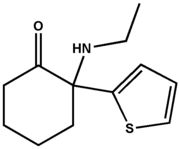
ABSTRACT. Objectives: Severe lactulose malabsorption causes osmotic diarrhea and decreased cecal cell proliferation. We tested the hypothesis that prefeeding with inulin, a prebiotic, would attenuate these effects. Methods: Piglets aged 10 days were randomized to 3 feeding groups (n = 6 each group): Control (CON), fed sow-milk replacement formula (SMR; lactose, 60 g/L) for 14 days; a lactulose-challenged group (LAC) that was fed SMR for 7 days and then a formula containing lactose (30 g/L) and lactulose (60 g/L) for 7 days; and a group prefed SMR containing inulin (3 g/L) for 7 days and then fed the lactulose-supplemented formula (INULIN). Groups CON and INULIN were pair-fed to LAC. Then, cecal tissue was collected for histology, determination of crypt cell proliferation index, apoptosis, and Western blot determination of expression of Bax, a pro-apoptotic protein. Results: The fraction of days when diarrhea was present (mean ± SD) was greater for LAG (0.87 ± 0.14; p = .004) than CON (0.28 ± 0.22; INULIN: 0.52 ± 0.44; p = .058 us LAC). Cell proliferation index for the total crypt was less for LAC (0.12 ± 0.04; p = .016) compared with CON (0.20 ± 0.04; INULIN: 0.15 ± 0.04; p = .06 vs LAC). BAX protein expression and apoptosis were similar in the 3 groups. Conclusions: We observed trends consistent with the hypothesis that prefeeding inulin attenuates diarrhea and the reduction in cell proliferation caused bylactulose. ( Journal of Parenteral and Enterai Nutrition 28:22-26, 2004)
Disaccharide malabsorption, secondary osmotic diarrhea, and even the short-chain fatty acid products of colonie fermentation may complicate recovery from enteric infection, hinder adaptation to enterai feeding after intestinal resection, and impair tolerance of liquid formulas in sick patients.1-8 We have been interested in the relative clinical merits and disadvantages of persistent enterai feeding, especially carbohydrate, in patients with apparent osmotic diarrhea from carbohydrate malabsorption. In an experimental model in piglets, severe disaccharide malabsorption was produced by feeding lactulose, a nondigestible disaccharide of galactose and fructose; this caused diarrhea, colitis, and lowering of cecal pH and cecal cell proliferation index.9 There is evidence that diversion colitis can be successfully treated with enemas containing butyric acid10; moreover, butyric acid deprivation of the colonie mucosa causes lower cell proliferation.11-15 Thus, we speculated that in our previous study, osmotic diarrhea reduced fermentation and particularly butyric acid production, which could in turn cause colitis and decreased cell proliferation in the colonie mucosa.9
Prefeeding adult humans a low dose of lactulose augmented fermentation and attenuated diarrhea during a subsequent challenge with a high dose of lactulose.16 Presumably, "priming" the colonie bacterial flora with a dose of indigestible carbohydrate that can be completely fermented might augment the overall capacity for fermentation (a form of adaptation), in part by inducing bacterial glucosidases.16 However, it also has been argued that prolonged feeding of a fermentable carbohydrate can lead to changes in the composition of the bacterial flora and thus in the preponderance of various bacterial pathways,17 which could quantitatively affect the production of individual organic acids during fermentation and then, secondarily, motility, perhaps via changes in secretion of neu ropeptides such as Peptide YY.18-20 According to this observation, prefeeding another fermentable carbohydrate that does not cause osmotic diarrhea also might attenuate some of the adverse effects of severe lactulose malabsorption.9 Oligosaccharides of fructose (including inulin and lactulose) are fermented and also have so-called prebiotic effects: they cause selective changes in the gut flora, particularly an increase in the bifidobacteria in the colon.21,22 Inulin has an average of 35 fructosyl units and will have much less of an osmotic effect on the colon than lactulose. Thus, we hypothesized that prefeeding with inulin would attenuate some of the adverse effects of subsequent severe disaccharide (lactulose) malabsorption (such as diarrhea and lower cecal cell proliferation). The purpose of this paper was to assess this overall hypothesis.
