* Massive hepatic necrosis following exposure to phenytoin and trimethoprim-sulfamethoxazole is a rare occurrence and to the best of our knowledge has not been reported previously. Acute hepatic failure following administration of trimethoprim-sulfamethoxazole has rarely been seen, and only 4 cases have been well documented pathologically. We report a case of acute liver failure in a 60-year-old woman following ingestion of phenytoin and trimethoprim-sulfamethoxazole concomitantly over a 9-day period. Autopsy findings revealed acute fulminant hepatic failure. This case demonstrates the effects of chemical-chemical interactions in the potentiation of hepatotoxicity of single agents and specifically illustrates the need for discontinuing trimethoprim-sulfamethoxazole in the presence of early liver injury.
(Arch Pathol Lab Med. 2000;124:1800-1803)
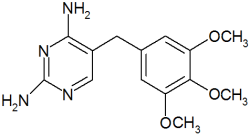
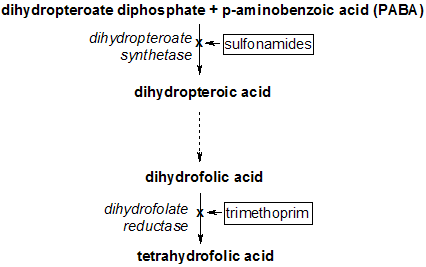
ydantoins and sulfonamides have been incriminated in hepatic damage either via idiosvncrasv or intrinsic toxicity. Phenytoin(DPH) is converted in the liver by parahydroxylation to 5-(p-hydroxyphenyl)-5-phenylhydantoin and then conjugated to glucuronic acid.' The hepatic injury varies from almost trivial to massive necrosis and is usually hepatocellular, although a mixed hepatocellular/cholestatic pattern is seen occasionally.2 Trimethoprim-sulfamethoxazole (TMP-SMZ) is also metabolized by glucuronide conjugation.3 The injury is usually cholestatic or mixed, although hepatocellular damage may occur, especially in immunocompromised patients.4,5 Trimethoprim-sulfamethoxazole, however, rarely causes acute fulminant liver failure with massive hepatocellular necrosis; only 4 cases have been well documented pathologically.69
We report a case of massive hepatic necrosis following exposure to DPH and TMP-SMZ and discuss the effects of chemical-chemical interactions in the potentiation of hepatotoxicity of single agents.
REPORT OF A CASE A 60-year-old black woman was transferred from another hospital because of a right-sided cerebral hemorrhage involving the region of the right basal ganglia and putamen. The patient had a history of untreated hypertension and diabetes of unknown duration. There was no history of tobacco use, alcohol consumption, intravenous drug abuse, or drug allergies.
The patient's temperature was 36.7C; pulse rate, 82 beats per minute; and respiration, 16 breaths per minute. Her blood pressure was 220/120 mm Hg. The patient complained of slurred speech and difficulty with movement on the left side. The neurological examination demonstrated small reactive pupils, leftsided facial palsy, increased left-sided reflexes, left hemiplegia, and a left Babinski sign. The rest of the physical examination was normal. Electrocardiogram showed normal sinus rhythm.
The white blood cell count, hemoglobin value, hematocrit level, and platelet count were all normal, as were the sodium, potassium, chloride, carbon dioxide, serum urea nitrogen, creatine, glucose, and liver enzyme studies.
