JP, a healthy 54-year-old woman, was running with friends when she experienced a sudden, severe headache; vomiting; left-sided hemiplegia; and a decreased level of consciousness. After she arrived at the hospital, her condition continued to deteriorate. She became less responsive, had difficulty maintaining an airway, and required intubation. Neurologically, her pupils were equal but sluggishly reactive to light; she had no cough reflex, a weak gag response, and withdrew only to noxious stimuli. Initial vital signs included a heart rate of 41 beats/min and a blood pressure of 200/96 mm Hg.
A 12-lead electrocardiogram revealed a type I second-degree heart block with no ST changes or Q waves. Computed tomography (CT) of the head showed a large subarachnoid hemorrhage (SAH) and intraparenchymal blood. Despite clinical and CT findings, no aneurysm was detected by emergent cerebral angiography. Although this result may seem unusual, the cause of up to 20% of SAHs cannot be detected angiographically. (1) JP was transferred to the trauma neurosurgical intensive care unit for hemodynamic and neurological monitoring and nursing assessment. According to the Hunt and Hess grading scheme (2) (scale 0-V; Table 1), she had a grade IV cerebral aneurysm. After placement of a central venous catheter and a ventriculostomy drain, JP was taken urgently to the operating room for clot evacuation. In surgery, an aneurysm of the right middle cerebral artery was located and clipped.
Postoperatively, JP returned to the trauma neurosurgical intensive care unit for observation and administration of nimodipine (Nimotop), dexamethasone (Decadron), and "triple H" therapy. Daily transcranial Doppler (TCD) studies were ordered to detect early signs of vasospasm.
Although Ms P was hypertensive at the time of admission, her systolic blood pressure dipped into the low 100s after surgery. Because cerebral perfusion pressure is equal to mean arterial pressure minus intracranial pressure, perfusion to her brain decreased dramatically despite an intracranial pressure that was never markedly elevated. A dopamine infusion was started, and the dosage was adjusted to maintain systolic blood pressure greater than 160 mm Hg and cerebral perfusion pressure greater than 75 mm Hg. On the third day after surgery, JP's central venous pressure remained persistently lower than the target level of 10 mm Hg despite a maintenance intravenous fluid rate of 200 mL/h and repeated boluses of 500 to 1000 mL of isotonic sodium chloride solution.
Although she had no clinical indications of vasospasm, TCD imaging on day 8 indicated decreased blood flow. Cerebral angiograms showed no abnormalities suggestive of vasospasm. Because increasing doses of dopamine did not maintain her systolic blood pressure at the desired level of 160 to 200 mm Hg, JP was switched to treatment with phenylephrine. When this intervention also was inadequate, administration of dobutamine was added to augment cardiac output.
On day 10, JP's nurse noted clinical signs of severe vasospasm, and cerebral angiograms indicated vasospasm. Balloon angioplasty was performed to reduce vessel narrowing. Subsequent TCD studies and angiograms indicated no further vasospasm. On day 12, JP returned to surgery for placement of a permanent ventriculoperitoneal shunt. Three weeks after admission, JP was functioning fairly independently, with some residual left-sided weakness, and she was discharged to a rehabilitation facility.
Subarachnoid Hemorrhage Incidence
In the United States, each year approximately 25 000 to 30 000 persons (6-11 per 100 000) experience SAH. (3-7) Rupture of cerebral artery aneurysms accounts for 50% to 80% of these cases. (5,6,8) Other causes include bleeding from a cerebral tumor, cocaine abuse, hypertensive cerebral hemorrhage, anticoagulant therapy, and ruptured arteriovenous malformations. (6,8) SAH has also been noted on the initial CT scans of approximately 40% of all patients with major head injuries. (9) However, traumatic SAHs tend to be less debilitating than those caused by aneurysmal rupture and are often associated with other, more devastating, intracranial abnormalities.
Etiology
Aneurysms are bulges or areas of outpouching that occur at a site of weakness in a vessel wall, most commonly at the junction or bifurcation of 2 vessels. Many are congenital; others are associated with vascular disorders caused by uncontrolled hypertension, bacterial or fungal infections, and connective tissue diseases such as Marfan or Ehlers-Danlos syndromes. (3) Regardless of the cause, when an aneurysm ruptures, arterial pressure forces blood into the subarachnoid space between the arachnoid mater and the surface of the brain. Free blood then travels through the fissures, into the basal cisterns, and across the surface of the brain. (6,8) When clotted, this blood can interfere with the circulation and reabsorption of cerebrospinal fluid (CSF), potentially causing an obstructive hydrocephalus and increased intracranial pressure.
