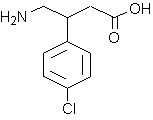
Cough is probably the most common symptom of respiratory disease. Cough associated with acute bacterial respiratory tract infection or chronic obstructive bronchitis facilitates the expulsion of mucus and other secretions from the airways. However, the dry, persistent cough occurring with viral respiratory tract infections, certain chronic pulmonary diseases, or with the use of angiotensin-converting enzyme inhibitors serves no apparent purpose, but causes significant morbidity. The impact of cough on the general population is highlighted by the fact that antitussive agents are the leading category of "over-the-counter" medications sold, accounting for over a half billion dollars in annual sales in the United states.[1]
An increase in the sensitivity of the cough reflex has been proposed as a potential etiology of chronic, nonproductive cough.[1] Often, this symptom is not responsive to standard antitussive therapy. Although codeine may be an effective cough suppressant, its sedating properties, inhibition of gastric motility, and potential for abuse render it an unsatisfactory option for chronic or even intermittent control of bothersome cough. Hence, a nonnarcotic agent with the ability to diminish the sensitivity of the cough reflex (ie, raise the cough threshold) could have significant therapeutic implications.
Gamma-Aminobutyric acid (GABA) is a central inhibitory neurotransmitter that also exists in peripheral tissues, including the lung.[2] The GABA-agonist baclofen has been shown, in animal studies, to inhibit cough via a central site of action,[3] with a potency equal to, or better than, codeine.[4] In this study, we investigated whether low-dose, oral baclofen can inhibit the cough response to inhaled capsaicin in healthy volunteers. The tussive agent capsaicin has been shown, in humans, to induce cough in a reproducible and dose-dependent manner,[5] thereby making it an excellent tool for the evaluation of the effect of a pharmacologic intervention on the cough reflex.
MATERIALS AND METHODS
Subjects
Twenty healthy, nonsmoking volunteers (10 male, 10 female; age range, 26 to 39 years) granted informed consent and participated in the study, which was approved by the Institutional Review Board of the Montefiore Medical Center. Subjects had not experienced symptoms of respiratory tract infection or seasonal allergy for at least 4 weeks prior to enrollment. No medications (other than the study drug) were used for at least 1 week prior to and during the study, except for one participant who continued the use of an oral contraceptive, and another subject who was maintained on a regimen of levothyroxine sodium. All subjects demonstrated normal pulmonary function as assessed by spirometry (KoKo Spirometer; Pulmonary Data Service Instrumentation Inc; Louisville, Colo).
Capsaicin Cough Challenge
Solutions of capsaicin (Sigma Chemical Co; St. Louis) were prepared as previously described.[6] Capsaicin (30.5 ma) was dissolved in ethanol (1 mL) and polyoxyethylenesorbitan mondoleate 80 (Tween 80) (1 mL) and then dissolved in physiologic saline solution (8 mL) to make a stock solution Of [10.sup.-2] M. This solution was further diluted with saline solution to make serial doubling concentrations ranging from 0.98 to 1,000 [micro]mol/L. Fresh dilutions were prepared on each day of testing.
Subjects inhaled single breaths of capsaicin solution from a compressed-air driven nebulizer (model 646; DeVilbiss Health Care Inc; Somerset, Pa) controlled by a dosimeter (KoKo DigiDoser; Pulmonary Data Service Instrumentation Inc). The nebulizer used in this study was modified by the addition of an inspiratory flow regulator valve (Pulmonary Data Service Instrumentation Inc) that regulated inspiratory flow rate to a consistent 0.5 L/s regardless of inspiratory force, thereby guaranteeing a consistent and reproducible amount of solution delivered with each breath. Under these conditions, the output of this nebulizer was determined to be 1.007 mL/min. The duration of aerosol delivery was set at 1.2 s, thereby providing 0.02 mL per breath. Single breaths of capsaicin solution were given in ascending order, with inhalations of saline solution randomly interspersed to increase challenge blindness, until the concentration inducing five or more coughs ([C.sub.5]) was reached. Breaths were delivered at 1-min intervals. The number of coughs in response to each concentration of capsaicin during the 1-min period immediately after each inhalation was recorded by a blinded observer. Subjects were unaware that the end point of the study was the number of coughs induced.
Study Protocol
After undergoing baseline capsaicin cough challenge, subjects were randomly assigned, in a double-blind manner, to take oral baclofen (Zenith Laboratories; Northvale, NJ), 10 ma, or placebo, three fumes daily for 14 days, after which the cough challenge was repeated in an identical manner. During the repeat challenge, doubling concentrations [is greater than] 1,000 [micro]mol/L were used, if necessary, to induce five or more coughs. A significant response was defined as a fourfold or greater increment in [C.sub.5].
