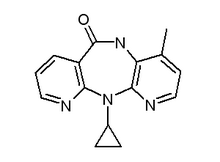
Nevirapine
Nevirapine, with trade name of Viramune®, is a controversial non-nucleoside reverse transcriptase inhibitors (NNRTIs) meant to treat HIV and AIDS. more...
History
Nevirapine was the first NNRTI (tenth antiretroviral) approved by the Food and Drug Administration (FDA). It was approved Jun 21, 1996 for adults and Sep 11, 1998 for pediatrics.
Controversy
Recently, concerns have surfaced that nevirapine may cause long-term resistance to further AIDS treatments and potentially lethal side effects like liver damage when taken in multiple doses over time.
These negative effects, however, were known upon the drugs release in 2000 when the FDA put a black box label on nevirapine warning that it could cause liver damage or rupturing of the blood and flesh.
After the negative assessment by the FDA, the drug's manufacturer, Boehringer Ingelheim, removed the drug from the intended market of pregnant American women. They moved the drug, instead, to the African market. Evidence of the drugs negative effects was found and buried in subsequent experimentation of the drug in Uganda.
Major controversy arose when South African President Thabo Mbeki accused the drug's maker, the United States, and President George W. Bush (whose $500 million plan for the African AIDS problem was grounded in the supposed antiretroviral nevirapine) of using Africans as "guinea pigs." Several African and African-American leaders, notably Rev. Jesse Jackson, followed Mbeki in denouncing the United States Government and health industry for pushing the drug in Africa.
Read more at Wikipedia.org