MATERIALS AND METHODS
Animals, Feedings, and Design
Eighteen standard Yorkshire/Hampshire piglets were used. The Children's Hospital Animal Care and Use Committee approved the research protocol. On day 7 of life, the piglets were transported from the pig farm to the laboratory. The piglets then were fed a carbohydrate-free sow milk replacement formula (SMR; Ross Products Division of Abbott Laboratories, Inc, Columbus, OH) supplemented with lactose (60 g/L) to accustom them to artificial nipple feeding. On day 10 of life, the piglets were randomized to 3 formula study groups. (1) The Control group (CON) received SMR formula with 60 g lactose/L for 14 days. (2) An experimental group (LAC), was fed SMR formula with 60 g lactose/L for 7 days and then the SMR formula containing lactose (30 g/L) and lactulose (60 g/L) for 7 days, and (3) the experimental group, INULIN, was fed SMR formula containing 60 g lactose/L and inulin (3 g/L) for 7 days and then fed the same lactulose-supplemented formula as the LAC group. Groups CON and INULIN were pair-fed to LAC. These 3 dietary groups were established according to the following rationale. The usual SMR formula contains 60 g/L of lactose. In the LAC group, we wanted to provide lactulose at a dose similar to our previous study where we observed that this dose caused osmotic diarrhea and decreased cell proliferation in the colonie mucosa. We included half the usual amount of lactose in order to meet minimal needs for glucose as suggested by a previous, preliminary study in piglets in which piglets were able to gain weight normally while receiving this dose of lactose.23 The dose of inulin was based on a previously published study in piglets which suggested that a dose of fructooligosaccharide of 3 g/L SMR formula augmented colonie mucosal cell proliferation.24 The piglets were studied in groups of 3 littermates. During the study, body weight, formula intake, and stool characteristics were monitored. If diarrhea was observed at any time during a day, this was counted as 1 day of diarrhea. The number of days so enumerated was divided by 7 to obtain the fraction of total days of observation when diarrhea was observed.
At age 24 days, the piglets were anesthetized with xylazine (4mg/kg IM; Rompun; Bayer Corp, Shawnee Mission, KS) and a combination of tiletamine HCl and zolazepam HCl (6mg/kg IM; Telazol; Fort Dodge Laboratories, Inc, Fort Dodge, IA). BrdU was injected into an ear vein.9 Two hours later, after the piglets were reanesthetized with the Telazol/Rompun combination, breath was collected from the endotracheal tube for determination of H2 CH4, and CO2 concentrations (Quintron Instrument Corp, Milwaukee, WI), and the concentrations of H2 and CH4 were expressed as ppm/% CO2. Cecal pH then was measured (Model Omega PHH-253; Omega Technologies Co, Stamford, CT). just before sacrifice, cecal tissue was obtained for histology and determination of cell proliferation, apoptosis, and Bax protein expression.
Histologic Damage Assessment
The tissues for light microscopy were fixed within a few seconds in 10% neutral buffered formalin, embedded in paraffin, and then sectioned at 5 µm. The slides were stained with hematoxylin-eosin for histologic assessment of injury or inflammation using a previously published scoring system25: Grade O, no abnormalities; 1, crypt dilatation and mucin retention; 2, cryptitis or 2 crypt abscesses; 3, multiple crypt abscesses; 4, fibrinopurulent debris and mucosal ulceration; 5, transluminal necrosis or perforation.