Clinical Findings
Blood in the subarachnoid space is a powerful meningeal irritant, and it is this irritation that causes most of the initial signs and symptoms of SAH. (6) Findings vary from insignificant to serious, depending on the extent of hemorrhage. (8) The characteristic initial symptom of patients with SAH is sudden headache described as the most painful ever experienced. (6,8,10) Changes in mental status are common. Initially, patients may vary from awake and alert to deeply comatose. Clinical findings include nuchal rigidity, photophobia, nausea and vomiting, hypertension, electrocardiographic changes, pyrexia, cranial nerve deficits, visual changes, sensory or motor deficits, fixed and dilated pupils, seizures, herniation, and sudden death. (6,8,10)
Outcome
Both severity of disability and length of rehabilitation after SAH are linked to the extent of the initial injury. (7,8) SAH takes a tremendous physical and emotional toll on patients and their families. Sadly, 35% of patients who experience SAH will not survive the initial hemorrhage. (11) Patients who outlive the original hemorrhage face a multitude of potential complications in the acute phase as well as functional limitations that require extensive rehabilitation and may result in permanent deficits.
Vasospasm After Rupture of Cerebral Aneurysm Incidence
Symptomatic vasospasm, also referred to as delayed ischemic deficits, (12) delayed ischemic neurological defects, (13) and cerebrovascular spasm, (4) is the most common serious complication after aneurysmal rupture and is the leading cause of death for patients who survive the rupture. (14) In fact, vasospasm-induced narrowing of cerebral vessels is estimated to occur in 70% to 90% of patients hospitalized for SAH. Vasospasm is symptomatic in 30% of these patients. (4-6,11,14-17)
Definition
Vasospasm has been described as a sustained arterial contraction unresponsive to vasodilator drugs. (17) This condition is commonly classified as either angiographic or clinical. Angiographic vasospasm refers to visible narrowing of the dye column in an artery, as shown on cerebral angiograms. Clinical vasospasm is the functional manifestation of cerebral ischemia produced by this arterial narrowing. (5,6)
Episodes of vasospasm can be benign or devastating. Among patients with SAH, 70% have angiographic evidence of vasospasm but no clinical evidence of vasospasm. The remainder (those with clinical vasospasm) have changes in findings on neurological examination. (14) Clinically, the onset of new or worsening neurological signs or symptoms is the most reliable indicator of vasospasm. (5) Assessment findings may be subtle and include headache, lethargy, and intermittent disorientation, which can progress to focal neurological deficits such as hemiparesis and speech dysfunction. (4) Deficits vary according to the degree of vessel constriction and the cerebral artery affected. (5) If the reduction in cerebral blood flow is severe or remains untreated, permanent disability and even death can occur. (4,6,14)
Clinical Course
A well-accepted predictive factor of vasospasm is the initial size of the SAH evident on CT scans. The presence of diffuse, thick, subarachnoid blood, particularly around the base of the brain, is correlated with a high incidence and severity of vasospasm; CT findings of focal, thin blood are associated with a lower vasospasm rate. (4,11,13) The scoring systems most commonly used (18) to grade acuity of SAHs are the Fisher Scale (Table 2) and the Hunt and Hess grading scheme (Table 1). For both of these instruments, studies have indicated a direct correlation between the severity of the clinical grade of cerebral aneurysm and the incidence of vasospasm after SAH. (6)
Vasospasm is also more prevalent if hemorrhage occurs in one of the vessels of the circle of Willis (5) (see Figure). Occasionally, spasm spreads across the entire distribution area of a cerebral artery, but it is usually localized to the vessels closest to the site of leakage. (8)
Although the incidence of vasospasm peaks at 5 to 12 days after rupture of the aneurysm, the actual time of onset can be anywhere from 3 to 21 days after hemorrhage. (4,5,10,17) Once initiated, the condition generally persists for 12 to 16 days. (5,10)
[FIGURE OMITTED]
Pathophysiology
The etiology and pathophysiology of cerebral vasospasm after SAH are complex and are only partially understood. Precise molecular mechanisms of vasospasm remain to be elucidated, but they include a combination of both increased constrictor mechanisms and decreased dilator functions. (5,11,14) Several theories have been offered to explain the multifaceted processes known to be involved.
