.812 and 0.816, and its boiling point is 78.15*C. It is miscible with chloroform, glycerin, and water. It should be stored in a cool place.5
Purified water is water that is obtained by distillation, ion exchange, reverse osmosis, or some other suitable process. It is miscible with most polar solvents and is chemically stable in all physical states (ice, liquid, and steam).6
References
1.United States Pharmacopeial Convention, Inc. United States Pharmacopeia 26/National Formulary2l. Rockville, Maryland:US Pharmacopeial Convention, Inc.; 2002:2197-2201, 2560.
2. [No author listed.] Physicians' Desk Reference. 48th ed. Montvale, New Jersey:Medical Economics Company, Inc.; 1994:1739-1740.
3. Kibbe AH. Lactose: In: Kibbe AH, ed. Handbook of Pharmaceutical Excipients. 3rd ed. Washington, DC:American Pharmaceutical Association; 2000:276-285.
4. Hamlow EE, Armstrong NA, Pickard A. Sucrose. In: Kibbe AH, ed. Handbook of Pharmaceutical Excipients. 3rd ed. Washington, DC:American Pharmaceutical Association; 2000:539-543.
5. Weller PJ. Alcohol. In: Kibbe AH, ed. Handbook of Pharmaceutical Excipients. 3rd ed. Washington, DC:American Pharmaceutical Association; 2000:7-9.
6. Ellison A, Nash RA, Wilkin MJ. Water. In: Kibbe AH, ed. Handbook of Pharmaceutical Excipients. 3rd ed. Washington, DC:American Pharmaceutical Association; 2000:580-584.
Copyright International Journal of Pharmaceutical Compounding May/Jun 2003
Provided by ProQuest Information and Learning Company. All rights Reserved