Cell Proliferation Index
Unstained, paraffin-embedded slides were stained for bromodeoxyuridine (BrdU) labeling (Cell Proliferation Kit from Amersham Life Science, no. RPN 20). Because cell proliferation in the normal colonie mucosa is much greater at the base of the crypt compared with the luminal surface,26'27 we estimated fractional proliferation index for the total cecal crypt and for the lower 40% of the crypt because, at least in the rat colon, butyrate significantly stimulates cell proliferation in the lower two-fifths of the crypt.27
Assessment of Apoptosis
DNA fragmentation (apoptosis) was assessed using the terminal deoxynucleotidyl transferase-mediated dUTPbiotin nick end labeling (TUNEL) technique (Tdt-FragEL DNA Fragmentation Detection Kit; catalog no. QIA33; Oncogene Research Products, Cambridge, MA).28
Bax Protein Expression
Expression of Bax, a pro-apoptotic protein, was determined by Western blot methodology.29 Previously, we speculated that osmotic diarrhea might inhibit fermentation and production of butyric acid per se.9 Thus, we studied Bax expression in the cecum specifically because there is evidence that depletion of butyric acid will stimulate Bax protein expression in colon.29 Briefly, after homogenization of the tissue sample, addition of proteinase inhibitors (to reduce degradation), and centrifugation, the protein concentration of the supernatant from the cell lysate was determined using the Pierce BCA (bicinchoninic acid) protein assay kit (Pierce Biotechnology, Rockford, IL). SDS-polyacrylamide gel electrophoresis and Western blotting were conducted using NuPage 4% to 12% Bis-Tris precast gel with MES Running Buffer, and the Xcell II Mini-Cell (EI9001) from Novex (Invitrogen Life Technologies, Carlsbad, CA). After blocking of nonspecific binding and washing, the membrane was incubated with the primary antibodies (Santa Cruz polyclonal anti-Bax, sc-493, prepared from rabbit; Santa Cruz Biotechnology, Santa Cruz, CA). After removal of free primary antibodies, the membrane was incubated with horseradish peroxidase-conjugated secondary antibodies (goat antirabbit). After removal of free secondary antibodies, the membrane was incubated with Luminol ECL Plus Reagent (Amersham Biosciences, Piscataway, NJ) to produce chemiluminescence. The image was developed with a phosphorimager (Molecular Dynamics Storm 860 at Blue Fluorescence/Chemifluoresence mode; Amersham Biosciences). The concentrations of the Bax protein were quantified by the Storm ImageQuant software.30
Data Analysis and Statistics
During histologic examination and other data analyses, the investigators were masked to the identity of the treatment group for each piglet. For assessing differences between groups, we used analysis of variance (ANOVA). When a significant group effect was identified by ANOVA, we used the least square differences test to compare means between specific groups. all results are expressed as mean ± SD (and not as SEM). Although we acknowledge that statistical significance is generally defined by a p value
RESULTS
In the LAC and INULIN groups, lactulose intake averaged, respectively, 15.4 g/kg/day (4.4 g/hour) and 13.8 g/kg/day. There was no effect of group on weight gain/formula intake (g/mL; CON, 0.20 ± 0.04; LAC, 0.21 ± 0.04; INULIN 0.22 ± 0.03).
Breath H^sub 2^ concentration (ppm/% CO2) ranged from 0.21 to 18.3 in the 18 piglets, and mean values were not significantly different in the 3 groups (CON, 0.4 ± 0.4; LAC, 2.5 ± 5.0; INULIN, 4.5 ± 7.1; Table I). The range in breath CH^sub 4^ (ppm/% CO2) was O to 15.5; the value (mean ± SD) in the INULIN group (6.4 ± 4.7) was significantly higher than in the LAC group (2.1 ± 2.2; p = .044) but not significantly different from CON (4.8 ± 2.8; Table I).
Cecal pH was lower in LAC (5.89 ± 0.66) than CON (6.71 ± 0.15; p = .014), but not significantly different from INULIN (6.36 ± 0.40; p = .13; Table I).
The fraction of days when diarrhea was present (mean ± SD) was greater for LAC (0.87 ± 0.14; p = .004) than for CON (0.28 ± 0.22; p = .004) or for INULIN (0.52 ± 0.44; p = .058 vs LAC; Table II).