Considerable evidence supports the hypothesis that oxyhemoglobin plays a primary role in the development of cerebral vasospasm associated with aneurysmal SAH. (13) After the initial hemorrhage, erythrocytes trapped in the subarachnoid cisterns slowly hemolyze, releasing oxyhemoglobin and other by-products of red cell lysis (eg, bilirubin and methemoglobin) to circulate within the subarachnoid space. (4) These spasminogens increase the influx of calcium into the vascular smooth muscle, altering myocyte function and causing prolonged contraction and vessel constriction. (4,5,13,20)
Oxyhemoglobin also contributes to release of free radicals and peroxidation of lipids. (4,13) These changes promote the synthesis of vasoactive eicosanoids and endothelin and inhibit endothelium-dependent relaxation of the arterial wall. (13,21) In addition, failure of nitric oxide--dependent relaxation mechanisms within cerebral arteries may be involved in vasospasm. (11) Studies of CSF after SAH indicated elevations in levels of free fatty acids, implicating these agents in the development of vasospasm as well. (22)
Evidence also supports the theory that vasospasm is associated with an inflammatory response in the subarachnoid space, because prostaglandins, thromboxane [A.sub.2], leukotrienes, and histamine are all present in the CSF after hemorrhage. (13) Another hypothesis is that sloughing of endothelial cells, which results in structural changes in the inner layer of the arterial wall, allows vasoconstrictive substances, including serotonin, catecholamines, and oxyhemoglobin, to reach the medial layer, thus producing vasospasm. (5) In addition to these mechanisms, the pathogenesis of cerebral vasospasm involves in part either an overexpression or an underexpression of specific proteins within the vessel wall. (23)
Regardless of the cellular mechanisms involved, SAH causes spasm of local vessels and disturbance of cerebral autoregulation. This impaired autoregulation reduces the ability of the brain to maintain adequate cerebral perfusion pressures and may also enhance the propensity of cerebral arteries to constrict in response to vasoactive substances. (22) The reduced size of the vessel lumen restricts blood flow to the brain tissue, causing temporarily irreversible vasoconstriction. (5,13) Prolonged increases in cerebral vascular resistance and decreased regional blood flow are followed by ischemia and potential infarction of the cerebral parenchyma. (6)
The blood-flooded subarachnoid space is clearly a "pathological factory" that stimulates vasospasm through a variety of mechanisms. Fortunately, this multiplicity of biochemical reactions offers many potential targets for antispastic drugs. (11)
Diagnosis
Signs and Symptoms Clinically, vasospasm can be recognized by means of frequent, thorough neurological examinations. Each patient's baseline assessment should be clearly documented. Signs and symptoms may be subtle, generally have a gradual onset, and tend to wax and wane. (5,6) These findings can include changes in level of consciousness, headache, periods of disorientation, inappropriate behavior, language impairment, hemiparesis, and seizures. (4,6) Nurses must quickly detect and report any changes in a patient's neurological status. If unrecognized, the deficits associated with vasospasm can become permanent disabilities. (4)
TCD Imaging TCD imaging is a noninvasive technique often used to diagnose vasospasm. (5) TCD studies are performed at the bedside, usually on a daily basis, after SAH. This imaging technique is used to measure the velocity of blood flow through segments of arterial vessels. Noting trends in flow velocity allows early identification of patients at risk for vasospasm, although many factors can affect the validity of TCD findings. (5,6) When correlated with the results of a patient's neurological examination, regular TCD studies can facilitate the rapid diagnosis and treatment of cerebral vasospasm. (5,6) TCD findings are most reliable as predictors of vasospasm of the middle cerebral and internal carotid arteries. (24)
Angiography Although use of both CT angiography and magnetic resonance angiography have been investigated, standard angiography remains the definitive study for the diagnosis of cerebral vasospasm. (5,11,14) Spasm is indicated by evidence of narrowing of arterial vessels, and these changes can be apparent on angiograms before the advent of clinical signs and symptoms. (4,6,14)
Interventions for Vasospasm
For more than 50 years, researchers and clinicians have sought to discover optimal ways to prevent and manage cerebral vasospasm, but no single therapy has been identified. (4) However, most authors (4,25,26) agree that clipping the aneurysm or using an endovascular intervention (eg, coiling, stenting, or angioplasty) within 48 hours of the initial hemorrhage is imperative to minimize the occurrence of both vasospasm and rebleeding. In addition, early surgical clot removal limits the amount of oxyhemoglobin released, reducing the incidence of vasospasm. (4)
Administration of calcium channel blockers and use of triple H therapy are both widely accepted interventions after clipping or coiling. (11) These measures appear to be most effective when used prophylactically. (6) Once vasospasm is established, even potent vasodilators do not effectively reverse the spasm. (17) Treatment of refractory vasospasm includes balloon angioplasty and direct arterial instillation of papaverine.
