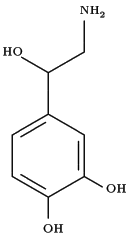
Approval of duloxetine hydrochloride by the Food and Drug Administration offers an additional drug for the treatment of major depressive disorder that affects both serotonin and norepinephrine.
In a statement announcing the approval, Dr. Stephen M. Stahl said duloxetine (Cymbalta), manufactured by Eli Lilly & Co., "offers physicians a new opportunity to help patients with depression, particularly those who experience the common physical symptoms of the disease, such as vague aches and pains."
But duloxetine "is not an analgesic, and it would be wrong to characterize it that way," Dr. Stahl told this newspaper.
Like selective serotonin reuptake inhibitors, duloxetine might take 2-8 weeks to take effect, said Dr. Stahl, who is chairman of the Neuroscience Education Institute, Carlsbad, Calif.; an adjunct professor of psychiatry at the University of California, San Diego; and a consultant to Lilly.
Venlafaxine hydrochloride (Effexor), also a selective serotonin and norepinephrine reuptake inhibitor, was approved in 1993. In addition to depression, venlafaxine, manufactured by Wyeth Pharmaceuticals, is indicated for the treatment of generalized and social anxiety disorders.
To date, no head-to-head trials have been conducted between venlafaxine and duloxetine.
In clinical trials, duloxetine was studied in doses of 40-120 mg, and the recommended daily dose is 60 mg. It was shown to effectively relieve depression as measured by the Hamilton Depression Rating scale. In support of its FDA application, Lilly studied duloxetine treatment in 6,000 adults with major depressive disorder, aged 18-83 years, as part of four randomized, double-blind, placebo-controlled studies, and in a 1-year open label safety study.
David Shaffer, a Lilly spokesperson, noted that the company has been presenting information about depression's "painful physical symptoms and Cymbalta for some time."
"If you look at the DSM-IV, painful physical symptoms are a part of depression, and Cymbalta is approved for the treatment of depression. It's not necessarily in the label in an explicit way. We did have secondary end points in many of our studies about painful physical symptoms," he said.
Two large, double-blind, placebo-controlled trials showed that duloxetine relieved diabetic neuropathic pain, Mr. Shaffer said. Those studies, which did not include depressed patients, show "an independent effect on pain that supports our assertion that the mechanism of action of duloxetine hitting on both serotonin and norepinephrine is believed to explain its effect on the painful physical symptoms of depression, as well as the emotional symptoms of depression."
Dr. Brendan Montano, an internist in Cromwell, Conn., said duloxetine is "a very worthy addition" to the drug choices that primary care physicians have for treating major depression.
"It's both balanced and potent with regard to serotonin and norepinephrine activity. There's a synergistic activity that helps the physical symptoms associated with depression," said Dr. Montano of the department of family medicine at the University of Connecticut. Farmington. In addition, he has served as a consultant to Lilly on duloxetine clinical trials.
In its statement announcing the approval, Eli Lilly said that it plans to petition the FDA for further indications for duloxetine for the treatment of diabetic neuropathic pain and stress urinary incontinence.
According to the drug's prescribing information, adverse events were similar in men and women studied, except when it came to sexual function.
Out of 1,916 participants (625 men, 1,291 women) in four placebo-controlled trials, a 2% incidence of sexual side effects was identified using the Arizona Sexual Experience Scale. But women did not experience more sexual dysfunction on duloxetine, compared with placebo.
In clinical trials, the most common side effects of duloxetine were nausea, dry mouth, constipation, decreased appetite, fatigue, sleepiness, and increased sweating.
The duloxetine label includes a warning about a possible increased risk of suicide, as requested by the FDA for this class of antidepressants.
Duloxetine should not be given to patients with any hepatic insufficiency, end-stage renal disease, or uncontrolled, narrow-angle glaucoma, or to patients with substantial alcohol use.
As a category C drug, duloxetine is not recommended for nursing women.
The drug has not been studied in children. The drug's effectiveness in hospitalized patients with major depression also has not been studied.
BY DEEANNA FRANKLIN
Senior Writer
COPYRIGHT 2004 International Medical News Group
COPYRIGHT 2004 Gale Group