The mean score for colonie injury in LAC was identical to CON (both, 1.5 ± 1.22) and to that seen in our previous study (Table II). In the INULIN group, the mean score was less than in the other 2 groups (0.83 ± 1.33), but this difference was not statistically different (Table II).
Cell Proliferation
Figure 1 depicts typical cecal crypts from each of the 3 groups; the BrdU labeling is noted by the very dense staining of the nuclei. Figure 1C illustrates the diminished frequency of BrdU-labeled cells in the lactulosetreated group, indicating a decreased labeling index (cell proliferation), compared with either the CON (Fig. IA) or INULIN groups (Fig. IB). Labeling index for the total crypt was 41% decreased in the LAC group (0.12 ± 0.04) compared with CON (0.20 ± 0.04; p = .0.005); there also was a significant (p = .007) 34% decrease in labeling index according to the lower 40% of the crypt (Table II). Labeling Index for the total crypt was 25% increased for the INULIN group (0.15 ± 0.04) compared with the LAC group (NS); labeling index results based on the lower 40% of the crypt indicated a 33% increase in the INULIN group compared with LAC (p = .057; Table II). For the total crypt analysis, there also was a borderline difference in labeling index between CON and INULIN (p = .06; Table II).
Apoptosis and Bax Protein Expression
We evaluated apoptosis in cecal sections from each piglet. In all but 2 piglets, there was no evidence of apoptosis in at least 20 crypts examined. In 1 piglet in CON, 6 of 10 crypts had > or =1 apoptotic cells identified according to the stain and the presence of morphologic criteria (range in these 6 crypts was 1 to 8 apoptotic cells per crypt). In 1 piglet in INULIN, 1 crypt of 10 showed 1 apoptotic cell. In another piglet in INULIN, 6 of 10 crypts had > or =1 apoptotic cell (range 1 to 5).
Bax protein expression was similar in the 3 groups (Table II).
DISCUSSION
Lactulose is a disaccharide of fructose and galactose that cannot be digested by mammalian enzymes. Experimentally, it permits one to simulate the effects of sugar maldigestion, both in the colon where it likely will be fermented in a very similar way to lactose31'32 and in the small intestine where fermentation also may occur (theoretically) and where undigested sugar also could exert an osmotic effect and could increase water flux through the bowel. As in our previous study,9 severe disaccharide malabsorption (produced by feeding lactulose) caused diarrhea and lowering of cecal pH and cecal cell proliferation index. Although the injury index was also the same in the LAC group compared with the similar group in our previous study, mean injury score was identical in the LAC and CON groups. We have no explanation for this latter observation, except for variance in the health of the piglets we were able to study; however, the degree of colonie injury in this control group also was greater than in a third study.23
This study also suggested that prefeeding with inulin tended to attenuate diarrhea, mucosal damage, and the reduction in cell proliferation and cecal pH produced by lactulose feeding. This conclusion is predicated on the acceptance of a probability value of .06 as reflecting statistical significance.
