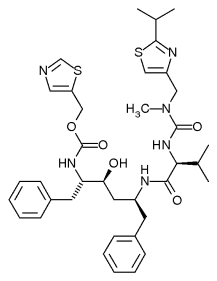
Norvir
Ritonavir, with trade name Norvir®, is an antiretroviral drug from the protease inhibitor class used to treat HIV infection and AIDS. more...
Ritonavir is frequently prescribed as the protease inhibitor component in HAART because it inhibits the same host enzyme that metabolizes NRTIs and NNRTIs. This inhibition leads to higher plasma concentrations of these latter drugs, allowing the clinician to lower their dose and frequency.
History
Ritanovir is manufactured as Norvir® by Abbott Laboratories. Research that led to the drug's development was financed in part by a $3,500,000 federal grant through the National Institutes of Health (NIH). The Food and Drug Administration (FDA) approved ritanovir on March 1, 1996, making it the seventh approved antiretroviral drug in the United States.
In 2003, Abbott raised the price of a Norvir course from USD $1.71 per day to $8.57 per day, leading to claims of price gouging by patients' groups and some members of Congress. Consumer group Essential Inventions petitioned the NIH to override the Norvir patent, but the NIH announced on August 4, 2004 that it would not invoke its legal right to allow generic production of Norvir, citing potential adverse effects on the pharmaceutical market.
Method of action
Ritanovir is exceptional as the only antiretroviral drug that inhibits a liver enzyme that normally metabolizes away protease inhibitors, Cytochrome P450-3A4 (CYP3A4). The drug's molecular structure inhibits CYP3A4, so a low dose can be used to enhance any other protease inhibitors. This effect does come with a price: it also affects the strength of numerous other medications, making it difficult to know how to administer them concurrently. In addition it can cause a large number of side-effects on its own. It is now rarely used for its own antiviral activity but remains widely used as a booster to other protease inhibitors.
Read more at Wikipedia.org