Calcium Channel Blockers
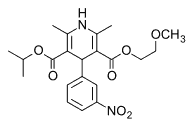
Nimodipine (Nimotop) is the calcium channel blocker of choice for cerebral vasospasm. (17) Because the drug is lipid soluble, it easily crosses the blood-brain barrier. (6) Nimodipine decreases spasm in the cerebral vascular bed and can lead to improvement in long-term outcomes after SAH. (4,10) After hemorrhage, patients are prophylactically given oral nimodipine at a dosage of 60 mg, every 4 hours, for 21 days. (6,10)
Triple H Therapy Use of triple H therapy--also referred to as HHH, THT, prophylactic hyperdynamic postoperative fluid therapy, and hemodynamic augmentation--has become routine practice in patients with SAH. (12,18,25,27,28) The 3 components of triple H therapy are hypertension, hypervolemia, and hemodilution. Importantly, these interventions affect the symptoms of vasospasm rather than the underlying cause. (27) A hypertensive state is maintained through the use of vasopressors. Hypervolemia is obtained by using volume expansion, which subsequently produces hemodilution. These maneuvers are all designed to increase cerebral perfusion pressure, improve blood flow to the brain, and decrease the risk of ischemia. (4,6,8,10,16)
Triple H therapy is generally started as soon as the aneurysm has been secured or clipped. Occasionally, a modified version of triple H therapy is used before securement of the aneurysm in order to prevent rebleeding or rupture. Therapy is continued for at least 14 days after the initial hemorrhage, the most common period for vasospasm. (6)
After SAH, patients lose autoregulatory function in ischemic brain tissue. Induced hypertension is designed to increase cerebral blood flow and thus enhance perfusion pressure. (17) In order to accomplish this goal, target systolic blood pressures may be as high as 200 mm Hg. In addition to fluids, vasopressor and inotropic agents such as dopamine, phenylephrine, and dobutamine are generally required to maintain systolic blood pressure at this elevated level. (10,12) Therapeutic hypertension is only initiated once the aneurysm has been surgically clipped or coiled. If the aneurysm has not yet been repaired surgically or is considered inoperable, systolic blood pressure is kept between 120 and 150 mm Hg to minimize the risk of rupture and rebleeding. (6,29) Many patients are surprisingly resistant to induced hypertension despite large fluid volumes and vasopressor therapy, and achieving the target systolic blood pressure is often difficult.
Hypervolemia is produced by infusing intravenous crystalloids or colloids sufficient to optimize a patient's Starling curve. Determining the point at which optimization occurs requires careful hemodynamic assessment. General parameters include pulmonary artery wedge pressure of 14 to 20 mm Hg, cardiac index (calculated as cardiac output in liters per minute divided by body surface area in square meters) of 2.2 or greater, and central venous pressure of 10 to 12 mm Hg. (12) Hetastarch (Hespan) and albumin have both been used to expand blood volume, but most clinicians recommend using simple isotonic crystalloid solutions. (10)
The goal for hemodilution is a hematocrit of 0.33 to 0.38, which is achieved by dilution with intravenous fluids. (6) Occasionally patients are anemic as a result of intraoperative blood loss or preexisting conditions. In this situation, packed red blood cells must be administered to reach the target hematocrit to ensure optimal cerebral oxygenation.