From our data, it appears that inulin improved colonie function, perhaps by enhancing fermentation. In this regard, short-chain fatty acids produced from fermentation stimulate water and sodium absorption in the colon,33-35 and especially butyrate is needed to maintain normal colonie structure and cell turnover.10'36 We used measurements of breath concentrations of H^sub 2^ and CH^sub 4^ to assess bacterial fermentation in the colon. There were no statistically significant differences among the groups in breath H^sub 2^ concentration, although the mean value was highest in the INULIN group. However, the breath concentration of CH^sub 4^ was also higher in the INULIN group compared with LAG (p = .044), suggesting that fermentation was stimulated in the INULIN group. Interestingly, in a previous study,23 we observed a statistically significant increase in breath CH^sub 4^ concentration in piglets fed a smaller supplement of lactulose, which did not cause diarrhea and tended to increase, not decrease, cell proliferation. Perhaps small doses of lactulose and other fructose oligosaccharides (like inulin) stimulate colonie fermentation, as has been shown in humans.16
It is intriguing to speculate about whether different effects of inulin might be observed if it were fed during lactulose feeding and prefed before lactulose challenge. In this study, we prefed inulin instead of administering it also during lactulose feeding for 2 reasons. First, we were interested in using a previously published experimental design16 and wanted to determine if a fermentable sugar that did not cause osmotic diarrhea would attenuate adverse effects of lactulose feeding, presumably by stimulating overall bacterial fermentation capacity (ie, feeding inulin would result in increased fermentable substrate without the possible decreased bacterial mass and fermentation activity caused by osmotic diarrhea).9 sec ond, despite its so-called prebiotic effects on bifidobacteria colonization, there is uncertainty regarding the effects of inulin on fermentation pathways and particularly on luminal synthesis of butyric acid.37'38 There is evidence that butyric acid could affect colonie mucosal function and cell proliferation, either as a "beneficial" luminal nutrient10'36 or as a toxic compound to gut mucosa.39'40 Therefore, we did not want to provide this additional substrate for fermentation during the 7-day challenge with lactulose, because in this study, we could not monitor luminal concentration or production of butyric acid.
In conclusion, this study provides the first preliminary evidence that prefeeding a prebiotic may prevent subsequent diarrhea and decreased cell proliferation associated with disaccharide-induced osmotic diarrhea. However, because the p value for each of these observed trends was only .06, there is a need to validate these results with a larger study or perhaps one with a different design. For example, it is plausible that the effects of inulin could be more pronounced if it were fed both before and during the lactulose feeding, as noted above.
ACKNOWLEDGMENTS
The study was supported in part by grants from Crohn's and Colitis Foundation of America and the Children's Research Institute, and the piglet formula was provided by Ross Products Division of Abbott Laboratories, Inc. Dr Rien also was supported by National Institutes of Health grant ROl DK61775 while the data were being analyzed and the manuscript was being written. The authors acknowledge the technical assistance of Jonathan Lash, with the animal procedures. The authors also are grateful to Florinda Jaynes and her staff for assistance with tissue preparation for microscopy and to Mark Luquette, MD, Department of Pathology, for his advice and assistance in selecting the optimal counterstain to quantify the index of apoptosis.
REFERENCES
1. Bhutta ZA, Hendricks KM. Nutritional management of persistent diarrhea in childhood: a perspective from the developing world. J Pediatr Gastroenterol Nutr. 1996;22:17-37.
2. Penny ME, Paredes P, Brown KH. Clinical and nutritional consequences of lactose feeding during persistent postenteritis diarrhea. Pediatrics. 1989;84:835-844.
3. Hammer HF, Fine KD, SantaAna CA, Porter JL, Schiller LR, Fordtran JS. Carbohydrate malabsorption: its measurement and its contribution to diarrhea. J Clin Invest. 1990;86:1936-1944.
4. Vanderhoof JA, Langnas AN, Pinch LW, Thompson JS, Kaufman SS. Invited review: short bowel syndrome. J Pediatr Gastroenterol Nutr. 1992;14:359-370.
5. Piche T, Zerbib F, Varannes SB, et al. Modulation by colonie fermentation of LES function in humans. Am J Physiol Gastrointest Liver Physiol. 2000;278:G578-G584.
6. Cuche G, Cuber JC, Malbert CH. Heal short-chain fatty acids inhibit gastric motility by a humoral pathway. Am J Physiol Gastrointest Liver Physiol. 2000;279:G925-G930.
7. Tohno H, Sarr MG, DiMagno EP. Intraileal carbohydrate regulates canine postprandial pancreaticobiliary secretion and upper gut motility. Gastroenterology: 1995;109:1977-1985.
8. Cherbut C, Ferrier L, Roze C, et al. Short-chain fatty acids modify colonie motility through nerves and polypeptide YY release in the rat. Am J Physiol. 1998;275:G1415-G1422.
