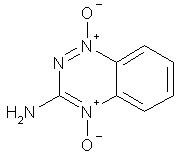
Photochemical Electron Transfer Reactions of Tirapazamine(para)
ABSTRACT
The absorption and fluorescence spectra of 3-aminobenzo-1,2,4-triazine di-N-oxide (tirapazamine) have been recorded and exhibit a dependence on solvent that correlates with the Dimroth E^sub T30^ parameter. Time-dependent density functional theory calculations reveal that the transition of tirapazamine in the visible region is pi > pi* in nature. The fluorescence lifetime is 98 +/- 2 ps in water. The fluorescence quantum yield is ~0.002 in water. The fluorescence of tirapazamine is efficiently quenched by electron donors via an electron-transfer process. Linear Stern-Volmer fluorescence quenching plots are observed with sodium azide, potassium thiocyanate, guanosine monophosphate and tryptophan (Trp) methyl ester hydrochloride. Guanosine monophosphate, tyrosine (Tyr) methyl ester hydrochloride and Trp methyl ester hydrochloride appear to quench the fluorescence at a rate greater than diffusion control implying that these substrates complex with tirapazamine in its ground state. This complexation was detected by absorption spectroscopy.
Abbreviations: AMP. adenosine 5' monophosphate; DABCO, 1,4diazabicyclo[2.2.21octane; GMP, guanosine monophosphate; 2MeTHF, 2-methyltetrahydrofuran; TCSPC, time-correlated single photon counting; TD-DFT, time-dependent density functional theory; tirapazamine, 3-aminobenzo-1,2,4-triazine di-N-oxide; Trp, tryptophan: Tyr, tyrosine.
INTRODUCTION
Since the late 1980s, a considerable amount of attention has been given to 3-aminobenzo-1,2,4-triazine di-N-oxide (tirapazamine; [11) as a potential antitumor agent, either alone or as part of combination therapies (1). Tirapazamine has been found to be capable of acting selectively in hypoxic environments found in rapidly growing tumors (2,3). One of the proposed mechanisms of action of this compound is shown in Scheme 1: enzymatic electron transfer and subsequent proton transfer to generate the radical 3, which undergoes beta-fission (4) to generate 3-aminobenzo- 1,2,4-triazine-N-oxide (4) and hydroxyl radical, which is well established as a promoter of nucleic acid cleavage (5). The fact that this reaction sequence may be quenched in the presence of oxygen gives rise to the potential in vivo selectivity of this agent.
The enzymatically triggered series of reactions can, in principle, be mimicked by light-induced electron-transfer reactions in aqueous solution. The present study was undertaken to investigate that possibility.
Acknowledgements-Support of this work by the NSF-Environmental Molecular Science Institute at The Ohio State University (NSF CHE-0089147 to M.S.P. and C.M.H.) and the American Cancer Society (RPG 00-028-01 to K.S.G.) is gratefully acknowledged, as are the computational resources made available from the Ohio Supercomputer Center. The authors would also like to thank Terry Gustafson and Frank DeLucia Jr. at OSU for performing the TCSPC experiments.
(para)Posted on the web site on January 28, 2002.
REFERENCES
1. Brown, J. M. (1999) The hypoxic cell: a target for selective cancer therapy-eighteenth Bruce F. Cain memorial award lecture. Cancer Res. 59, 5863-5870.
2. Costa, A. K., M. A. Baker, J. M. Brown and J. R. Trudell (1989) In vitro hepatotoxicity of SR 4233 (3-amino-1,2,4-benzotriazine-1,4-dioxide), a hypoxic cytotoxin and potential antitumor agent. Cancer Res. 49, 925-929.
3. Brown, J. M. (1993) SR 4233(tirapazamine): a new anticancer drug exploiting hypoxia in solid tumors. Br. J. Cancer 67, 1163-1170.
4. Daniels, J. S, and K. S. Gates (1996) DNA cleavage by the antitumor agent 3-amino-1,2,4-benzotriazine 1,4-dioxide (SR4233): evidence for involvement of hydroxyl radical. J Am. Chem. Soc. 118, 3380-3385.