The patient underwent inferior vena caval filter placement for deep vein thrombosis prophylaxis. There were no complications to the procedure. Treatment with DPH (100 mg 3 times daily) and cimetidine (400 mg 2 times daily) was begun. In addition, she was given heparin (500 units subcutaneously every 8 hours), docusate sodium (100 mg 3 times daily), and clonidine hydrochloride (0.1 mg every 6 hours). Eleven days after admission, the patient was transferred to rehabilitation. Her overall condition was good, her cognitive functions were improving, and she was alert and oriented. On day 13, the patient spiked a temperature of 39.5C. Urinalysis showed 15 to 30 white blood cells with 1+ protein. Blood culture was negative. At this time, the aspartate aminotransferase, alkaline phosphatase, and total bilirubin levels became elevated (Table 1). The patient was still taking DPH and cimetidine. She had no rash, lymphadenopathy, or hepatosplenomegaly. On day 14, the patient was treated with TMP (400 mg)-SMZ (800 mg) 2 times daily for urinary tract infection. On day 21, the patient suddenly developed a spiking fever of 38.3C. Her mental status changed, and she appeared somnolent and lethargic. It was believed that the patient may have suffered another intracerebral hemorrhage, and she was admitted to the Medicine Service 23 days after her initial admission.
The patient's temperature was 37.2C; pulse rate, 90 beats per minute; and respiration, 20 breaths per minute. Her blood pressure was 130/90 mm Hg. The patient was lethargic. Neurological examination showed left-sided motor weakness, a decrease in sensory responsiveness, and hyperreflexia. The rest of the physical examination was normal. The patient's alkaline phosphatase, aspartate aminotransferase, alanine aminotransferase, total bilirubin, and y-glutamyltransferase levels were markedly elevated at this time (Table 1).
The patient was treated for possible urosepsis with ceftriaxone (1 g intravenously every 8 hours). Additionally, as a precaution, TMP-SMZ, DPH, and cimetidine were discontinued, although the patient displayed no signs of an allergic reaction or eosinophilia. On day 24, the patient spiked a temperature of 41.2C. Computed tomographic scan of the brain revealed no changes. On day 26, the patient's liver enzyme levels dramatically increased, and the patient went into hypotensive shock. She was sent to the intensive care unit, where she was intubated. Tests for hepatitis B surface antigen and antibody, hepatitis C antibody, and human immunodeficiency virus were negative. Ceftriaxone was discontinued, and imipenem-cilastatin and vancomycin were administered. The patient developed renal and hepatic insufficiency and died 26 days after admission.
PATHOLOGIC FINDINGS A complete autopsy was done 24 hours after death. The most striking gross findings were submassive hepatic necrosis, duodenal ulcer with hemorrhage, and hemorrhage into the right basal ganglia. There was no evidence of pulmonary embolism or venous thrombosis. The liver weighed 2080 g. The capsule was shrunken and had focal petechiae. Cut sections revealed a yellow-brown parenchyma with confluent areas of necrosis (Figure 1). Microscopic sections showed massive hepatic necrosis with collapse and minimal fibrosis predominantly in Rappaport zones 2 and 3 (Figure 2). The portal triads contained a mild, chronic lymphoplasmacytic infiltrate with occasional eosinophils and bile duct proliferation (Figure 3). COMMENT There are 2 types of chemical-chemical interactions that may be involved in hepatotoxicity: (1) toxicokinetic, involving an alteration in the disposition of the chemical, and (2) toxicodynamic, involving alterations in the response of a tissue to the chemical or the injury it causes.10 Toxicokinetic interactions act primarily through enhancement or inactivation of enzyme systems that govern drug metabolism and involve the modification of factors that influence the disposition of the chemical (absorption, distribution, metabolism, and excretion). Toxicodynamic interactions act through induction or depletion of tissue factors and involve alterations in inflammatory response, tissue repair mechanisms, or the hemodynamics of the chemical. The present case represents a predominantly toxicokinetic chemical interaction with a toxicodynamic component.
The temporal sequence of clinical symptoms and concomitant elevations in hepatic enzymes strongly suggest that DPH may have initiated the hepatic damage, but that TMP-SMZ was responsible for the patient's fulminant hepatic failure. During her first 14 days in the hospital, the patient received DPH and did not manifest clinical signs indicative of DPH hypersensitivity. Phenytoin is known to elevate alkaline phosphatase levels and with severe toxicity exhibits any one of the findings of eosinophilia, rash, lymphadenopathy, and hepatic necrosis.11-17 Our patient did not display eosinophilia, rash, or lymphadenopathy.
