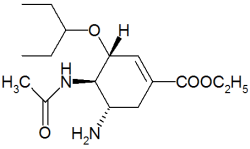
For marketers of seasonal products in a category as competitive and unpredictable as cold and flu remedies, it's crucial to be first. Just ask the folks at Hoffman-La Roche Pharmaceuticals, Nutley N.J, manufacturers of the prescription anti-viral flu medication Tamiflu.
At the start of last year's flu season, Tamiflu's chief competitor, Glaxo-Weilcome's Relenza, had received approval from the Food & Drug Administration and was building consumer awareness via a TV campaign featuring Wayne Knight, a.k.a. Newman from Seinfeld. While Tamiflu is a pill and Relenza is an inhaler, both had been approved to ease influenza symptoms if taken within the first 48 hours.
To make up for lost time, Roche teamed with Momentum Worldwide, New York, in a promotional tour to bolster its own ad campaign. Tour organizers hit the streets in heavy flu markets, armed not only with informational brochures but a plan to give passersby a live demonstration of flu prevention.
Well, sort of. Call it a day-in-the-life of a healthy person. An actor on display in a 9-by 20-foot, glass-enclosed, fully-furnished apartment mounted on the back of a flat-bed truck. The subject rose from bed in his pajamas, brushed his teeth, read the newspaper and ate breakfast. Later he watched TV, snacked, played videogames and worked on his hobbies, never acknowledging the hordes of people staring inward.
Besides attracting attention, the demonstration conveyed the message that while this man living in an enclosed, germ-free environment was therefore safe from the flu bug, the rest of us were at risk of getting sick. "We were looking for ideas that would resonate with people and [contrast with] how miserable they feel when they're sick," said Dan Stevenson, creative director at Momentum.
To ensure that even onlookers peering out from building windows had a clear view of the demonstration, Momentum hired Broadway set designer David Gallow to create the model apartment. Signage on top of the studio read, "One person in this town who can probably feel safe from the flu ... For the rest of us flu sufferers, there's Tamiflu."
Rather than the usual perky young volunteers, kindly grandma types stood outside distributing the Tamiflu brochures and specially packaged envelopes of "Granny's" chicken soup, complete with a picture of a white-haired Granny on the packet.
The tour ran from Jan. 1 through March 15, weaving its way into the country's 71 heaviest flu markets using eight mobile apartments. Meanwhile, in-store collateral buttressed the effort with an interactive moveable "symptom wheel" display to address whether or not one was suffering from the flu or just a cold.
With last year's severe flu outbreaks making national news, the Tamiflu tour accounted for more than 100 million brand impressions. In a fortuitous twist for Roche, Glaxo skipped a direct-to-consumer Relenza campaign this year when those Newman spots came under scrutiny by the FDA. Glaxo had altered the spots in 1999 when officials deemed the campaign "misleading" in its suggested effectiveness, and the company was forced to revise its labeling to reflect reports of serious respiratory problems in people using the inhaler.
From an unknown, late-market entry just three months prior to the tour, Tamiflu went on to dominate the anti-viral flu category with a 58% market share. The drug manufacturer rang up $41 million in U.S. sales from Nov. 1999 through April 2000, versus Relenza at $20 million, per IMS Health. Marketers are considering whether to export the Tamiflu promotion to cities outside the U.S.
CATEGORY: National Consumer Promotion (Between $3,000,000 and $6,000,000 )
PROGRAM: Launch of Tamiflu
MARKETER: Hoffman-La Roche Pharmaceuticals, Nutley, N.J.
AGENCY: Momentum Worldwide, New York
KEY PLAYERS:
Roche:
Momentum:
Nancy Serkow, consumer products director
Sean O'Malley, ass't creative director
Dan Stevenson, creative director
COPYRIGHT 2001 BPI Communications, Inc.
COPYRIGHT 2001 Gale Group