9. Kien CL, Murray RD, Qualman SJ, Marcon M. Lactulose feeding in piglets: a model for persistent diarrhea and colitis induced by severe sugar malabsorption. Dig Dis Sd. 1999;44:1476-1484.
10. Harig JM, Soergel KH, Komorowski RA, Wood CM. Treatment of diversion colitis with short-chain-fatty acid irrigation. N Engl J Med. 1989;320:23-28.
11. Sakata T. Depression of intestinal epithelial cell production rate by hindgut bypass in rats. Scared J Gastroenterol. 1988;23:1200-1202.
12. Bartram HP, Scheppach W, Schmid H, et al. Proliferation of human colonie mucosa as an intermediate biomarker of carcinogenesis: effects of butyrate, deoxycholate, calcium, ammonia, and pH. Cancer Res. 1993;53:3283-3288.
13. Sakata T. Stimulatory effect of short-chain fatty acids on epithelial cell proliferation in the rat intestine: a possible explanation for trophic effects of fermentable fibre, gut microbes and luminal tropic factors. Br J Nutr. 1987;58:95-103.
14. Goodlad RA, Wright NA. Effects of addition of kaolin or cellulose to an elemental diet on intestinal cell proliferation in the mouse. Br J Nutr. 1983;50:91-98.
15. Komai M, Takehisa F, Kimura S. Effect of dietary fiber on intestinal epithelial cell kinetics of germ-free and conventional mice. Nutr Rep Int. 1982;26:255-261.
16. Flourie B, Briet F, Florent C, Pellier P, Maurel M, Rambaud JC. Can diarrhea induced by lactulose be reduced by prolonged ingestion of lactulose? Am J Clin Nutr. 1993;58:369-375.
17. Florent C, Flourie B, Leblond A, Rautureau M, Bernier J-J, Rambaud J-C. Influence of chronic lactulose ingestion on the colonie metabolism of lactulose in man (an in vivo study). J Clin Invest. 1985;75:608-613.
18. Layer P, Peschel S, Schlesinger T, Goebell H. Human pancreatic secretion and intestinal motility: effects of ileal nutrient perfusion. AmJPhysiol Gastroiiitest Liver Physiol. 1990;258:G196-G201.
19. Wen J, Phillips SF, Sarr MG, Kost LJ, Holst JJ. PYY and GLP-I contribute to feedback inhibition from the canine ileum and colon. Am J Physiol Gastroiiitest Liver Physiol. 1995;269:G945-G952.
20. Longo WE, Ballantyne GH, Savoca PE, Adrian TE, Bilchick AJ, Modlin IM. Short-chain fatty acid release of peptide YY in the isolated rabbit distal colon. Scand J Gastroenterol. 1991;26:442-448.
21. Gibson GR, Roberfroid MB. Dietary modulation of the human colonie microbiota: introducing the concept of prebiotics. JNutr. 1995;125:1401-1412.
22. Roberfroid MB, Van Loo JA, Gibson GR. The bifidogenic nature of chicory inulin and its hydrolysis products. J Nutr. 1998;128: 11-19.
23. Kien CL, Cooper JR, Frankel WL. Moderate disaccharide malabsorption does not affect weight gain and cecal cell proliferation in piglets. JPEN. 2003;27:323-326.
24. Howard MD, Gordon DT, Pace LW, Garleg KA, Kerley MS. Effects of dietary supplementation with fructooligosaccharides on colonie microbiota populations and epithelial cell proliferation in neonatal pigs. JPediatr Gastroenterol Nutr. 1995;21:297-303.
25. Qualman S, Pysher T, Schauer G. Hirschsprung disease: differential diagnosis and sequelae. In: Dahms WT, Qualman SJ, eds. Perspectives in Pediatrie Pathology. Basel: Karger; 1997;111-126.
26. Lipkin M. Proliferation and differentiation of normal and diseased gastrointestinal cells. In: Johnson LR, ed. Physiology of the Gastrointestinal Tract. New York: Raven Press; 1985;255-284.