Although triple H therapy has become the standard of care in many institutions, its use is not without controversy. In some studies of patients with vasospasm, improvement in neurological status occurred after initiation of triple H therapy, but not every patient responds to this therapy. (6,25) Importantly, despite apparent reversal of neurological deficits, no evidence indicates that morbidity or mortality is reduced after triple H therapy. (15,25,28) In fact, in studies (28) in pigs, outcomes were worse in the animals subjected to triple H therapy after induced SAH. Triple H therapy is also associated with a substantial number of complications. (6)
The complications include pulmonary edema, myocardial ischemia, congestive heart failure, electrolyte imbalances, coagulopathies, and rupture of unsecured aneurysms. (10,16,25,27-31) Costs associated with triple H therapy are substantial and are related to additional nursing care requirements (Table 3), extended critical care unit lengths of stay, aggressive fluid management, frequent complications, and expenses associated with placement and monitoring of pulmonary artery catheters. (6,25)
Angioplasty Vasospasm unresponsive to nimodipine and maximal triple H therapy requires more invasive interventions. Balloon angioplasty directly treats narrowed cerebral vessels by widening the stenotic segment. This widening can dramatically improve a patient's neurological status. (10) Fortunately, vessels rarely restenose after angioplasty. Most likely, along with opening the vessel, the balloon causes functional impairment of smooth muscle in the vessel wall, reducing or eliminating further vasospasm. (10,14) An important limitation of balloon angioplasty is that it can be used to widen only larger vessels, such as the distal internal carotid, proximal middle cerebral, proximal anterior cerebral, distal vertebrobasilar, and proximal posterior cerebral arteries. The most devastating complication associated with angioplasty is vessel rupture. (10,14)
Papaverine Papaverine, a synthetic alkaloid of opium, has an antispasmodic effect on smooth muscle. For patients with vasospasm refractory to triple H therapy, selective intra-arterial injection of papaverine directly into spastic vessels can reverse the spasm. (11,14,18) The main advantage of papaverine is that it can be used to treat distal vessels, those beyond the reach of an angioplasty balloon. (14) Use of this agent is controversial; significant increases in intracerebral pressure can occur after instillation, and some investigators (10) think that this intervention has no proven long-term benefit.
New Directions in Management of Vasospasm
Several newer therapies are being investigated in hope of eventually providing better treatment for patients with vasospasm.
Recombinant Tissue Plasminogen Activator Administration of intracisternal recombinant tissue plasminogen activator after aneurysm clipping is considered controversial because complications include devastating cerebral hemorrhage and bleeding from the craniotomy site. (4) After aneurysm clipping, recombinant tissue plasminogen activator can be injected into the basal cisterns to dissolve clots and reduce the amount of spasmogenic substances present in the CSF. (4) Despite the substantial risks, an incidence of vasospasm of less than 5% after this therapy has been reported. (11)
Oxygen Free Radical Scavengers With the breakdown of erythrocytes after SAH, large quantities of oxyhemoglobin are released into the sub-arachnoid space. This release in turn stimulates the production of oxygen free radicals, which can damage endothelial and neuronal membranes and lead to vasospasm. In theory, the administration of a scavenger of oxygen free radicals, such as tirilazad mesylate, would decrease the number of free radicals available. However, in clinical trials, use of this intervention did not result in a reduction in the incidence of vasospasm. (11)
Nitric Oxide Blockers Another possible treatment for vasospasm is the use of nitric oxide blockers. Researchers have theorized that cerebral artery vasospasm is caused in part by a failure of the nitric oxide--dependent relaxation mechanism. In animal models, agents that blocked nitric oxide decreased arterial spasm. (11)
Hypothermia Elevated intracranial pressure and vasospasm after SAH are more easily managed in patients who have mild hypothermia. However, achieving a hypothermic state may be difficult and requires intensive nursing care. In addition, rewarming is associated with significant adverse effects such as hypotension and increased systemic vascular resistance, which can worsen cerebral ischemia. (32,33)
Intra-Aortic Balloon Pumps Intra-aortic balloon pumps have been used to optimize cardiac performance in patients with SAH who have heart failure refractory to fluids and vasopressors. (16) Use of the pump allows continuation of triple H therapy in patients who would otherwise be unable to tolerate it. The pump decreases cardiac workload, reduces myocardial oxygen consumption, and improves perfusion to coronary arteries and systemic tissues. Use of intra-aortic balloon pumps has not been studied in patients with vasospasm, and its use involves significant risks. (16,34)
Gene Therapy Future treatment of vasospasm may involve the use of gene therapy. In this therapy, genetic material is introduced into cells to direct the cells to prevent or treat disease. Because vasospasm is a relatively short-term process, it is considered compatible with the temporary aspects of somatic gene therapy. (23) Investigators think that genetic material could be injected into the CSF, where vessels at or near the circle of Willis would be accessible to gene delivery. These genes would then interact with cells in the vessel walls to prevent or relieve vasospasm. To date, this therapy has been tested only in animal models. (23)
Summary
Despite every effort, 13% of patients with SAH still die or are permanently disabled as a consequence of vasospasm. (11) Optimal outcome after aneurysmal SAH depends on careful assessment and management of patients throughout the course of hospitalization. Critical care nurses play a crucial role in this process. Efforts continue to understand the cascade of events that lead to cerebral vasospasm and to develop more effective treatments. Many therapies, both traditional and new, are being investigated to reduce the incidence of symptomatic vasospasm and improve the lives of patients who experience this devastating condition.