RESULTS
Study subjects were evenly distributed between the two groups, whose mean age and gender distribution were similar: 30.6 [+ or -] 1.34 (SEM) years, six male and four female subjects in the baclofen group; and 30.3 [+ or -] 0.92 years, four male and six female subjects in the placebo group. The two groups did not differ with respect to baseline sensitivity to capsaicin (p = 0.178 by unpaired Student's t test). The baclofen group demonstrated a significant elevation of capsaicin cough threshold (pretherapy and posttherapy log [C.sub.5], 0.93 [+ or -] 0.27 and 1.41 [+ or -] 0.36, respectively; p = 0.029 by paired Student's t test). No change in cough sensitivity occurred in the subjects receiving placebo (prestudy and poststudy log [C.sub.5], 1.44 [+ or -] 0.24 and 1.38 [+ or -] 0.26, respectively; p = 0.62). Comparison of the two groups revealed that the change in cough threshold ([Alpha] log [C.sub.5]) after treatment was significantly greater in the baclofen group than in subjects receiving placebo (0.48 [+ or -] 0.19 vs. -0.06 [+ or -] 0.12; p = 0.024 by unpaired Student's t test). Six of 10 subjects receiving baclofen, but none of the members of the placebo group, demonstrated a significant increase (fourfold or greater) in capsaicin cough threshold ([C.sub.5]) (p = 0.0054 by Fisher's Exact Test [Fig 1]).
[Figure 1 ILLUSTRATION OMITTED]
All subjects reported compliance with the study protocol. Four members of the baclofen group and two subjects receiving placebo reported mild fatigue or drowsiness at some time during the initial 3 days of the study.
DISCUSSION
We have demonstrated, in a randomized, double-blind, placebo-controlled study, the ability of low-dose oral baclofen to suppress the cough reflex in healthy volunteers. A 14-day course of baclofen resulted in a significant increase in the cough threshold to inhaled capsaicin. Readers may note that, although the difference is not statistically significant, the mean baseline capsaicin cough threshold in subjects receiving baclofen was lower than that of the placebo group, whereas poststudy values were quite similar. We believe that this issue does not alter the interpretation of the results of this study, for the following reasons. As in the present investigation, previous, larger studies have demonstrated a very wide range in sensitivities to inhaled capsaicin among healthy, asymptomatic volunteers.[7,8] Therefore, an isolated measure of cough sensitivity to capsaicin is of little significance. However, in a specific individual, capsaicin cough threshold is dose-dependent and reproducible over time.[5] Hence, in assessing the effect of a pharmacologic intervention on the sensitivity of the cough reflex, a significant increase in cough threshold over a relatively short period of time is attributable to the investigational agent, with each subject serving as his/her own control. Conversely, a viral upper respiratory tract infection could transiently lower cough threshold. It is noteworthy, therefore, that 6 of 10 subjects receiving baclofen, but none of the 10 subjects receiving placebo, demonstrated a significant (fourfold or greater) increase in capsaicin cough threshold. Furthermore, as shown in Figure 1, subjects with the greatest response to baclofen included those with the lowest, as well as those with the highest, baseline cough thresholds. Therefore, the contention that the baclofen group may have selected out subjects with somewhat lower cough thresholds who were thus more likely to demonstrate a positive effect of the study drug is unfounded.
The GABA-agonist baclofen is a commonly used agent for the relief of muscle spasm, especially in patients with multiple sclerosis and spinal cord injury. The recommended method of baclofen administration is to begin therapy at a low dosage (5 mg three times daily) and to increase gradually until optimal subjective effect is achieved. Most patients attain adequate relief of muscle spasm at daily dosages of 40 to 80 ma, but occasionally larger doses are necessary. The most common adverse reactions associated with baclofen use include transient drowsiness, dizziness, weakness, and fatigue,9 which often subside despite continued use of the drug.
Animal studies have demonstrated the ability of baclofen to inhibit bronchoconstriction induced by various stimuli,[10-12] as well as to suppress both mechanically stimulated and capsaicin-induced cough by a central site of action.[3] The antitussive potency of baclofen in those investigations has been shown to be equal to or greater than that of codeine or dextromethorphan.[4]
Studies investigating the respiratory effects of GABA-agonist therapy in humans are limited. Baclofen, when administered long term, has been shown to inhibit bronchial hyperresponsiveness to methacholine, which results from cervical spinal cord injury.[13] A recent report has described the ability of baclofen to suppress cough due to angiotensin-converting enzyme inhibition.[14] In that study, initial symptomatic relief was noted after 3 to 6 days of baclofen therapy, and maximal antitussive effect was achieved by 5 to 14 days. The 14-day course of baclofen used in the present study was based on e these data.