Trimethoprim was administered from the 14th to the 23rd hospital day concomitantly with DPH. Although TMP-SMZ can decrease DPH metabolism and increase its toxicity when given concurrently,"' there was no indication of DPH hypersensitivity during these 8 days. We believe rather that depletion of glucuronic acid by DPH most likely increased the hepatotoxicity of TMP-SMZ, resulting in acute fulminant hepatic failure. In addition, depletion of glucuronic acid may have exhausted the detoxifying mechanisms of endogenous antioxidants and metals, potentiating the hepatotoxicity of TMP-SMZ. This patient did not have the clinical, gross, and microscopic findings usually associated with DPH fulminant liver failure.
Adverse side effects to TMP-SMZ are rare. They include anaphylaxis, cutaneous eruptions, thrombocytopenia, leukopenia, and hemolytic anemia.3 Rarely, TMPSMZ has caused acute liver failure. Four cases are well documented in the literature (Table 2).69 The exact mechanism of liver failure due to TMP-SMZ is unknown. The inferred mechanism of TMP-SMZ toxicity is consistent with hypersensitivity, and hepatic injury has been reprovoked by challenge doses of TMP-SMZ.19
Symptoms of TMP-SMZ toxicity usually develop within 10 days of ingestion.6,7 In some patients who have had previous exposure to the medication, clinical symptoms have appeared 1 day later? In the present case jaundice developed 10 days after ingestion. Symptoms and signs may vary depending on previous exposure to the drug and the time course over which the drug was given. Most patients are jaundiced on admission. Rarely, patients may develop jaundice many days after discontinuation of TMP6 Patients with TMP-SMZ-related hepatic failure typically have markedly elevated transaminase levels. Aspartate aminotransferase and alanine aminotransferase values have ranged from 2000 U/L to 19 520 U/L and 957 U/L to 9400 U/L, respectively (Table 2). Hemorrhagic diathesis and coagulopathy, which were present in our patient, have also been commonly reported. The pathologic findings in the present case were similar to those reported previously. The injury was mainly hepatocellular with massive necrosis of Rappaport zones 2 and 3, and with partial sparing of zone 1 and reticulin collapse with early fibrosis. Mild cholestatic injury and a sparse lymphoplasmacytic and eosinophilic infiltrate were also present. A diffuse mottled pattern of patchy necrosis was grossly evident, similar to the patient described by Colucci and Cicero.6
Other possible causes of hepatic failure were excluded. The cerebrovascular accident was not related to the acute liver failure. Erratic cardiac output has been known to cause acute fulminant hepatic failure; however, this patient's blood pressure was stable throughout her hospital course. There were no signs of cardiogenic shock, and there was no history of heart disease or peripheral vascular disease. Serologic studies for hepatitis B and C were negative. There was no history of acetaminophen abuse prior to initial hospital admission, and the patient took only 1 acetaminophen tablet during her hospital stay. The patient did not have septicemia.
This case of massive hepatic necrosis appears to follow exposure to DPH and TMP-SMZ and was most likely directly due to TMP-SMZ. This conclusion is consistent with the time relationship of administration of TMP-SMZ and the clinical course, as well as with the transaminase increase and the histologic picture of massive centriacinar necrosis. It was felt the patient developed a mild hepatitis from DPH ingestion, which set the stage for further hepatic damage. Ten days following TMP-SMZ administration, she became acutely ill with fever, irritability, somnolence, and lethargy. She was transferred back to the medical ward in hepatic coma and died. This case demonstrates the general need for increased vigilance toward potential chemical-chemical hepatotoxic interactions when administering multiple drugs and specifically illustrates the need for discontinuing TMP-SMZ in the face of early liver injury.