References
1. Wilcock D, Jaspan T, Holland J, Cherryman G, Worthington B. Comparison of magnetic resonance angiography with conventional angiography in the detection of intracranial aneurysms in patients presenting with subarachnoid haemorrhage. Clin Radiol. 1996;51:330-334.
2. Hunt WE, Hess RM. Surgical risk as related to time of intervention in the repair of intracranial aneurysms. J Neurosurg. 1968;28:14-20.
3. Pfohman M, Criddle L. Epidemiology of intracranial aneurysm and subarachnoid hemorrhage. J Neurosci Nurs. 2001;33:39-41.
4. Bell TE, Kongable GL. Innovations in aneurysmal subarachnoid hemorrhage: intracisternal t-PA for the prevention of vasospasm. J Neurosci Nurs. 1996;28:107-113.
5. Falyar CR. Using transcranial Doppler sonography to augment the neurological examination after aneurysmal subarachnoid hemorrhage. J Neurosci Nurs. 1999;31:285-293.
6. Rusy KL. Rebleeding and vasospasm after subarachnoid hemorrhage: a critical care challenge. Crit Care Nurse. February 1996;16:41-48.
7. Kirkness CJ, Thompson JM, Ricker BA, et al. The impact of aneurysmal subarachnoid hemorrhage on functional outcome. J Neurosci Nurs. 2002;34:134-141.
8. Rees G, Shah S, Hanley C, Brunker C. Subarachnoid haemorrhage: a clinical overview. Nurs Stand, July 3-9. 2002;16:47-54.
9. Servadei F, Murray GD, Teasdale GM, et al. Traumatic subarachnoid hemorrhage: demographic and clinical study of 750 patients from the European Brain Injury Consortium survey of head injuries. Neurosurgery. 2002;50:261-267.
10. Corsten L, Raja A, Guppy K, et al. Contemporary management of subarachnoid hemorrhage and vasospasm: the UIC experience. Surg Neurol. 2001;56:140-148.
11. Chiappetta F, Brunori A, Bruni P. Management of intracranial aneurysms: "state of the art." J Neurosurg Sci. 1998;42(1 suppl 1):5-13.
12. Aiyagari V, Cross DT III, Deibert E, Dacey RG Jr, Diringer MN. Safety of hemodynamic augmentation in patients treated with Guglielmi detachable coils after acute aneurysmal subarachnoid hemorrhage. Stroke. 2001;32:1994-1997.
13. Warnell P. Advanced concepts in the management of cerebral vasospasm associated with aneurysmal subarachnoid hemorrhage. Axone. 1996;17:86-92.
14. Smith TP, Enterline DS. Endovascular treatment of cerebral vasospasm. J Vasc Interv Radiol. 2000;11:547-559.
15. Romner B, Reinstrup P. Triple H therapy after aneurysmal subarachnoid hemorrhage: a review. Acta Neurochir Suppl. 2001;77:237-241.
16. Apostolides PJ, Greene KA, Zabramski JM, Fitzgerald JW, Spetzler RF. Intra-aortic balloon pump counterpulsation in the management of concomitant cerebral vasospasm and cardiac failure after subarachnoid hemorrhage: technical case report. Neurosurgery. 1996;38:1056-1059.
17. Levati A, Solaini C, Boselli L. Prevention and treatment of vasospasm. J Neurosurg Sci. 1998;42(1 suppl 1):27-31.
18. Hickey J, Buckley D. Cerebral aneurysms. In: Hickey J, ed. The Clinical Practice of Neurological and Neurosurgical Nursing. 5th ed. Philadelphia, Pa: Lippincott Williams & Wilkins; 2003;523-548.
