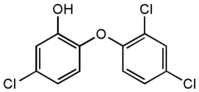
Triclosan
Triclosan (chemically 5-chloro-2-(2,4-dichlorophenoxy)phenol) is a potent wide-spectrum antibacterial and antifungal agent. more...
Chemistry
This organic compound is a white powdered solid with a slight aromatic/phenolic odor. It is a chlorinated aromatic compound which has functional groups representative of both ethers and phenols. Phenols often show anti-bacterial properties. Triclosan is slightly soluble in water, but soluble in ethanol, diethyl ether, and stronger basic solutions such as 1 M sodium hydroxide, like many other phenols.
Uses
It is found in soaps, deodorants, toothpastes and is impregnated in an increasing number of consumer products, such as kitchen utensils, toys, bedding, socks, and trash bags. It has been shown to be effective in reducing and controlling bacterial contamination on the hands and on treated products.
Triclosan is regulated by both the U.S. Food and Drug Administration and by the European Union. In the environment, triclosan is removed during normal waste treatment processes as shown by extensive environmental studies, and any of it that remains after waste treatment quickly breaks down into other compounds in the environment. However, one study showed that triclosan was broken down into dioxins in river water, because of the presence of sunlight (PMID 15779749).
Mechanism of action
Triclosan appears to kill bacteria mainly by inhibiting fatty acid synthesis. Triclosan binds to bacterial enoyl-acyl carrier protein reductase (encoded by FabI) and increases the enzyme's affinity for nicotinamide adenine dinucleotide (NAD+). This results in the formation of a stable ternary complex of FabI-NAD+-triclosan, which is unable to participate in fatty acid synthesis. Some bacterial strains are resistant to triclosan due to FabI mutations which decrease triclosan's effect on FabI-NAD+ binding. (PMID 10196195) Another way for bacteria to gain resistance to triclosan is to overexpress FabI (PMID 11418506).
Resistance concerns
An article coauthored by Dr. Stuart Levy in the August 6, 1998 issue of Nature (PMID 9707111) warned that its overuse could cause resistant strains of bacteria to develop, in much the same way that antibiotic-resistant bacterial strains are emerging, based on speculation that triclosan behaved like an antibiotic. Based on this speculation, in 2003, the Sunday Herald newspaper reported that some UK supermarkets and other retailers were considering phasing out products containing triclosan.
It has since been shown that the laboratory method used by Dr. Levy was not effective in predicting bacterial resistance for biocides like triclosan, based on work by Dr. Peter Gilbert in the UK (PMID 12957932). At least seven peer-reviewed and published studies have been conducted demonstrating that triclosan is not significantly associated with bacterial resistance, including one study coauthored by Dr. Levy, published in August of 2004 in Antimicrobial Agents and Chemotherapy (PMID 15273108). In addition, there is 30 years of experience with triclosan without any incidences of acquired bacterial resistance reported, and there are no studies showing acquired resistance after long-term use.
Read more at Wikipedia.org