References
1. Olanow CW, Finn AL. Phenytoin: pharmacokinetics and clinical therapeutics. Neurosurgery. 1981;8:112-117.
2. Mullick FG, Ishak KG. Hepatic injury associated with diphenylhydantoin therapy: a clinicopathologic study of 20 cases. AmJ Clin Pathol. 1980;74:442452.
3. Cockerill FR, Edson RS. Trimethoprim-sulfamethoxazole. Mayo Clin Proc. 1991;66:1260-1269.
4. Zimmerman HJ. Update of hepatotoxicity due to classes of drugs in common clinical use: non-steroidal drugs, anti-inflammatory drugs, antibiotics, anti hypertensives, and cardiac and psychotropic agents. Semin Liver Dis. 1990; 10:322-337.
5. Gordin FM, Simon GL, Wofsy CB, Mills JM. Adverse reactions to trimethoprim-sulfamethoxazole in patients with the acquired immunodeficiency syndrome. Ann Intern Med. 1984;100:495-499.
6. Colucci CF, Cicero ML. Hepatic necrosis and trimethoprim-sulfamethoxazole. JAMA. 1975;233:952-953.
7. Ransohoff DF, Jacobs G. Terminal hepatic failure following a small dose of sulfamethoxazole-trimethoprim. Gastroenterology. 1981;80:816-819.
8. Alberti-Flor JJ, Hernandez ME, Ferrer JP, Howell S, Jeffers L. Fulminant liver failure and pancreatitis associated with the use of sulfamethoxazole-trimethoprim. Am J Gastroenterol. 1989;84:1577-1579.
9. Simma B, Meister B, Deutsch J, et al. Fulminant hepatic failure in a child as a potential adverse effect of trimethoprim-sulphamethoxazole. Eur] Pediatr 1995;154:530-533.
10. Sauer JM, Stine ER, Gunawardhana L, Hill DA, Sipes IG. The liver as a target for chemical-chemical interactions. Adv Pharmacol. 1997;43:37-63.
11. Lee TJ, Carney CN, Lapis JL, Higgins T, Fallon HJ. Diphenylhydantoininduced hepatic necrosis. Gastroenterology. 1976;70:422-424.
12. Dhar Gj, Pierach CA, Ahamed PN, Howard RB. Diphenylhydantoin-induced hepatic necrosis. Postgrad Med. 1974;56:128-134.
13. Crawford SE, Jones CK. Fatal liver necrosis and diphenylhydantoin sensitivity. Pediatrics. 1962;30:595-600.
14. Cropper AL. Diphenylhydantoin sensitivity. N Engl J Med. 1956;254: 522-523.
15. Dubois El. Toxic hepatic necrosis associated with taking dilantin, tridione, and phenobarbital: report of a case. Am) Clin Pathol. 1950;20:153-158.
16. Ch'ien LT, Ceballos R, Benton JW. Diphenylhydantoin fatal hepatic necrosis: a review of literature and report of a case treated exchange transfusion. Ala I Med Sci. 1970;7:318-322.
17. Carro JA, Senior J, Rubio CE, Torres EA. Phenytoin induced fatal hepatic injury. Bol Asoc Med P R. 1989;81:359-360.
18. Arky R. Physician's Desk Reference. 53rd ed. Montvale, NJ: Medical Economics Data Production Co; 1999,
19. Tanner AR. Hepatic cholestasis induced by trimethoprime. Br Med J. 1986;293:1072-1073.
Marius J. M Ilario MD Jose E Ruiz MD Constantine A Axiotis MD
Accepted for publication May 18, 2000.
From the Department of Pathology, State University of New York Health Science Center at Brooklyn and The Kings County Hospital Center, Brooklyn, NY.
Reprints: Constantine A. Axiotis, MD, Department of Pathology, Box 25, State University of New York Health Science Center at Brooklyn, 450 Clarkson Ave, Brooklyn, NY 11203.
Copyright College of American Pathologists Dec 2000
Provided by ProQuest Information and Learning Company. All rights Reserved