5. Von Sonntag, C., U. Hagen, A. Schoen-Bopp and D. Schulte-- Frohlinde (1981) Radiation-induced strand breaks in DNA: chemical and enzymic analysis of end groups and mechanistic aspects. Adv. Radiat. Biol. 9, 109-142.
6. Fuchs, T., G. Chowdhury. C. L. Barnes and K. S. Gates (2001) 3-Amino-1,2,4-ben zotri azine 4-oxide: characterization of a new metabolite arising from bioreductive processing of the antitumor agent 3-amino-1,2,4-benzotriazine 1,4-dioxide (tirapazamine). J. Org. Chem. 66, 107-114.
7. Buterbaugh, J. S., J. P. Toscano, W. L. Weaver, J. R. Gord, C. M. Hadad, T. L. Gustafson and M. S. Platz (1997) Fluorescence lifetime measurements and spectral analysis of adamantyldiazirine. J. Am. Chem. Soc. 119, 3580-3591.
8. Bauernschmitt, R. and R. Ahlrichs (1996) Treatment of electronic excitations within the adiabatic approximation of time dependent density functional theory. Chem. Phys. Lett. 256, 454-464.
9. Stratmann, E. R., G. E. Scuseria and M. J. Frisch (1998) An efficient implementation of time-dependent density-functional theory for the calculation of excitation energies of large molecules. J. Chem. Phys. 109, 8218--8224.
10. TD-DFT has been used successfully to simulate electronic spectra (vertical transitions) of organic molecules. For example, see Wiberg, K. B., R. E. Stratmann and M. J. Frisch (1998) A timedependent density functional theory study of the electronically excited states of formaldehyde. acetaldehyde and acetone. Chem. Ph-vs. Lett. 297. 60-64.
11. van Gisbergen, S. J. A, A. Rosa, G. Ricciardi and E. J. Baerends (1999) Time-dependent density functional calculations on the electronic absorption spectrum of free base porphyrin. J. Chem. Phys. 111, 2499-2506.
12. Becke, A. D. (1988) Density-functional exchange-energy approximation with correct asymptotic behavior. Phys. Rev. A 38, 3098-3100.
13. Becke, A. D. (1993) Density-functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 98, 5648-5652. 14. Lee, C., W. Yang and R. G. Part (1988) Development of the
Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B 37, 785-789.
15. Frisch, M. J., G. W. Trucks, H. B. Schlegel, G, E. Scuseria, M. A. Robb, J. R. Cheeseman, V. G. Zakrzewski, J. A. Montgomery, Jr., R. E. Stratmann, J. C. Burant, S. Dapprich, J. M. Millam, A. D. Daniels, K. N. Kudin, M. C. Strain, 0. Farkas, J. Tomasi, V. Barone, M. Cossi, R. Cammi, B. Mennucci, C. Pomelli, C. Adamo, S. Clifford, J. Ochterski, G. A. Petersson, P. Y. Ayala, Q. Cui, K. Morokuma, D. K. Malick, A. D. Rabuck, K. Raghavachari, J. B. Foresman, J. Cioslowski, J. V. Ortiz, B. B. Stefanov, G. Liu, A. Liashenko, P. Piskorz, I. Komaromi, R. Gomperts, R. L. Martin, D. J. Fox, T. Keith, M. A. AI-Laham, C. Y. Peng, A. Nanayakkara, C. Gonzalez, M. Challacombe, P. M. W. Gill, B. Johnson, W. Chen, M. W. Wong, J. L. Andres, C. Gonzalez, M. Head-Gordon, E. S. Replogle and J. A. Pople (1998) Gaussian 98, Revision A.6. Gaussian, Inc. Pittsburgh, PA.
16. McDowell, J. A. and D. H. Turner (1996) Investigation of the structural basis of thermodynamic stabilities of tandem GU mismatches: solution structure of (rGAGGUCUC)2 by two-dimensional NMR and simulated annealing. Biochemistry 35, 1407714089.
17. Dimroth, K. and C. Reichardt (1969) Erweiterung der 16sungsmittelpolarititsskala dutch verwendung alkyl-substituierter pyridinium-N-phenol betaine. Liebigs Ann. Chem. 727, 93-99.