The proposed mechanism of cough involves the stimulation of two types of receptors within the respiratory tract: rapidly adapting pulmonary stretch receptors (RARs) with thin, myelinated fibers in the vagus nerves, and C-fiber receptors with unmyelinated afferent fibers.[15] RARs are believed to cause cough directly by sending impulses to the brainstem, whereas C-fiber receptors may mediate cough by releasing sensory neuropeptides within the airway, which subsequently activate RARs to cause cough.[15] The antitussive effect of baclofen observed in this study can be explained by central inhibition, as shown in animal studies,[3] but may also involve peripheral inhibitory mechanisms. For example, baclofen has been shown to suppress the release of the protussive neuropeptide, substance P, from primary afferent neurons in rat trachea.[16]
The results of the present study document the antitussive activity of a GABA-agonist in healthy human subjects. The demonstrated efficacy of low-dose oral baclofen supports further investigation of a potential therapeutic use for this drug, or other GABA-agonists, in the treatment of pathologic cough.
REFERENCES
[1] Choudry NB, Fuller RW. Sensitivity of the cough reflex in patients with chronic cough. Eur Respir J 1992; 5:296-300
[2] Ong J, Kerr DIB. GABA-receptors in peripheral tissues. Life Sci 1990; 46:1489-1501
[3] Bolser DC, DeGennaro FC, O'Reilly S, et al. Peripheral and central sites of action of GABA-B agonists to inhibit the cough reflex in the cat and guinea pig. Br J Pharmacol 1994; 113: 1344-48
[4] Chapman RW, Hey JA, Rizzo CA, et al. GABA-B receptors in the lung. Trends Pharmacol Sci 1993; 14:26-9
[5] Midgren B, Hansson L, Karlsson J-A, et al. Capsaicin-induced cough in humans. Am Rev Respir Dis 1992; 146:347-51
[6] Fujimura M, Kamio Y, Hashimoto T, et al. Cough receptor sensitivity and bronchial responsiveness in patients with only chronic nonproductive cough: in view of effect of bronchodilator therapy. J Asthma 1994; 31:463-72
[7] Fujimura M, Kasahara K, Kamio Y, et al. Female gender as a determinant of cough threshold to inhaled capsaicin. Eur Respir J 1996; 9:1624-26
[8] O'Connell F, Thomas VE, Studham JM, et al. Capsaicin cough sensitivity increases during upper respiratory tract infection. Respir Med 1996; 90:279-86
[9] Package insert. Lioresal (baclofen). Ardsley, NY: Ciba-Geigy, December 1988
[10] Chapman RW, Danko G, Rizzo C, et al. Prejunctional GABA-B inhibition of cholinergic, neurally mediated airway contractions in guinea pigs. Pulm Pharmacol 1991; 4:218-24
[11] Belvisi MG, Ichinose M, Barnes PJ. Modulation of nonadrenergic, non-cholinergic neural bronchoconstriction in guinea pig airways via GABA-B receptors. Br J Pharmacol 1989; 97:1225-31
[12] Luzzi S, Franchi-Micheli S, Folco G, et al. Effect of baclofen on different models of bronchial hyperreactivity in the guinea pig. Agents Actions 1987; 20:307-09
[13] Dicpinigaitis PV, Spungen AM, Bauman WA, et al. Inhibition of bronchial hyperresponsiveness by the GABA-agonist baclofen. Chest 1994; 106:758-61
[14] Dicpinigaitis PV. Inhibition of angiotensin-converting enzyme inhibitor-induced cough by the GABA-agonist baclofen [abstract]. Eur Respir J 1995; 8:347S
[15] Widdicombe JG. Neurophysiology of the cough reflex. Eur Respir J 1995; 8:1193-1202
[16] Ray NJ, Jones AJ, Keen P. GABA-B receptor modulation of the release of substance P from capsaicin-sensitive neurones in the rat trachea in vitro. Br J Pharmacol 1991; 102:801-04
(*) From the Department of Medicine, Pulmonary Division, Albert Einstein College of Medicine, Bronx, NY.
Supported by a grant from the American Lung Association of New York.
Presented in part at the Annual Congress of the European Respiratory Society, Stockholm, Sweden, September 8, 1996.
Manuscript received June 27, 1996; revision accepted October 11.
Reprint requests: Dr. Dicpinigaitis, Albert Einstein Hospital, 1825 Eastchester Road, Bronx, NY 10461
COPYRIGHT 1997 American College of Chest Physicians
COPYRIGHT 2004 Gale Group