27. Velazquez OC, Zhou D, Seto RW, et al. In vivo crypt surface hyperproliferation is decreased by butyrate and increased by deoxycholate in normal rat colon: associated in vivo effects on c-Fos and c-Jun expression. JPEN. 1996;20:243-250.
28. McKay BC, Ljungman M, Rainbow AJ. Persistent DNA damage induced by ultraviolet light inhibits p21wafl and bax expression: implications for DNA repair, UV sensitivity and the induction of apoptosis. Oncogene. 1998;17:545-555.
29. Hass R, Busche R, Luciano L, Reale E, Engelhardt WV. Lack of butyrate is associated with induction of Bax and subsequent apoptosis in the proximal colon of guinea pig. Gastroenterology. 1997;112:875-881.
30. Qu X, Yang Z, Zhang S, et al. The human DEVH-bax protein Ski2w from the HLA is localized in nucleoli and ribosomes. Nucleic Acids Res. 1998;26:4068-4077.
31. Mortensen PB. Holtug K, Rasmussen HS. Short-chain fatty acid production from mono- and disaccharides in a fecal incubation system: implications for colonie fermentation of dietary fiber in humans. J Nutr. 1988;118:321-325.
32. Mortensen PB, Rasmussen HS, Holtug K. Lactulose detoxifies in vitro short-chain fatty acid production in colonie contents induced by blood: implications for hepatic coma. Gastroenterology. 1988;94:750-754.
33. Argenzio RA, Miller N, VonEngelhardt W. Effect of volatile fatty acids on water and ion absorption from the goat colon. Am J Physiol. 1975;229:997-1002.
34. Demigne C, Remesy C. Stimulation of absorption of volatile fatty acids and minerals in the cecum of rats adapted to a very high fiber diet. J Nutr. 1985;115:53-60.
35. Roediger WE, Rae DA. Trophic effect of short chain fatty acids on mucosal handling of ions by the defunctioned colon. Br J Surg. 1982;69:23-25.
36. Roediger WEW. Utilization of nutrients by isolated epithelial cells of the rat colon. Gastroenterology. 1982;83:424-429.
37. Djouzi Z, Andrieux C. Compared effects of three oligosaccharides on metabolism of intestinal microflora in rats inoculated with a human faecal flora. Br J Nutr. 1997;78:313-324.
38. Kleessen B, Sykura B, Zunft H-J, Blaut M. Effects of inulin and lactose on fecal microflora, microbial activity, and bowel habit in elderly constipated persons. Am J Clin Nutr. 1997;65:1397-1402.
39. Argenzio RA, Meuten DJ. Short-chain fatty acids induce reversible injury of porcine colon. Dig Dis Sei. 1991;36:1459-1468.
40. Butel MJ, Roland N, Hibert A, et al. Clostridial pathogenicity in experimental necrotising enterocolitis in gnotobiotic quails and protective role of bifidobacteria. J Med Microbiol. 1998;47:391-399.
C. Lawrence Kien, MD, PhD*[dagger][double dagger]J. C. Chang, PhD*; James R. Cooper, DVM, PhD*; and Wendy L. Frankel, MD§
From the * Children's Research Institute, the [dagger] Department of Pediatrics, § the Department of Pathology, The Ohio State University, Columbus, OH; and the [double dagger] Department of Pediatrics, University of Texas Medical Branch, Galveston, Texas
Received for publication December 28, 2002.
Accepted for publication September 15, 2003.
Correspondence: C. Lawrence Kien, MD, PhD, Department of Pediatrics, Gastroenterology Division, University of Texas Medical Branch, Children's Hospital, 301 University Blvd., Galveston, TX 77555-0352. Electronic mail may be sent to clkien@utmb.edu.
Copyright American Society for Parenteral and Enteral Nutrition Jan/Feb 2004
Provided by ProQuest Information and Learning Company. All rights Reserved