18. Reichardt, C, (1979) Solvent Effects in Organic Chemistry, pp. 225-262. Verlag Chemie, Weinheim.
19. Reichardt, C. (1979) Solvent Effects in Organic Chemistry, pp. 189-205. Verlag Chemie, Weinheim.
20. Bist, H. D., J. S. Parihar and J. C. D. Brand (1976) The 341 nm band system of pyridine-N-oxide. Analysis of the in-plane vibrational structure. J. Mol. Spectrosc. 59, 435-441.
21. Brand, J. C. D. and K. T. Tang (1971) Rotational analysis of the 342 nm band of pyridine-N-oxide. J. Mol. Spectrosc. 39, 171-174.
22. Demas, J. N. and S. Snyder (1989) Single Photon Counting Software.
23. Fery-Forgues, S. and D. Lavabre (1999) Are fluorescence quantum yields so tricky to measure? A demonstration using familiar stationery products. J. Cherry. Ed. 76, 1260-1264.
24. Birks, J. B. (1970) Photophysics of Aromatic Molecules, pp. 443-448. Wiley-Interscience, London.
25. Chen, T., E. Voelk, M. S. Platz and R. P. Goodrich (1996) Photochemical and photophysical studies of 3-amino-6-iodoacridine and the inactivation of X phage. Photochem. Photobiol. 64, 622-631.
26. Wilson, W. D., F. A. Tanious, M. Fernandez-Saiz and C. T. Rigl (1997) Evaluation of Drug-Nucleic Acid Interactions by Thermal Melting Curves, Drug-DNA Interaction Protocols Vol. 90, (Edited by K. R. Fox), pp. 219-239. Humana, Totowa, NJ.
27. Jenkins, T. (1997) Optical Absorbance and Fluorescence Techniques for Measuring DNA-Drug Interactions; Vol. 90 (Edited by K. R. Fox), pp. 195-218. Humana Press, Totawa, NJ.
28. Daniels, J. S., T. Chatterji. L. R. MacGillivray and K. S. Gates (1998) Photochemical DNA cleavage by the antitumor agent 3amino-1,2,4-benzotriazine 1,4-dioxide (tirapazamine, WIN 59075, SR4233). J. Org. Chem. 63, 10027-10030.
29. Olmstead, J. III (1979) Calorimetric determinations of absolute fluorescence quantum yields. J. Phys. Chem. 83, 2581-2584. 30. Martin, M. M. (1975) Hydrogen bond effects on radiationless
electronic transitions in xanthene dyes. Chem. Phys. Lett. 35, 105-111.
31. DeFelippis, M. R; C. P. Murthy, F. Broitman, D. Weinraub, M. Farraggi and M. H. Klapper (1991) Electrochemical properties of tyrosine phenoxy and tryptophan indolyl radicals in peptides and amino acid analogs. J. Phys. Chem. 95, 3416-3419.
32. Jovanovic, S. V; S. Steenken and M. G. Simic (1991) Kinetics and energetics of one-electron-transfer reactions involving tryptophan neutral and cation radicals. J. Phys. Chem. 95, 684-687.
33. Steenken, S. and S. V. Jovanovic (1997) How easily oxidizable is DNA? One-electron reduction potentials of adenosine and guanosine radicals in aqueous solution. J. Am. Chem. Soc. 119, 617-618.
34. Priyadarsini, K. L, M. Tracy and P. Wardman (1996) The one-- electron reduction potential of 3-amino-1,2,4-benzotriazine 1,4dioxide (tirapazamine): a hypoxia-selective bioreductive drug. Free Rad. Res. 25, 393-399.
James S. Poole1, Christopher M. Hadad1, Matthew S. Platz*1, Zachary P. Fredin1, Laura Pickard1, Elisa Levya Guerrero1, Margarita Kessler1, Goutam Chowdhury 2, Delshanee Kotandeniya2 and Kent S. Gates2
1Department of Chemistry, The Ohio State University, Columbus, OH and
2Department of Chemistry and Biochemistry, University of Missouri, Columbia, MO
Received 22 August 2001; accepted 17 January 2002
*To whom correspondence should be addressed at: Department of Chemistry, Ohio State University, 100W 18th Avenue. Columbus, OH 43210, USA. Fax: 614-292-5151: e-mail: platz.1@osu.edu
Copyright American Society of Photobiology Apr 2002
Provided by ProQuest Information and Learning Company. All rights Reserved