19. Fisher CM, Kistler JP, Davis JM. Relation of cerebral vasospasm to subarachnoid hemorrhage visualized by computerized tomographic scanning. Neurosurgery. 1980;6:1-9.
20. Macdonald RL. Pathophysiology and molecular genetics of vasospasm. Acta Neurochir Suppl. 2001;77:7-11.
21. Chow M, Dumont AS, Kassell NF, Endothelin receptor antagonists and cerebral vasospasm: an update. Neurosurgery. 2002;51:1333-1341.
22. Pilitsis JG, Coplin WM, O'Regan MH, et al. Free fatty acids in human cerebrospinal fluid following subarachnoid hemorrhage and their potential role in vasospasm: a preliminary observation. J Neurosurg. 2002;97:272-279.
23. Findlay JM. Gene therapy for vasospasm. Acta Neurochir Suppl. 2001;77:83-87.
24. Suarez JI, Qureshi AI, Yahia AB, et al. Symptomatic vasospasm diagnosis after subarachnoid hemorrhage: evaluation of transcranial Doppler ultrasound and cerebral angiography as related to compromised vascular distribution. Crit Care Med. 2002;30:1348-1355.
25. Egge A, Waterloo K. Sjoholm H, Solberg T, Ingebrigtsen T, Romner B. Prophylactic hyperdynamic postoperative fluid therapy after aneurysmal subarachnoid hemorrhage: a clinical, prospective, randomized, controlled study. Neurosurgery. 2001;49:593-605.
26. Gumprecht H, Winkler R, Gerstner W, Lumenta C. Therapeutic management of grade IV aneurysm patients. Surg Neurol. 1997;47:54-58.
27. Choudhari K. Prophylactic hyperdynamic postoperative fluid therapy after aneurysmal subarachnoid hemorrhage: a clinical, prospective, randomized, controlled study [letter; comment]. Neurosurgery. 2002;50:1170-1172.
28. Dueck M, Ebel H, Volz M, Lee JY, Semmelmann G. Triple "H" therapy does not increase cerebral blood flow after induced subarachnoid hemorrhage in pigs: results of a pilot [.sup.99m]Tc-HMPAO-single photon emission computed tomography study. Acta Neurochir Suppl. 2001;77:243-246.
29. Hoh BL, Carter BS, Ogilvy CS. Risk of hemorrhage from unsecured, unruptured aneurysms during and after hypertensive hypervolemic therapy. Neurosurgery. 2002;50:1207-1211.
30. Krayenbuhl N, Hegner T, Yonekawa Y, Keller E. Cerebral vasospasm after subarachnoid hemorrhage: hypertensive hypervolemic hemodilution (triple-H) therapy according to new systemic hemodynamic parameters. Acta Neurochir Suppl. 2001;77:247-250.
31. Gupta D, Sharma BS, Gupta SK, Bapuraj R, Khosla VK. Postoperative hypertensive-hypervolaemic-haemodilution (triple H) therapy in the treatment of vasospasm following aneurysmal subarachnoid haemorrhage. Neurol India. 2000;48:126-131.
32. Inamasu J, Ichikizaki K. Mild hypothermia in neurologic emergency: an update. Ann Emerg Med. 2002;40:220-230.
33. Thome C, Schubert G, Piepgras A, Elste V, Schilling L, Schmiedek P. Hypothermia reduces acute vasospasm following SAH in rats. Acta Neurochir Suppl. 2001;77:255-258.
34. Rosen CL, Sekhar LN, Duong DH. Use of intra-aortic balloon pump counterpulsation for refractory symptomatic vasospasm. Acta Neurochir (Wien). 2000;142:25-32.
Katie Oyama, RN, BSN, CCRN
Laura Criddle, RN, MS, CCRN, CCNS
Katie Oyama is a staff nurse in the neuro/trauma intensive care unit at Oregon Health & Science University, Portland, Ore.
Laura Criddle is the clinical nurse specialist for the emergency department and the neuro/trauma intensive care unit at Oregon Health & Science University, Portland, Ore.
To purchase reprints, contact The InnoVision Group, 101 Columbia, Aliso Viejo, CA 92656, Phone, (800) 809-2273 or (949) 362-2050 (ext 532); fax, (949) 362-2049; e-mail, reprints@aacn.org.
COPYRIGHT 2004 American Association of Critical-Care Nurses
COPYRIGHT 2004 Gale Group